Translate this page into:
Acute graft versus host disease
Correspondence Address:
Sujay Khandpur
Department of Dermatology, Venereology and Leprology, All India Institute of Medical Sciences (AIIMS), New Delhi - 110 029
India
| How to cite this article: Kumawat MK, Khandpur S. Acute graft versus host disease. Indian J Dermatol Venereol Leprol 2011;77:217-219 |
Sir,
Hematopoietic cell transplantation is the preferred therapy for certain life-threatening diseases of the lympho-hematopoietic system, including acute leukemia′s, aplastic anemia, and certain immunodeficiency diseases. Moderate to severe acute graft versus host disease (GVHD) affects 9-35% patients undergoing allogenic stem cell transplantation despite using human leukocyte antigen (HLA)-matched sibling donors and immunosuppression after grafting. The main targets of acute GVHD in humans are the skin and gastrointestinal tract including the gut and liver, of which the skin is the earliest and the most frequent target. Herein we report a case of acute GVHD presenting as widespread erythematous intensely pruritic macular rash associated with watery to bloody diarrhea and jaundice, following allogenic stem cell transplantation for aplastic anemia.
A 30-year-old patient with severe aplastic anemia underwent allogenic peripheral blood stem cell transplantation from an HLA-identical sibling sister. Following transplantation, he was put on fludarabin 30 mg/m 2 for 5 days, cyclophosphamide 60 mg/kg/day for 2 days, anti-thymocyte globuline (ATG) 30 mg/kg/day for 3 days, inj. cyclosporine-A 1.5 mg/kg/day from day 1 until oral intake was possible and methotrexate 10 mg/kg on day +1 and methotrexate 7 mg/kg on days 3 and 6, as a preparative regimen. Post-transplantation, the course was uneventful for first 30 days. On 31st day, he developed high grade fever followed by pruritic erythematous nonblanchable macular rash over the chest, with watery diarrhea (four to five times/h). During next 3-4 days, the erythematous rash extended to involve the shoulders, back, abdomen, neck and proximal extremities and also involved the palms and soles [Figure - 1]. Oral mucosa was normal. Watery diarrhea changed to bloody diarrhea (8-10 episode/h) with persistence of fever. The patient also developed clinical jaundice and serum bilirubin ranged from 5.8 to 17.3 mg/dl and SGOT/SGPT from 61/103 and 85/155, respectively. A skin biopsy from the erythematous macule revealed focal parakeratosis, dyskeratotic keratinocytes, spongiosis, exocytosis, basal cell damage, pigment incontinence, and dermal lympho-histiocytic infiltrate. Similar changes also involved the hair follicle [Figure - 2]. Endoscopic examination of the GIT showed sloughing of entire mucosa of the second part of duodenum. A diagnosis of acute GVHD involving the skin (Stage 2), duodenum and liver was made and patient initiated on methylprednisolone 2 mg/kg/day, with resolution of the rash with exfoliation and reduction in frequency of stool. However the patient died after 25 days due to sepsis.
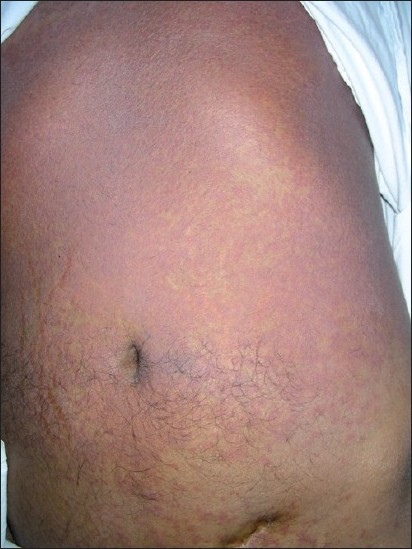 |
| Figure 1: Erythematous macular rash diffusely involving the whole abdomen |
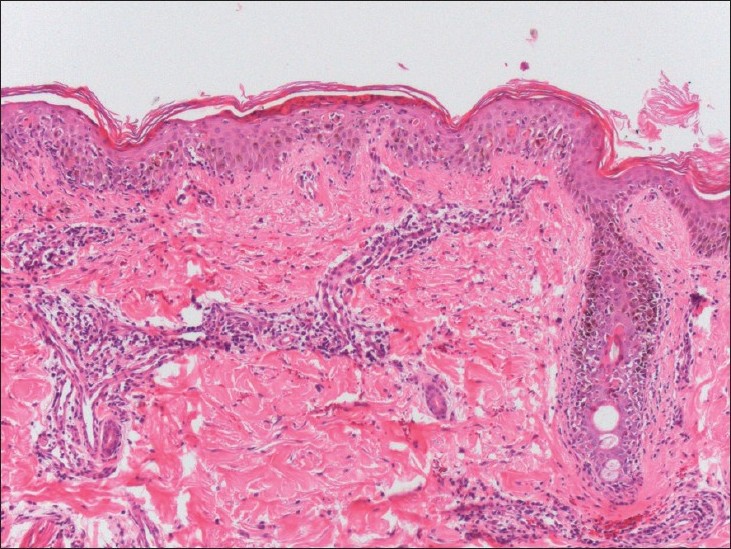 |
| Figure 2: Dermal edema and mucin deposition with lymphocytic vasculitis, consistent with Degos disease (H and E, ×40) |
Barnes and Loutit first described GVHD in mice as a syndrome called "secondary disease" to differentiate it from primary disease of radiation sickness, after observing fatal disease (skin abnormalities and diarrhea) on introducing allogeneic spleen cells. [1] Acute GVHD is a distinctive syndrome of dermatitis, hepatitis and enteritis developing within 100 days of allogeneic hematopoietic-cell transplantation (HCT). Chronic GVHD is a more diverse syndrome developing after day 100. Acute GVHD affect 9-35% patients undergoing standard allogeneic stem cell transplantation despite using HLA-matched sibling donors and immunosuppression after grafting. [2] Allogeneic stem cell transplantation is the most common clinical scenario although it may arise after maternal to fetal transfer of lymphocytes, blood transfusion and solid organ transplantation, most commonly of liver. [3],[4] The skin is the earliest and most frequent target, and involvement begins with faint erythematous macules at any site, often the palms, soles, and pinna. Edema and tenderness often accompany. Pruritus is an uncommon complaint. As the acute GVHD evolves, the distribution of erythematous macules increases, becoming confluent especially over upper back. At a later stage, erythroderma and toxic epidermal necrolysis may ensue. A staging scheme has been proposed based on body surface area (BSA) involvement. Stage 1-cutaneous eruption involving < 25% BSA, Stage 2-cutaneous eruption involving 25-50 % BSA, Stage 3-erythroderma, Stage 4-vesicles and bullae. [5]
Watery diarrhea may develop within days to weeks after the cutaneous lesions, its severity often paralleling skin involvement; it may become bloody and protracted. Liver involvement is expressed clinically by jaundice and tender hepatomegaly. With successful treatment, the eruption resolves with desquamation in the same general progression as its appearance.
In our case, on 31 st day of stem cell transplantation, patient simultaneously developed fever, watery diarrhea and severely pruritic macular erythematous rash over chest which progressively involved other sites. Palmoplantar involvement was not the first feature in our case. Pruritus is an uncommon complaint in acute GVHD but in our case the rash was intensely pruritic. It was associated with diarrhea and clinical jaundice.
The clinical differential diagnosis of acute cutaneous GVHD include drug hypersensitivity eruption, e.g. to cyclosporine or recombinant cytokines, viral exanthem, eruption of lymphocyte recovery, and transient acantholytic dermatosis. A skin biopsy helps to differentiate GVHD from its mimickers. Early changes include focal basal cell vacuolation and sparse superficial perivascular lymphocytic infiltrate with exocytosis into epidermis and follicular epithelium. Established lesions show more pronounced vacuolation, focal spongiosis, lymphocytic infiltration and necrotic epidermis. The grading scheme for the histopathologic features of acute cutaneous GVHD is as follows: Grade 0-normal skin or changes not referable to graft-versus-host disease, Grade 1-basal vacuolization of the dermo-epidermal junction, Grade 2-basal vacuolization, necrotic epidermal cell, lymphocytes in the dermis and/or epidermis, Grade 3-sub-epidermal cleft formation plus grade 2 changes, Grade 4-separation of epidermis from dermis plus grade 2 changes. [6]
Corticosteroids are the mainstay of treatment of acute GVHD. When this approach fails, second line therapy like cyclosporine 2-15 mg/kg/day, mycophenolate mofetil 2 g/day, tacrolimus 4-20 mg/day, ATG 15 mg/kg/day, etanercept 25 mg biweekly, infliximab 10 mg/kg/week, and OKT3 5 mg/day may be required. The overall grade of acute GVHD is predictive of the patient′s outcome, with the highest rates of mortality in those with grade IV GVHD. The response to treatment is also predictive of outcomes. Patients with no response or with progression have a mortality rate as high as 75% compared with 20-25% in those with a complete response.
| 1. |
Barnes DW, Loutit JF, Micklem HS. "Secondary disease" of radiation chimeras: A syndrome due to lymphoid aplasia. Ann N Y Acad Sci 1962;99:374-85.
[Google Scholar]
|
| 2. |
Margolis J, Vogelsang G. An old drug for a new disease: Pentostatin (nipent) in acute graft versus host disease. Semin Oncol 2000;27:72-7.
[Google Scholar]
|
| 3. |
Grogan TM, Brovghton DD, Doyle WF. Graft-versus-host reaction (GVHR): A case report suggesting GVHR occurred as a result of maternofetal cell transfer. Arch Pathol 1975;99:330-4.
[Google Scholar]
|
| 4. |
Schmuth M, Vogel W, Weinlich G. Cutaneous lesions and the presenting sign of acute graft-versus-host disease following liver transplantation. Br J Dermatol 1999;141:901-4.
[Google Scholar]
|
| 5. |
Glucksberg H, Storb R, Fefer A, Buckner CD, Neiman PE, Clift RA, et al. Clinical manifestations of graft-versus host disease in human recipients of marrow from HLA-matched sibling donors. Transplantation 1974;18:295-304.
[Google Scholar]
|
| 6. |
Lerner KG, Kau GF, Storb R. Histopathology of graft versus host reaction in human recipients of marrow from HLA-matched sibling donors. Transplantation 1974;18:367.
[Google Scholar]
|
Fulltext Views
2,209
PDF downloads
1,061





