Translate this page into:
An ambispective cross-sectional descriptive study to characterise the cutaneous and histological features of pediatric Langerhans cell histiocytosis (LCH)
Corresponding author: Dr. Neetu Bhari, Department of Dermatology and Venereology, All India Institute of Medical Sciences, New Delhi, India. drntbhari@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Mondal A, Gaurav V, Khandpur S, Gupta AK, Agarwal S, Seth R, et al. An ambispective cross-sectional descriptive study to characterise the cutaneous and histological features of pediatric Langerhans cell histiocytosis (LCH). Indian J Dermatol Venereol Leprol. doi: 10.25259/IJDVL_1217_2024
Abstract
Background
Langerhans cell histiocytosis (LCH) is an idiopathic disorder characterised by the presence of pathological Langerhans cells derived from myeloid precursor cells. There is a scarcity of data regarding its diverse cutaneous and histological presentations, as well as their prognostic implications.
Objectives
To investigate the clinical and histological features in children with LCH and explore the correlation between the frequency and severity of systemic involvement and various clinical and histopathological findings.
Methods
A two-year ambispective cross-sectional descriptive study was conducted at a tertiary referral centre to identify and describe the clinical and histological features of LCH. Twenty patients (10 prospective and 10 retrospective) were diagnosed based on histopathology.
Results
This study included 20 patients with LCH, majority in the age range of 1-5 years and with male predominance (75%). The primary complaint was fever (100%), accompanied by itching (65%), abdominal pain (45%), or respiratory issues (40%). Scalp involvement was universal, with erythematous papules (95%) being the most common type of lesions. Systemic examination revealed hepatomegaly (65%), splenomegaly (40%), and cervical lymphadenopathy (50%). Histopathological evaluation from varied skin lesions revealed dense dermal infiltrates of Langerhans cells, with papillary dermal oedema and significant RBC extravasation. Immunohistochemistry confirmed Langerhans cells. Systemic evaluation indicated organ involvement in varying percentages.
Limitations
Small sample size and retrospective nature of half of the cases are major limitations of our study.
Conclusion
This study shows skin lesions may develop as crops and fever is a consistent feature found on history or at presentation which may act as important clues towards clinical diagnosis of LCH. In terms of histological features, papillary dermal oedema and RBC extravasation are some prominent findings. Histopathological features does not predict the progression of disease and the extent of systemic involvement.
Keywords
Langerhans cell histiocytosis
paediatric oncology
histopathology
multisystem involvement
immunohistochemistry
Introduction
Langerhans cell histiocytosis (LCH) is an idiopathic disorder characterised by the presence of pathological Langerhans cells (LCs) derived from immature myeloid precursor cells.1-3 There is heterogeneity in the clinical presentation and progression of LCH. It can manifest as a single-system disease that may resolve spontaneously or as a multisystem treatment-resistant disease with a mortality rate of 20%.4 Lesions can be solitary or multiple, and they may remain localised or undergo dissemination.5-7 A histological examination is necessary to confirm diagnosis in case of a clinical suspicion of LCH. The diagnosis can be confirmed by evaluating the expression of CD1a, S100, and CD207 (langerin), or by employing electron microscopy to identify Birbeck granules.8 Limited structured information is available regarding the diverse cutaneous and histological presentations of LCH. In this study, we analysed the cutaneous and histological features of LCH in children and the correlation between histological features and systemic involvement.
Methods
An ambispective, cross-sectional, descriptive study was conducted in the Department of Dermatology at a tertiary care centre in India from January 2014 to May 2023, following approval by the institutional ethics committee (IECPG-327/28.05.21, RT-09/23.06.21). Twenty cases (10 prospective and 10 retrospective) were recruited considering the inclusion and exclusion criteria. All children (age ≤ 18 years) with histopathological diagnosis of LCH from January 2014 to May 2023, whose parents gave consent for participation, were included in this study. Children with a doubtful diagnosis of LCH and cases with inadequate skin tissue samples were excluded. For prospective cases, we screened 15 children suspected of having LCH based on cutaneous examination from June 2021 to May 2023. An active lesion was chosen for the biopsy, and in cases with polymorphic lesions, biopsies were done from morphologically different lesions to increase the diagnostic yield. Five cases showed only spongiotic tissue reaction on histology and were subsequently excluded. The included patients underwent a comprehensive evaluation, including routine tests (complete blood count, liver function test, kidney function test, skeletal survey, and chest X-ray) and Positron emission tomography-computed tomography (PET-CT), if deemed necessary, in consultation with a paediatric oncologist. Histopathological features, including immunohistochemistry (CD1a, langerin, S-100) were analysed by two dermatopathologists independently. For retrospective cases, skin biopsy slides were collected from the Pathology department using the keyword “Langerhans cell histiocytosis”, from year 2014 to 2020. This search yielded 12 cases, and the corresponding biopsy samples were retrieved. Fresh haematoxylin and eosin (H&E) slides were prepared from the residual tissue of these cases and screened. Two cases were excluded from the study due to diagnostic uncertainty in histology. Biopsy forms for the included cases were retrieved from the pathology department and contact information of the patients was obtained using the Unique Health Identification (UHID) system. Out of 10, three patients were able to come and were asymptomatic. Telephonic communication was conducted for the rest seven patients, and the parents were interviewed after obtaining consent to corroborate the clinical information provided in the biopsy forms collected from the pathology department. We were able to retrieve retrospective clinical images from our dermatopathology WhatsApp (Meta, California) group. Finally, the slides were reviewed independently by two aforementioned dermatopathologists [Figure 1]. Statistical analysis was carried out for descriptive statistics, including mean, median, and ranges (IBM SPSS version 27, SPSS Inc., Chicago, IL, USA) and correlation was seen using the scatter plot.
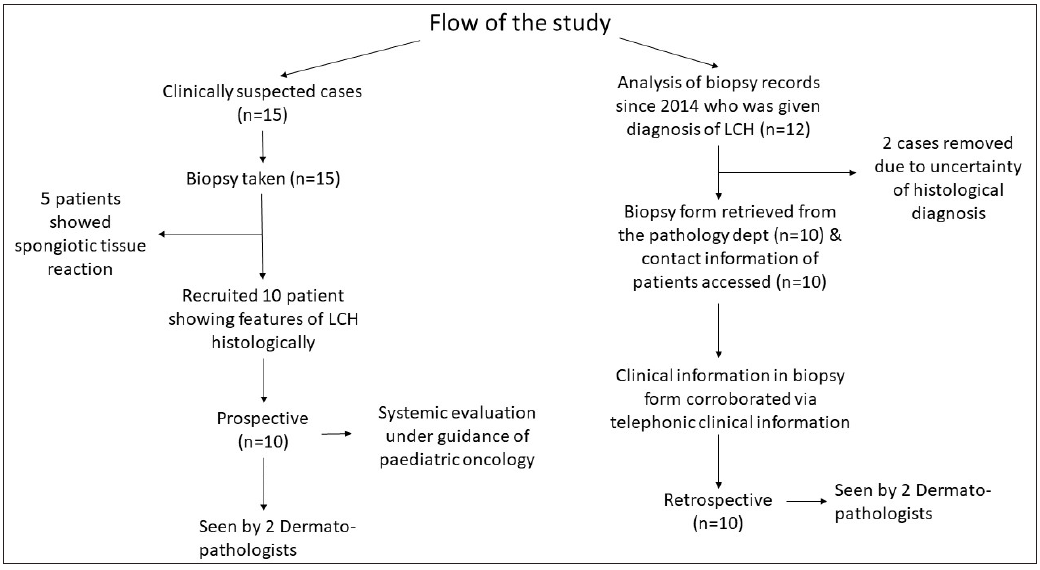
- Flowchart showing the study methodology.
Results
Among the 20 recruited patients (10 prospective and 10 retrospective), 14 (70%) were in the 1-5 years age group with a mean age of 2.4±1.9 years. The male-to-female ratio was 3:1. The age of onset ranged from the 7th day of life to 5.5 years, with a mean age of onset being 1.85±1.5 years. The disease started from the scalp in 12 patients (60%) and from the trunk in 3 (15%), while 5 patients (25%) showed concomitant scalp and trunk involvement at presentation. The mean interval between disease onset and diagnosis was 8 ± 5 months (range 2 months to 1.5 years). The clinical features are presented in Table 1 [Figures 2a to 2f]. Though most of the patients (18; 90%) had erythematous lesions, 10 (50%) had hypopigmented lesions [Figure 2e], and 6 (30%) had purpuric lesions.
| Characteristic | N=20 | |
|---|---|---|
| Symptoms | ||
| Cutaneous |
Itching Pain Oozing Dryness |
13 (65%) 3 (15%) 5 (25%) 2 (10%) |
|
Systemic |
History of fever Abdominal distension and pain Dyspnoea and recurrent cough Watery ear discharge |
20 (100%) 9 (45%) 8 (40%) 6 (30%) |
| Site of involvement |
Scalp Trunk Flexures Palms and soles Perianal region |
20 (100%) 18 (90%) 7 (35%) 2 (10%) 2 (10%) |
| Morphology of lesions |
Papules Pustules Plaques Purpuric Macules Vesicles Ulcer |
19 (95%) 2 (10%) 10 (50%) 2 (10%) 9 (45%) 1 (5%) 2 (10%) |
| Surface changes |
Scaling: Fine loose Fine semi-adherent Fine adherent Coarse adherent Coarse semi-adherent Crusting Fissuring Excoriation |
20 (100%) 3 (15%) 4 (20%) 2 (10%) 5 (25%) 6 (30%) 14 (70%) 3 (15%) 13 (65%) |
| Other sites |
Nail involvement Genital involvement (Scrotum, perineum) Ear involvement |
1 (5%) 2 (10%) 6 (30%) |
| Systemic Examination Findings |
Cervical lymph node enlargement Hepatomegaly Splenomegaly Diminished vesicular breath sounds, crackles, and wheezing Palpable bony swelling over the skull |
10 (50%) 13 (65%) 8 (40%) 4 (20%) 8 (40%) |
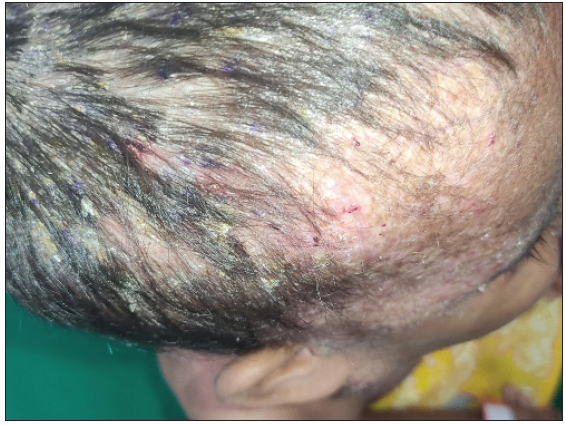
- Scalp involvement in a 3.5 year old female child with multisystem LCH (MS-LCH) in the form of erythematous excoriated papules and yellowish semi-adherent scales.
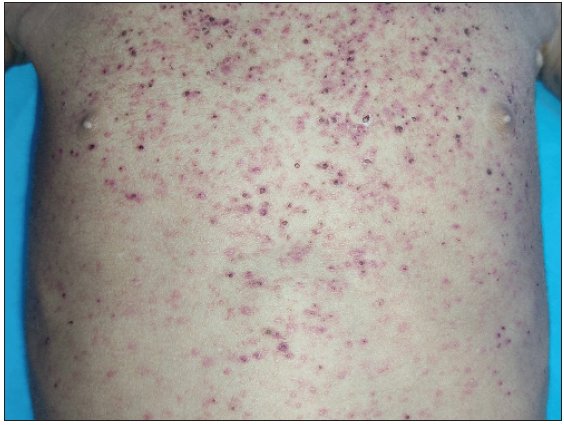
- Discrete to coalescent erythematous and purpuric papules over the chest and abdomen with abdominal distension in a 4-year-old male child with MS-LCH.
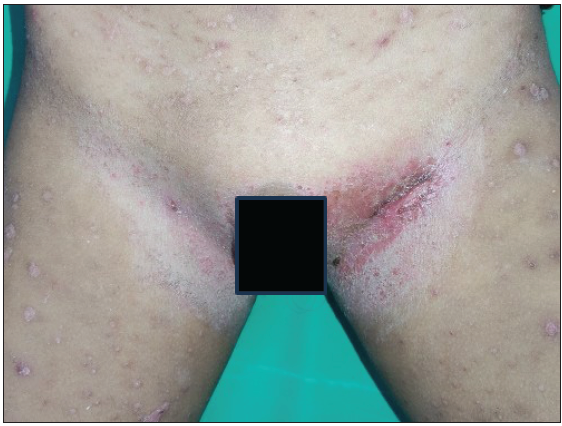
- Linear plaques with fissuring & moist scales over the bilateral inguinal folds in a 5-year-old male child with MS-LCH.
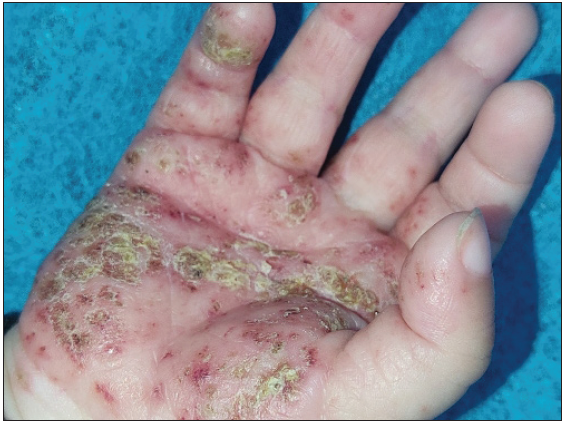
- Erythematous papules and plaque along with a yellowish adherent scale crust seen over the right palm in a 1-year-old male child with MS-LCH.

- Well-defined hypopigmented macules seen over the upper chest in a 2-year-old male child with MS-LCH.
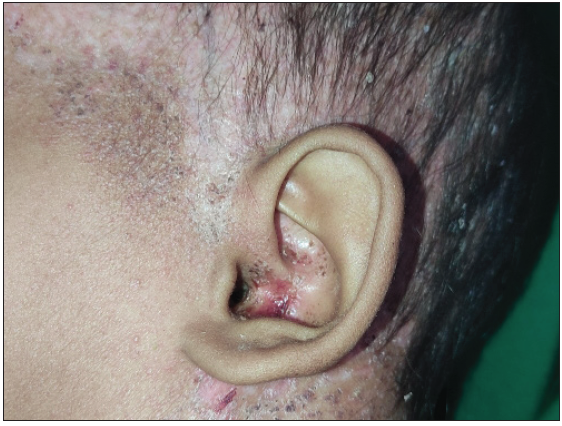
- Ear involvement in the form of erythema, crusting, and depigmentation in a 2.5-year-old male child with MS-LCH.
The histological features are presented in Table 2 [Figures 3a to 3f]. The density of LCs in the dermis was described based on the span of the papillary dermis. Involvement of fewer than 3 dermal papillae or rete pegs was classified as mild, between 3-6 as moderate, and more than 6 as dense. The immunohistochemical staining patterns showed confluent, multifocal, and focal distributions, indicative of the heterogeneous pathological presentation of LCH [Table 3, Figures 4a to 4e].
| Characteristic | N= 20 (100%) | |
|---|---|---|
| Epidermal features | 19 (95%) | |
| Stratum corneum | Compact/basket weave/basket weave to compact | 6 (30%)/9 (45%)/4 (20%) |
| Hyperkeratosis/focal parakeratosis/confluent parakeratosis | 5 (25%)/7 (35%)/7 (35%) | |
| Scale crust (fibrin globules) | 3 (15%) | |
| Epidermis | Normal/atrophic/hyperplastic | 3 (15%)/8 (40%)/8 (40%) |
| Spongiosis; mild/moderate/marked | 12 (75%); 6 (30%)/3 (15%)/6 (30%) | |
| Langerhans cells in epidermis; isolated/clustered/both (isolated + clustered) | 13 (65%); 4 (20%)/6 (30%)/3 (15%) | |
| Epidermal Necrotic keratinocytes | 4 (20%) | |
| Exocytosis (other cells) | 6 (30%) | |
| Atypical mitosis (ring and caterpillar type) | 2 (10%) | |
| Basal layer | Vacuolar (focal) interface dermatitis/focal basal cell degeneration/ confluent basal cell degeneration | 1 (5%)/4 (20%)/14 (70%) |
| Dermal features | 20 (100%) | |
| Infiltrate | Density: Mild (<3 dermal papillae or rete ridges)/moderate (3-6)/dense (>6) | 3 (15%)/2 (10%)/15 (75%) |
| Distribution: Perivascular in upper reticular dermis/interstitial | 2 (10%)/20 (100%) | |
| Disposition: horizontal/both (Horizontal + Vertical) | 17 (85%)/3 (15%) | |
| Location: papillary only/combined (papillary + upper reticular)/extending to deep dermis | 14 (70%)/5 (25%)/1 (5%) | |
| Other cells: neutrophils/eosinophils/lymphocytes/foamy cells/giant cells | 9 (45%)/9 (45%)/15 (75%)/2 (10%)/3 (15%) | |
| Other features | Papillary dermal oedema | 12 (60%) |
| RBC extravasation | 11 (55%) | |
| Fibrosis | 1 (5%) | |
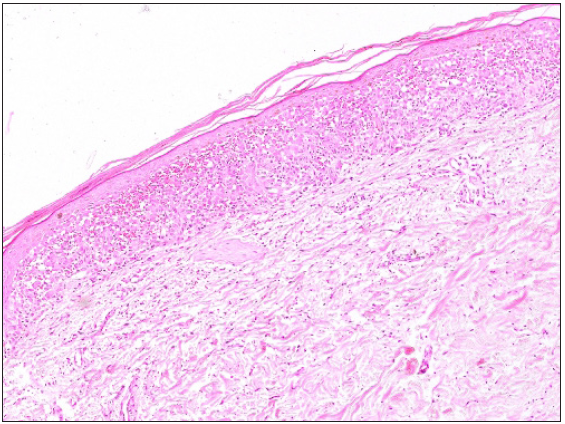
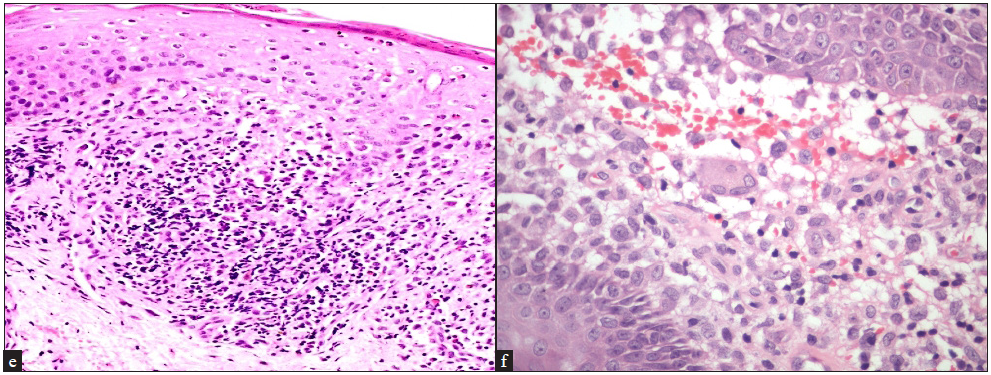
- Focal parakeratosis, basket weave to compact stratum corneum, atrophic epidermis with confluent basal cell damage, dense band-like infiltrate of Langerhans cell in the papillary dermis and significant RBC extravasation (Haematoxylin & eosin, 100x).
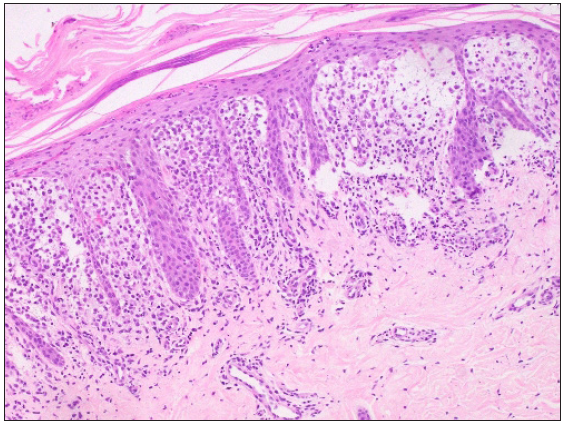
- Compact hyperkeratosis, multifocal parakeratosis, epidermal hyperplasia, dense infiltrate of Langerhans cells in the papillary dermis with marked papillary dermal oedema (Haematoxylin & eosin, 200x) eyepiece 10x and objective 20x (200x).
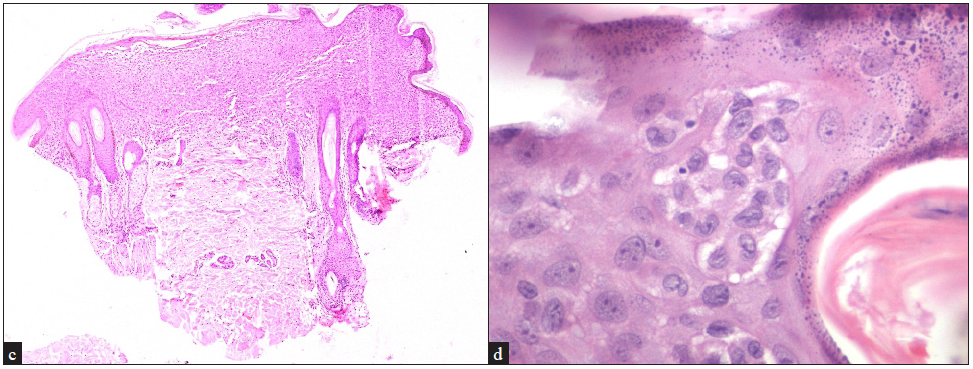
- (c) Confluent (band-like) infiltrate of Langerhans cells in the papillary and upper reticular dermis horizontal disposition with overlying marked basal cell degeneration (Haematoxylin & eosin, 40x), (d) Clustered Langerhans cells in the epidermis beneath the granular layer (Haematoxylin & eosin, 400x).
- (e) Isolated Langerhans cells exocytosis in the basal layer of the epidermis, eosinophilic exocytosis in the upper, mid and lower layers of the epidermis and multiple eosinophils in the upper dermis, (Haematoxylin & eosin, 200x), (f) Langerhans cells forming a giant cell along with significant RBC extravasation and marked oedema (Haematoxylin & eosin, 400x).
| Staining marker | Positive cases | Regions of positivity | Dense infiltrate |
|---|---|---|---|
| CD1a | 20 (100%) | - Both epidermal and dermal: 14 (70%) | 13 (65%) |
| - Predominantly dermal: 6 (30%) | |||
| Langerin | 20 (100%) | - Both regions: 15 (75%) | 10 (50%) |
| S-100 | 18 (90%) | - Both regions: 12 (60%) | 12 (60%) |
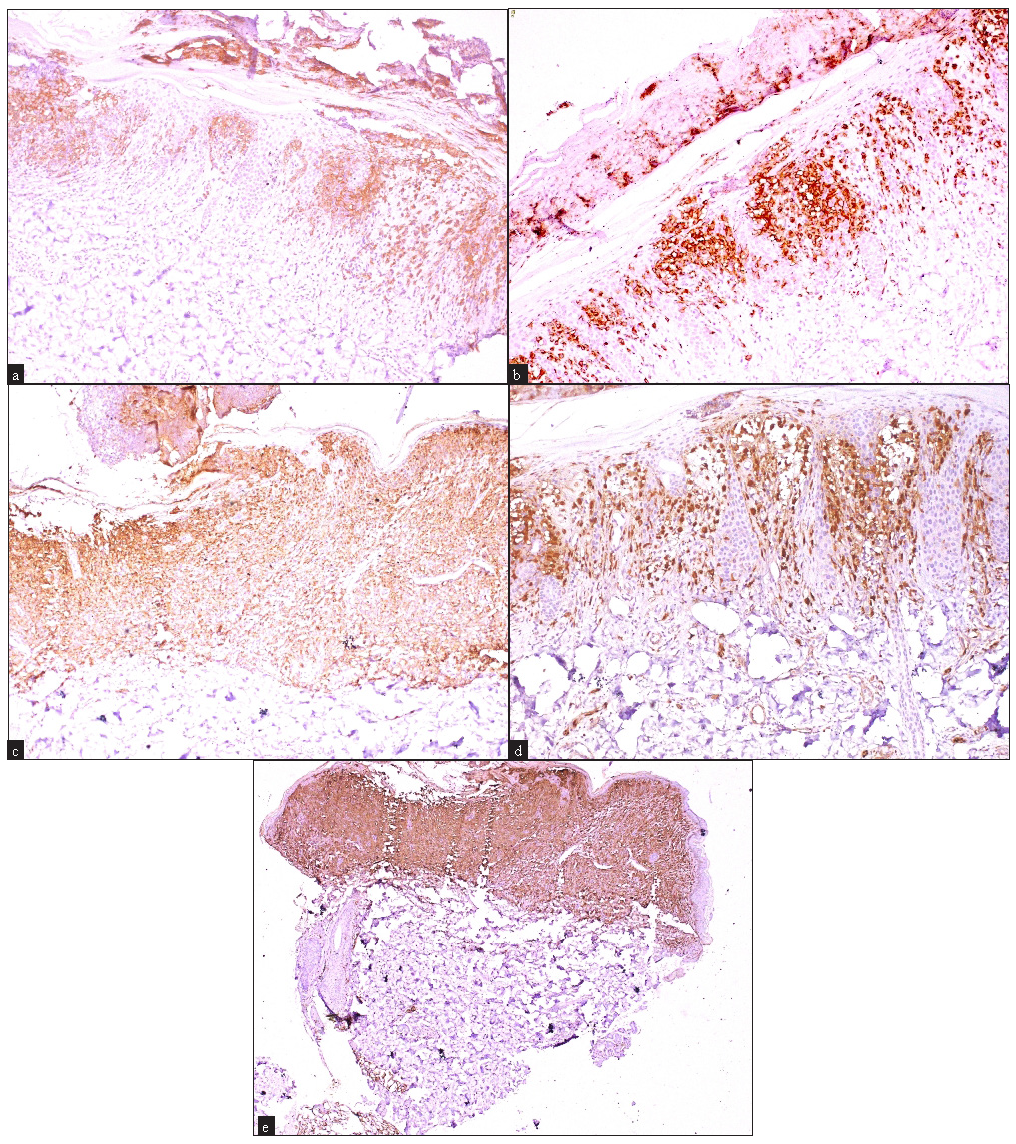
- Immunohistochemistry findings showing (a) patchy (multifocal) epidermal and dermal CD1a positivity (100x), (b) patchy (multifocal) epidermal and dermal langerin positivity (100x), (c) epidermal and dermal confluent and dense langerin positivity in the papillary dermis (100x). (d) patchy (multifocal) epidermal and dermal S-100 positivity (100x) and (e) epidermal and dermal confluent and dense S-100 positivity in the papillary dermis (40x).
Table 4 details the laboratory, radiological, lymph node and bone marrow findings. Post-evaluation, 2 (10%) patients were classified as single-system disease (skin limited SS-LCH), 5 (25%) as multisystemic low-risk disease and 13 (65%) as multisystemic high-risk disease. On analysis, we could not find any correlation between histologic findings and systemic involvement.
| Parameter | Prevalence |
|---|---|
| Hematological findings | |
| - Anaemia (<10 gm%) | 11 (55%) |
| - Pancytopenia | 1 (5%) |
| - Reactive thrombocytosis | 6 (30%) |
| Liver function tests | |
| - Transaminitis | 7 (35%) |
| - Hypoproteinaemia | 4 (20%) |
| Renal function | |
| - Reduced urine osmolality | 5 (25%) (associated with polyuria and polydipsia) |
| Chest X-Ray findings | |
| - Multiple cystic lesions | 4 (20%) |
| - Reticular opacity | 3 (15%) |
| - Multiple tiny nodular opacities | 1 (5%) |
| Skeletal X-Ray findings | |
| - Skull involvement | 10 (50%) |
| - Vertebral involvement | 6 (30%) |
| - Long bone involvement | 3 (15%) |
| HRCT thorax (n=16) | |
| - Ground glass opacity | 7 (35%) |
| - Multiple cystic lesions | 5 (25%) |
| Whole-body PET-CT findings | |
| - Metabolically active cervical lymph nodes | 8 (40%) |
| - Cervical and abdominal lymph nodes | 2 (10%) |
| - Metabolic activity limited to skull | 5 (25%) |
| - Uptake in skull and long bones | 7 (35%) |
| Lymph node and bone marrow studies | |
| - Lymph node aspiration/biopsy (n=9) | 1 (11%) positive for CD1a, langerin, and S-100 |
| - Bone marrow biopsy (n=10) | 2 (20%) suggestive of LCH |
HRCT: High-resolution computed tomography, PET-CT: Positron emission tomography-computed tomography.
Discussion
LCH is a rare disease; the incidence is estimated to occur at 2 to 9 per million children under the age of 15 years, with the highest incidence being observed between the ages of 1 and 3 years.9 Diagnosis is usually delayed, as noted by Uppal et al., where the establishment of diagnosis took an average of 9.4 ± 9.3 months which was longer compared to our patient population (mean 7.9±5.2 months).10
Regarding the systemic symptoms, we noted a history of fever associated with the development of cutaneous lesions in crops in all the patients, indicating a potential relation between cutaneous involvement and pyrexia, a potential clue for the diagnosis. Uppal et al., had mentioned a history of fever or recurrent episodes of fever in 50% of the cases, while respiratory and abdominal complaints were noted in 31% and 12.5 % patients respectively.10
Existing literature shows a similar trend with either the scalp or the trunk being the commonest site of involvement.9,11,12 Battistella et al. noted that flexural involvement (intertrigo) was predominantly seen in the non-self-regressing form of LCH, contrary to the present study where both the patients with flexural involvement had skin-limited LCH.13 In the present study, vesiculo-pustular palmoplantar lesions at initial presentation were seen in one patient of skin-limited LCH, who later progressed to multisystem disease. Palmoplantar lesions have been reported by Battistella et al., and Morren et al., in one patient each with self-healing and multisystem LCH, respectively.11,13
Surface changes in the form of erosions (37.5%) and haemorrhagic crusting were also reported.11 Hypopigmented macules are relatively common in dark-skinned patients with LCH and have been reported as an initial presenting complaint.14 Battistella et al. noted that mono-lesional forms, necrotic lesions, hypopigmented macules, and distal topography of limb lesions were associated with a self-regressive form of the disease.13
Our study did not reveal any case with mucosal involvement, contrary to studies by Battistella et al., Uppal et al., and Morren et al., which have shown mucosal involvement in 9.5%, 6%, and 9.5% of patients, respectively.10,11,13 Similarly, a lesser frequency of nail (1.5%) and ear involvement (6.30%) was seen in our study compared to 18% nail involvement in a study by Uppal et al. and 19% ear involvement by Morren et al.10,11 It has been stated that nail involvement, mucosal lesions, and otitis externa are markers of multisystem disease, though, we did not find any such association.11,15
The presence of large cells with kidney-shaped or reniform vacuolar nuclei with abundant pale eosinophilic cytoplasm is the characteristic histopathological appearance of LCs .12 Battistella et al. reported a milder infiltrate in 43 % of cases and equal involvement of the papillary and reticular dermis.13 A predilection for papillary dermis was also noted by Hussein et al.16 In most of our cases, the infiltrate demonstrated a predominantly interstitial pattern, a horizontal disposition and papillary dermal oedema was a prominent finding in our study.
In contrast to our study, Punia et al., reported eosinophils in all five patients in either clustered form or scattered.17 Battistella et al., mentioned absent to moderate eosinophils in 70% of patients while many to numerous eosinophils in 30%.13 Kapur et al., found many to numerous eosinophils in 31% of patients and noticed a denser eosinophil infiltrate, necrosis, and ulceration in the cases of self-healing disease compared to multisystem disease.18 Plasma cells were not detected in any of our cases, which had been reported as a feature by Ng et al.12 A significantly higher number of giant cells has been reported in previous studies by Hussein et al. (85%), Punia et al. (60%), Kapur et al. (63%) and Widodo et al., (80%) as compared to our study, .16-19 Among these studies, only Kapur et al. noted foamy cells in 36% of cases.18 Significant extravasation of red blood cells (RBCs) was observed in our study, which is considerably higher compared to the 20% reported in a study by Punia et al.17
Epidermal ulceration was a prominent finding in studies done by Punia et al. (20%) and Kapur et al. (52%).17,18 None of those studies mentioned spongiosis, isolated or clustered LCs in the epidermis as histological features. Hussein et al., mentioned epidermal infiltration by LCs in 28% of cases.16 Necrotic keratinocytes in 20% of the cases noted in our study, similar to the study by Kapur et al.18 Basal cell degeneration was a prominent finding in our study which has not been described earlier.
We did not observe any correlation between histopathological features and systemic involvement. This finding aligns with the observations made by Risdall et al., where the authors concluded that histopathological analysis of biopsy material in LCH does not differentiate between the various clinical presentations of the disease and is not useful in predicting its progression.20
Limitations: Small sample size and retrospective nature of half of the cases are major limitations of our study.
Conclusion
This study attempts to provide analysis of the clinical, histopathological, and immunohistochemical features of LCH in Indian children. Notable findings include the significance of fever as an important historical point, occurrence of lesions in crops, and frequent association of fever with cutaneous involvement. Future studies with larger sample sizes and longer follow-ups are essential to unravel the intricacies of the disease and pave the way for advancements in treatment modalities.
Ethical approval
The research/study was approved by the Institutional Review Board at All India Institute of Medical Sciences, New Delhi, number IECPG-277/27.04.2022, RT-18/26.05.2022, dated 27-05-2022.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript, and no images were manipulated using AI.
References
- Progress in understanding the pathogenesis of Langerhans cell histiocytosis: Back to Histiocytosis X? Br J Haematol. 2015;169:3-13.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Expansion of regulatory T cells in patients with Langerhans cell histiocytosis. PLoS Med. 2007;4:e253.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Cell-specific gene expression in Langerhans cell histiocytosis lesions reveals a distinct profile compared with epidermal Langerhans cells. J Immunol. 2010;184:4557-67.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Langerhans cell histiocytosis: Malignancy or inflammatory disorder doing a great job of imitating one? Dis Model Mech. 2009;2:436-9.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The natural history of skin-limited Langerhans cell histiocytosis: A single-institution experience. J Pediatr Hematol Oncol. 2014;36:613-6.
- [PubMed] [Google Scholar]
- Two case report studies of Langerhans cell histiocytosis with an analysis of 918 patients of Langerhans cell histiocytosis in literatures published in China. Int J Dermatol. 2010;49:1169-74.
- [CrossRef] [PubMed] [Google Scholar]
- BRAFV600E mutation in cutaneous lesions of patients with adult Langerhans cell histiocytosis. J Eur Acad Dermatol Venereol. 2015;29:1205-11.
- [CrossRef] [PubMed] [Google Scholar]
- Immunohistochemical analysis of langerin in langerhans cell histiocytosis and pulmonary inflammatory and infectious diseases. Am J Surg Pathol. 2007;31:947-52.
- [CrossRef] [PubMed] [Google Scholar]
- Diverse cutaneous manifestation of Langerhans cell histiocytosis: A 10-year retrospective cohort study. Eur J Pediatr. 2019;178:771-6.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical profile of Langerhans Cell Histiocytosis at a tertiary centre: A prospective study. Indian J Pediatr. 2012;79:1463-7.
- [CrossRef] [PubMed] [Google Scholar]
- Diverse cutaneous presentations of Langerhans cell histiocytosis in children: A retrospective cohort study. Pediatr Blood Cancer. 2016;63:486-92.
- [CrossRef] [PubMed] [Google Scholar]
- Cutaneous Langerhans cell histiocytosis: Study of Asian children shows good overall prognosis. Acta Paediatr. 2013;102:e514-8.
- [CrossRef] [PubMed] [Google Scholar]
- Neonatal and early infantile cutaneous langerhans cell histiocytosis: Comparison of self-regressive and non-self-regressive forms. Arch Dermatol. 2010;146:149-56.
- [CrossRef] [PubMed] [Google Scholar]
- Langerhans cell histiocytosis presenting with hypopigmented macules. Indian J Dermatol Venereol Leprol. 2008;74:670-2.
- [CrossRef] [PubMed] [Google Scholar]
- Nail changes in Langerhans cell histiocytosis: A possible marker of multisystem disease. Pediatr Dermatol. 2008;25:247-51.
- [CrossRef] [PubMed] [Google Scholar]
- Skin-limited Langerhans’ cell histiocytosis in children. Cancer Invest. 2009;27:504-11.
- [CrossRef] [PubMed] [Google Scholar]
- Langerhans cell histiocytosis of skin: A clinicopathologic analysis of five cases. Indian J Dermatol Venereol Leprol. 2006;72:211-4.
- [CrossRef] [PubMed] [Google Scholar]
- Congenital self-healing reticulohistiocytosis (Hashimoto-Pritzker disease): Ten-year experience at Dallas children’s medical center. J Am Acad Dermatol. 2007;56:290-4.
- [CrossRef] [PubMed] [Google Scholar]
- Case series of cutaneous Langerhans cell histiocytosis in Indonesian children; The clinicopathological spectrum. Dermatol Reports. 2020;12:8777.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Histiocytosis X (Langerhans’ cell histiocytosis) Prognostic role of histopathology. Arch Pathol Lab Med. 1983;107:59-63.
- [Google Scholar]







