Translate this page into:
Breast carcinoma arising in ectopic breast tissue presenting as an enlarging axillary nodule
2 Department of Pathology, Massachusetts General Hospital, Boston, MA, USA
3 Department of Dermatology, Massachusetts General Hospital, Boston, MA, USA
Correspondence Address:
Marina Shuster
Harvard Medical School, 89 Park Drive, Apt 2, Boston, MA 02215
USA
| How to cite this article: Shuster M, Tse JY, Smith GP. Breast carcinoma arising in ectopic breast tissue presenting as an enlarging axillary nodule. Indian J Dermatol Venereol Leprol 2015;81:422-424 |
Sir,
A 37-year-old Asian American woman with systemic lupus erythematosus and type IV lupus nephritis on mycophenolate mofetil, hydroxychloroquine and prednisone presented to the department of dermatology at Massachusetts General Hospital, Boston, Massachusetts, with a 2-month history of an enlarging non-tender nodule in the left axilla. The nodule had been present for 2 years but had recently increased in size. Hot compresses recommended by her primary care physician did not reduce the nodule and she was referred to us for further evaluation.
Physical examination revealed a 1.2 cm, firm, rubbery, subcutaneous, semi-mobile nodule [Figure - 1]. The overlying skin was slightly erythematous, but there was no punctum or scale.
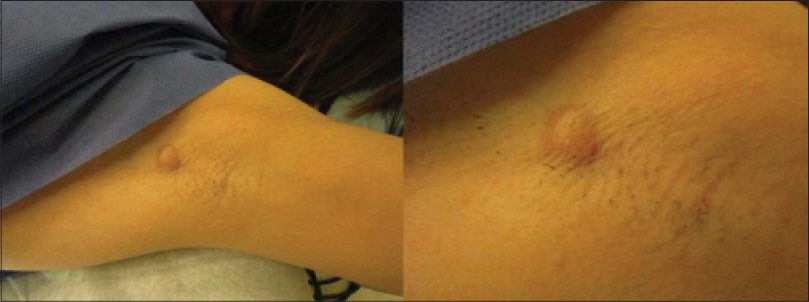 |
| Figure 1: Distant and close up view of 1.2 cm firm, rubbery, subcutaneous semi-mobile nodule in the left axilla |
A punch biopsy of the axillary nodule showed ectopic breast tissue with a focus of invasive ductal carcinoma (grade 1) with intraepidermal involvement (microscopic Paget′s disease) on a background of ductal carcinoma in situ [Figure - 2] and [Figure - 3]. The pattern of immunostaining with calponin, smooth muscle myosin heavy chain, p63, keratin 5/6, and keratin 14 supported the diagnosis of ductal carcinoma of ectopic breast tissue. Keratin 7 immunostain highlighted tumor cells within the dermis and focally in the epidermis. The majority of in situ neoplastic cells stained strongly with Her2, while the invasive component stained weakly.
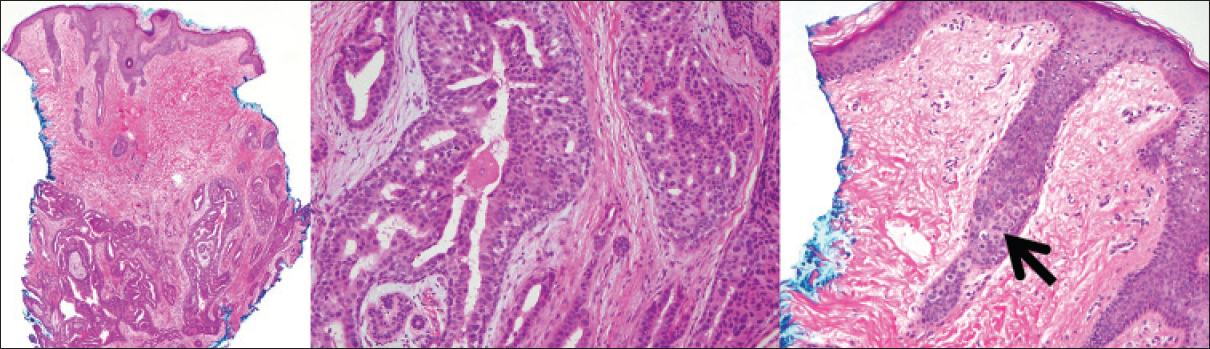 |
| Figure 2: At low power, histologic evaluation of the punch biopsy reveals a dermal lesion composed of large cribriform glands and small infiltrating tubules (left,hematoxylin and eosin, ×40). At high power, the architecture and cytology of the cribriform glands was reminiscent of glandular breast tissue (middle,hematoxylin and eosin, ×200). A high power view of the epidermis overlying the dermal nodule reveals pagetoid spread of carcinoma cells (right, arrow, hematoxylin and eosin, ×200) |
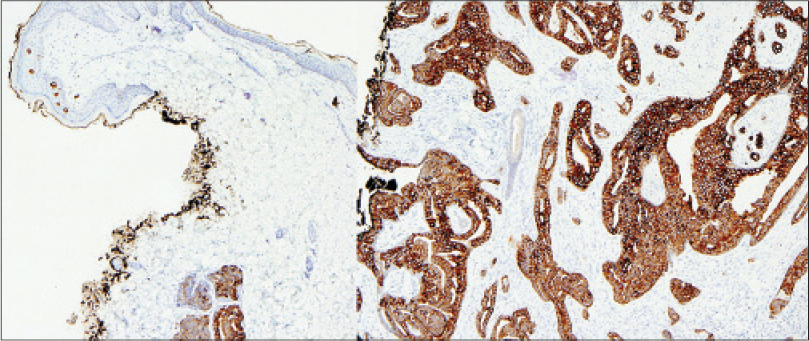 |
| Figure 3: Cytokeratin 7 also highlights the intraepidermal component (microscopic Paget's disease) at the surface of the biopsy (left, ×100). Cytokeratin 7 highlights the ectopic breast tissue in the deep aspect of the skin biopsy, containing in situ and invasive breast carcinoma (right, ×200) |
Further workup revealed suspicious calcifications in the left breast and therefore, a nipple-sparing simple mastectomy was performed along with left axillary sentinel lymph node biopsy and excision of the ectopic breast tissue. Histopathology of excised tissue showed ductal carcinoma in situ in the mastectomy specimen, negative lymph nodes, and a 0.9 cm grade 2 invasive ductal carcinoma (Estrogen Receptor/Progesterone Receptor (ER/PR) positive, Human Epidermal Growth Factor Receptor-2 (HER-2) negative) within the ectopic breast tissue (T1b N0 M0, Stage 1A). The patient was treated with tamoxifen and ovarian suppression with leuprolide. The patient continues to do well and remains disease-free one and a half years later.
Ectopic breast tissue results from improper regression of the milk line, an ectodermal thickening from axilla to groin, formed during fetal development, which regresses to leave behind anatomic breasts. [1] The reported prevalence of ectopic breast tissue ranges from 0.2% to 6%, and is most common in Asian persons. [1]
Ectopic breast tissue is subject to the same potential for malignant transformation as eutopic breast tissue. The incidence of primary ectopic breast carcinoma and rate of malignant transformation of ectopic breast tissue is unknown. [1] Axillary involvement is predominant; axillary ectopic breast carcinoma has been reported to account for 58%, 71% and 91% of all primary ectopic breast carcinomas. [2] It is diagnosed at a median age of 51 years, 10 years earlier than breast carcinoma, despite a 40.5 month average diagnostic delay. [1] About 72% of axillary primary ectopic breast carcinomas are invasive ductal carcinomas while 12% each are invasive lobular carcinomas and medullary carcinomas. [1]
Axillary primary ectopic breast carcinoma commonly presents as a unilateral, non-tender, firm, red, persistent, enlarging subcutaneous/dermal nodule. [1] All such masses warrant strong clinical suspicion for malignant disease. Diagnostic delay may result in increased tumor size and lymph node involvement with resultant higher morbidity and more extensive therapy. [1]
Initial workup of an axillary nodule clinically suspicious for primary ectopic breast carcinoma includes ultrasound imaging and mammogram. [1] Fine needle aspiration or core biopsy may be informative. [3] However, in practice most patients receive definitive diagnosis via excisional biopsy. [2] Primary ectopic breast carcinoma histology is notable for benign breast lobules interspersed with cutaneous adnexal structures and malignant glands, which stain positively for Estrogen receptors (ER), Progesterone receptors (PR) or Gross cystic disease fluid protein-15 (GCDFP-15). [1],[2] Of note, Human Epidermal Growth Factor Receptor-2/NEU (HER-2neu), carcinoembryonic antigen, and cytokeratins can also be expressed in adnexal tumors. [1] The absence of malignancy in eutopic breast tissue also favours the diagnosis of a primary ectopic breast carcinoma. [1]
Once the diagnosis is confirmed on histopathology, lymph node involvement must be evaluated. Primary ectopic breast carcinoma is most likely to spread to the ipsilateral axillary nodes, with subsequent spread to the supraclavicular nodes. [1] The prognosis is similar to a breast carcinoma of comparable stage, with a similar rate of lymph node involvement. [4]
Currently, no guidelines exist for management of this malignancy or the role of prophylactic ectopic breast tissue excision. Patients staged I, II, and IIIA-B should receive radical excision of the malignancy and axillary lymphadenectomy with adjuvant radiotherapy, and if indicated, hormonal treatment and/or chemotherapy. [1] Such an approach was most successful in providing long-term cancer-free survival and mastectomy offered no additional survival benefit to patients with no evidence of disease in eutopic breast tissue. [1],[4] In fact, patients with axillary ectopic breast carcinoma almost never have ipsilateral breast disease, even those with distant metastases. [1],[5]
Our patient′s invasive ductal carcinoma arising within ectopic breast tissue was diagnosed on skin biopsy. Despite annual breast examinations, this nodule was never considered worrisome. This case highlights how strong suspicion on the part of clinicians is integral to the diagnosis of primary ectopic breast carcinoma, and secondly, how it may rarely present with concomitant disease of the eutopic breast, warranting complete evaluation.
| 1. |
Visconti G, Eltahir Y, Van Ginkel RJ, Bart J, Werker PM. Approach andmanagement of primary ectopic breast carcinoma in the axilla: Where are we? A comprehensive historical literature review. J PlastReconstrAesthet Surg 2011;64:e1-11.
[Google Scholar]
|
| 2. |
Nihon-Yanagi Y, Ueda T, Kameda N, Okazumi S. A case of ectopic breast cancer with a literature review. Surg Oncol 2011;20:35-42.
[Google Scholar]
|
| 3. |
Velanovich V. Fine needle aspiration cytology in the diagnosis and management of ectopic breast tissue. Am Surg 1995;61:277-8.
[Google Scholar]
|
| 4. |
Evans DM, Guyton DP. Carcinoma of the axillary breast. J Surg Oncol 195;59:190-5.
[Google Scholar]
|
| 5. |
Cogswell HD, Czerny EW. Carcinoma of aberrant breast of the axilla. Am Surg 1961;27:388-90.
[Google Scholar]
|
Fulltext Views
1,735
PDF downloads
1,008





