Translate this page into:
Childhood onset dermatofibrosarcoma protuberans
Correspondence Address:
B D Sathyanarayana
Department of Skin - STD, Government Medical College, Mysore - 570001, Karnataka
India
| How to cite this article: Sathyanarayana B D. Childhood onset dermatofibrosarcoma protuberans. Indian J Dermatol Venereol Leprol 2004;70:310-312 |
Abstract
Dermatofibrosarcoma protuberans (DFSP) is a rare, slow growing, locally aggressive dermal tumor of disputed histogenesis with a tendency for local recurrence. A 16-year-old female presented with hyperpigmented macules, papules, plaques, erythematous nodules and atrophic, indurated lesions ranging from a few millimeters to a few centimeters. Biopsy revealed thinned out epidermis and a diffuse dermal infiltration by fascicles of spindle cells arranged in storiform pattern. The pseudoseptae formed by the tumor cells interdigitating between the subcutaneous adipocytes resulted in a honeycomb pattern. Histochemical findings are helpful, but the chronicity, size, indolent nature, recurrence after surgery and histopathology are more important to arrive at a diagnosis. Wide and deep surgical excision is the treatment of choice.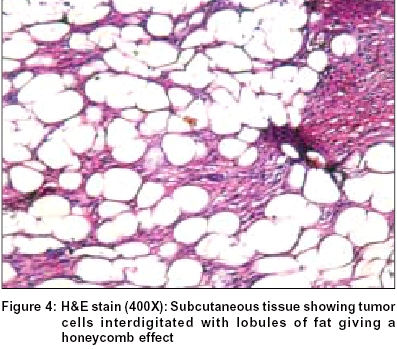 |
 |
 |
 |
 |
 |
 |
 |
INTRODUCTION
Dermatofibrosarcoma protuberans (DFSP) is a rare, slow growing, locally aggressive dermal tumor of disputed histogenesis with a marked tendency for local recurrence.[1],[2] Childhood onset of DFSP is rare but familial and even congenital occurrences are known.[3],[4],[5],[6] We came across a 16 year old girl with this condition who had onset of the disease in infancy.
CASE REPORT
A 16-year-old girl complained of skin lesions on the anteromedial aspect of the upper part of the right thigh since the age of 5 months. It had started as hyperpigmented macules and a raised lesion about 3 cm in diameter that had increased in size and number over years.
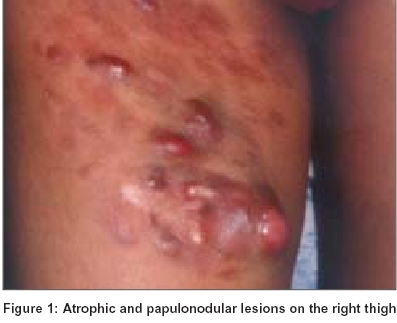
On examination, multiple papules, plaques and nodules varying from a few millimeters to a few centimeters in size were present over the anteromedial aspect of right thigh. Some of these were erythematous, mobile and tender, while the rest were skin colored and adherent to the skin. These were superimposed over several atrophic or indurated plaques in that region. Additionally there were several hyperpigmented atrophic macules and patches in and around the papulonodular lesions; all the lesions covering totally an area of about 12 x 15 cm [Figure - 1].
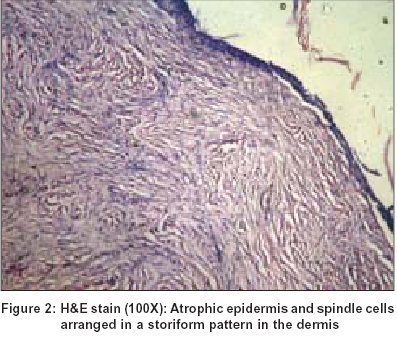
Fine needle aspiration cytology showed benign fibroblastic proliferation. Biopsy revealed thinned out epidermis [Figure - 2] and a diffuse dermal monomorphic infiltration by fascicles of spindle cells arranged in short interweaving (storiform) pattern [Figure - 3]. The pseudoseptae formed by the tumor cells interdigitating between the adipocytes of subcutaneous fat resulted in a honeycomb pattern typical of DFSP [Figure - 4].
DISCUSSION
DFSP lesions occur most commonly on the trunk but may affect any region of the body. They are usually flesh colored, but are occasionally violaceous, blue or red in color. They may present as solitary or multiple polypoid nodules often superimposed over an indurated plaque. The size varies from one to several centimeters in diameter. Atrophic depressed scar like plaque is an uncommon variant. It is difficult to diagnose clinically and is often mistaken for sclerosing basal cell carcinoma, morphoea, anetoderma or scar.[3],[7] The neoplasm is locally invasive and even destructive but distant metastasis is extremely rare. Four early clinical variants of DFSP can exist. These are: i) confluent nodular lesions forming a sclerotic plaque, ii) keloid-like sclerotic plaque, iii) tumor-like, and iv) atrophic plaque form.[3]
Histopathologically, dermatofibrosarcoma protuberans has a monomorphous appearance with thin spindle cells, no or mild nuclear atypia with fewer secondary elements like giant and inflammatory cells than either benign or malignant fibrohistiocytoma. The spindle cells are arranged in short interweaving fascicles giving rise to a storiform appearance. Involvement of subcutaneous tissue with pseudoseptae formation (honeycomb appearance) is a striking feature. Malignant fibrous histiocytoma is not often confused with DFSP because of its rapid growth and is characterized by far more nuclear pleomorphism, mitotic activity, necrosis and its typical deep location affecting muscle.[8] Variable histologic patterns consisting of myxoid, neuroid, fibrosarcomatous, myoid and granular cell types depending upon the tissue admixed in DFSP have been described.[9]
Electron microscopy, tissue culture and histochemical studies of DFSP have led to conflicting observations. Presence of ring chromosome and vimentin in tumor cells are consistent findings.[10] CD-34, the human progenitor cell antigen, is the most important marker; it stains 50 to 100% of the cells. In contrast it stains only a focal area in a small percentage of cases in dermatofibroma and some neural tumors.[11] Factor XIIIa, which stains most dermatofibromas, is only focally positive in small number of cases of DFSP.[12]
Since immunohistochemistry may not be always available, features like chronicity, size, indolent nature, recurrence after surgery and histopathology are very important to reach a diagnosis of DFSP. Wide and deep surgical excision including a minimum 2 to 3 cm of uninvolved skin around the tumor and reaching the superficial fascia is the treatment of choice.[1] Recently, imatinib has been recommended for the treatment of DFSP as a molecularly targeted treatment option for non-resectable tumors and for reducing the recurrence rate after surgery for other cases.[13]
The clinical, histologic and biologic features of congenital, childhood and adult forms of DFSP are similar. To the best of our knowledge, this is the first report of childhood onset atrophic, papulo-nodular DFSP from India.
| 1. |
Garcia C, Clark RE, Buchanan M. Dermatofibrosarcoma protuberans. Int J Dermatol 1996;35:867-71.
[Google Scholar]
|
| 2. |
Gloster HM Jr. Dermatofibrosarcoma protuberans. J Am Acad Dermatol 1996;35:355-74.
[Google Scholar]
|
| 3. |
Martin L, Combemale P, Dupin M, Chouvet B, Kanitakis J, Bouyssou Gauthier ML. The atrophic variant of dermatofibrosarcoma protuberans in childhood: A report of six cases. Br J Dermatol 1998;139:719-25.
[Google Scholar]
|
| 4. |
Gardner TL, Elston DM, Wotowic PJ. A familial dermatofibrosarcoma protuberans. J Am Acad Dermatol 1998;39:504-5.
[Google Scholar]
|
| 5. |
Checketts SR, Hamilton TK, Baughman RD. Congenital and childhood dermatofibrosarcoma protuberans: A case report and review of literature. J Am Acad Dermatol 2000;42:907-13.
[Google Scholar]
|
| 6. |
McKee PH, Fletcher CD. Dermatofibrosarcoma presenting in infancy and childhood. J Cutan Pathol 1991;18:241-6.
[Google Scholar]
|
| 7. |
Davis DA, Sanches RL. Atrophic and plaque like dermatofibrosarcoma protuberans. Am J Dermatopathol 1998;20:498-501.
[Google Scholar]
|
| 8. |
Enzinger FM, Weiss SW. Soft tissue tumors. 3rd Ed. Mosby-Year Book; 1995. p. 325-35.
[Google Scholar]
|
| 9. |
From L, Assad D. Dermatofibrosarcoma protuberans. In: Freedberg IM, Eisen AZ, Wolff K, Austen KF, Goldsmith LA, Katz SI, et al, editors. Dermatology in general medicine. 5th Ed. New York: McGraw-Hill; 1999. p. 1169-70.
[Google Scholar]
|
| 10. |
Bridge JA, Neff JR, Sandberg AA. Cytogenetic analysis of dermatofibrosarcoma protuberans. Cancer Genet Cytogenet 1990;49:199-202.
[Google Scholar]
|
| 11. |
Haycox CL, Odland PB, Olbricht SM, Piepkorn M. Immunohistochemical characterization of dermatofibrosarcoma protuberans with practical application for diagnosis and treatment. J Am Acad Dermatol 1997;37:438-44.
[Google Scholar]
|
| 12. |
Cohen PR, Rapini RP, Farhood Al. Expression of the human hematopoietic progenitor cell antigen CD-34 in dermatofibrosarcoma protuberans, other spindle cell tumors and vascular lesions. J Am Acad Dermatol 1994;30:147-8.
[Google Scholar]
|
| 13. |
McArthur G. Molecularly targeted treatment of dermatofibrosarcoma protuberans. Semin Oncol 2004;31:30-6.
[Google Scholar]
|
Fulltext Views
2,409
PDF downloads
481





