Translate this page into:
Clinicopathological and management outcome in 70 patients with pityriasis rubra pilaris: A retrospective analysis
Corresponding author: Dr. Nermin Karaosmanoğlu, Department of Dermatology, Health Sciences University, Ankara Training and Research Hospital, Ankara, Turkey. nermin_kara@yahoo.com
-
Received: ,
Accepted: ,
How to cite this article: Karaosmanoglu N, Ozdemir Cetinkaya P, Yüksel İ. Clinicopathological and management outcome in 70 patients with pityriasis rubra pilaris: A retrospective analysis. Indian J Dermatol Venereol Leprol. doi: 10.25259/IJDVL_1607_2024
Abstract
Background
Pityriasis rubra pilaris (PRP) is a rare papulosquamous disease with a characteristic clinical appearance, including follicular salmon-coloured papules, which can sometimes progress into erythroderma. Both clinical and histopathological findings are important in diagnosing the disease and should be evaluated together.
Objectives
There are no guidelines for diagnosis and treatment of the disease. The aim of the present study was to contribute to the clinical diagnosis and treatment approach of this rare disease, which could serve as a clinical guide for dermatologists.
Methods
A total of 70 patients diagnosed with PRP in the last 20 years were included in this retrospective study. Clinical, demographic, and histopathologic profiles; and treatment strategies were analysed. Patients were divided into two groups with ‘definite’ or ‘probable’ diagnoses; those diagnosed to have PRP based on both clinical and histopathological findings were classified as having a ‘definite’ diagnosis; and those with histopathologically non-specific features but clinical findings compatible with PRP were classified as having a ‘probable’ diagnosis.
Results
Forty (57.1%) patients were females and 30 (42.9%) were males. Their age ranged from 8 to 79 years (mean age 39.57±18.80 years). The median duration of the disease (before patients received the diagnosis) was 7 months (IQR=24). According to biopsy reports, the most common epidermal changes were hyperkeratosis (78.6%) and focal parakeratosis (75.7%), while the ‘checkerboard pattern,’ which is considered specific for the disease, was detected in only 16 (22.9%) cases. Only 8 biopsy samples (11.4%) had an eosinophilic infiltrate. While 47 (67.1%) patients were classified under ‘definite diagnosis and 23 (32.9%) were classified under ‘probable diagnosis.’ The disease duration was longer in patients with definite diagnosis than in patients with probable diagnosis (p=0.045). A total of 33 (47.14%) patients needed systemic therapy in addition to topical treatment. While 4 of these 33 patients were classified as Type 3, the remaining patients were classified as Type 1, classical adult type. Only three Type 1 (classical adult PRP) patients had erythroderma. Oral retinoids, especially acitretin, were the most preferred and effective treatment. Five patients showed improvement with methotrexate and two needed biological agents.
Limitation
The present study is retrospective and includes a relatively limited number of patients.
Conclusion
PRP is still a rare, difficult-to-manage disease. More studies are required to standardise and improve the diagnosis and treatment approaches for the disease.
Keywords
Pityriasis rubra pilaris
management
acitretin
methotrexate
biologics
Introduction
PRP is a rare papulosquamous disease that may occur at any age.1 The disease has a bimodal distribution in the first and fifth decades and is equally distributed between both genders. Its characteristic clinical appearance includes follicular salmon-coloured papules, which can sometimes progress into erythroderma with islands of spared skin and an accompanying palmoplantar keratoderma.1,2
Griffiths divided PRP into five clinical subsets depending on certain features.3 It was defined with subtitles from type 1 to 5: Type 1 as classical adult type, Type 2 as atypical adult type, Type 3 as classical juvenile type, Type 4 as circumscribed juvenile type, and Type 5 as atypical juvenile type.4 Subsequently, another type (Type 6) was defined as HIV-associated PRP.5 Both clinical and histopathological findings are important in diagnosing the disease and should be evaluated together. Histopathology shows non-specific findings like psoriasiform dermatitis with irregular hyperkeratosis and alternating vertical and horizontal ortho-parakeratosis, specifically called a ‘checkerboard pattern’.6 Treatment strategies are similar to those for psoriasis vulgaris. These options include topical emollients, topical steroids, oral retinoids, immunosuppressants, and biologics.2
PRP is rarely encountered in clinical practice and has delays in diagnosis and treatment due to its clinical and histopathological similarities to psoriasis vulgaris. It is also revealed in the literature that it shares a cytokine network with psoriasis.7 Due to the rarity of the disease and the difficulties experienced in diagnosis, the present literature is mostly limited to case reports and reviews. There are no guidelines for diagnosis and treatment, which makes the standardisation of disease management more difficult.
This study collected and examined 20 years of patient data for PRP. Clinical and histopathological features and treatment strategies were recorded and compared with the current literature. In this way, we aimed to contribute to the clinical diagnosis and treatment approach of this rare disease.
Methods
This retrospective descriptive study consisted of 70 patients, evaluated and diagnosed with PRP in the Department of Dermatology of Ankara Training and Research Hospital over the last 20 years. The local ethics committee approved the study (Date:07.08.2024, No: E-24-179). The study was performed in accordance with the latest version of the ‘Helsinki Declaration’ and ‘Guidelines for Good Clinical Practice.’
All data of PRP patients from the last 20 years were analyzed. Patients’ data were accessed through the hospital’s information processing system and evaluated in detail. Age, sex, accompanying diseases, drug usage, clinical features, subtype of PRP, preliminary diagnoses of skin biopsies, a detailed analysis of histopathological findings, and treatment agents were investigated and recorded. The patients <18 years were categorised under the paediatric group, and all the patients were divided into Griffiths’ types.3 The clinical lesions of the participants were described under subheadings, such as erythematous scaly plaques, either limited or widespread, palmoplantar keratoderma, intertriginous involvement, and erythroderma. The patients were divided into two groups having ‘definite’ or ‘probable’ diagnoses. Specific clinical findings of the disease included salmon-coloured erythematous scaly plaques, orange-red plaques, erythroderma with islands of spared skin, prominent erythema, follicular hyperkeratosis, and palmoplantar keratoderma [see Figures 1 and 2]. Specific histopathological findings included hyperkeratosis, focal parakeratosis, alternating vertical and horizontal parakeratosis, and orthokeratosis (checkerboard pattern) [see Figure 3]. The patients who were diagnosed with PRP based on both clinical and histopathological findings were classified as having a ‘definite’ diagnosis; and those with non-specific histopathological features but PRP-compatible clinical findings were classified as having a ‘probable’ diagnosis. The number of biopsies needed to confirm a definite or certain diagnosis was recorded. Treatment agents, including topical agents (emollients, steroids, D vitamin analogues), conventional systemic agents (retinoids, immunosuppressants), phototherapy, biological agents, or combination therapy were analysed.
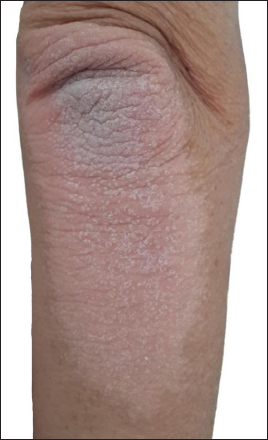
- A salmon-coloured erythematous plaque with follicular hyperkeratosis, located on the elbow.
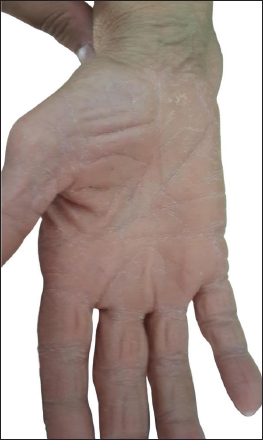
- Palmar keratoderma in a patient with PRP.
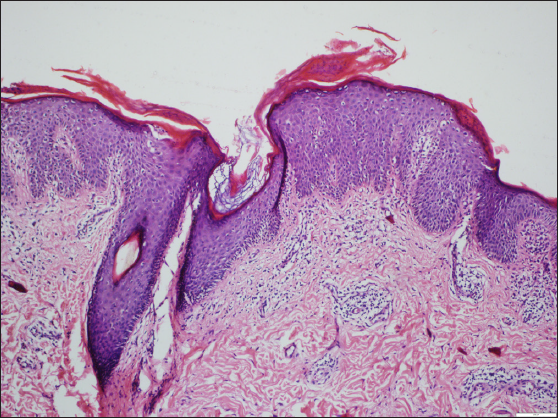
- Acanthosis consisting of thick, blunt, short rete ridges, focal hypergranulosis and shoulder parakeratosis. In addition, there is expansion and keratin plug in the follicle, as well as moderate dermal lymphocytic infiltration. (Haematoxylin and eosin, 100x).
Statistical analysis
All analyses were performed using IBM SPSS Statistics for Windows, Version 20.00 (IBM Corp.), and a p-value of less than 0.05 was considered statistically significant. The normality of the data was tested with the Kolmogorov-Smirnov test. Continuous variables were expressed as mean ± standard deviation and median (minimum-maximum), interquartile range (IQR) with parametric and non-parametric distribution, respectively. Categorical variables were expressed as numbers and percentages. Independent samples were compared using the Student’s t-test and Mann-Whitney U test according to their distribution. The Chi-Square test was employed to compare categorical variables.
Results
A total of 70 patients with PRP diagnosis were included in this study. Forty (57.1%) were female and 30 (42.9%) were male. The age of the patients ranged from 8 to 79 years old, and the mean age was 39.57±18.80 years. The median disease duration (time until patients received diagnosis) was 7 months (IQR=24). While 47 (67.1%) patients were classified with ‘definite diagnosis,’ 23 (32.9%) patients were classified with ‘probable diagnosis.’ The age and sex of patients with definite and probable PRP diagnoses were similar (p>0.05, The Student’s t-test and Chi-Square test were used, respectively). The duration of the disease in patients with definite PRP diagnosis was longer than in patients with probable diagnosis (p=0.045, The Mann-Whitney U test was used).
Fifty-one (72.8%) patients had adult-onset PRP, while 19 (27.2%) patients had juvenile-onset PRP. Among the 19 (27.2%) juvenile patients, one was 8 years old and classified as a paediatric patient with type 3, classical juvenile-onset disease. The remaining patients were adolescents aged 14-18, with all but one diagnosed with type 3 disease; the exception was a patient with type 4, circumscribed juvenile-onset disease. The most common clinical type was Type 1, followed by Type 3. Only one patient presented with a rare and atypical clinical type, as Type 4 disease. Fourteen patients (20%) had an accompanying disease, while 56 (80%) had no diseases. Twelve of 14 patients had multiple accompanying diseases such as hypertension, diabetes mellitus, thyroid diseases, etc. None of the patients had a family history of PRP. The clinical picture of the disease was diverse, most commonly presenting as erythematous scaly plaques on extensor surfaces of the extremities (68.6%), widespread erythematous scaly plaques (67.1%), and palmoplantar keratoderma (30%). Intertriginous involvement and erythroderma were rare presentations detected in four and three patients, respectively. All three patients with erythroderma had Type 1, classical adult PRP. Two had accompanying palmoplantar keratoderma, and all had intertriginous involvement with widespread erythematous scaly plaques on the body and extensor surfaces of extremities. The demographic and clinical characteristics of patients with PRP are presented in Table 1.
| PRP group (n=70) | |
|---|---|
| Sex (n/%) | |
| Female | 40 (57.1%) |
| Male | 30 (42.9%) |
| Age [Mean±SD, years] | 39.57±18.80 |
| Duration of disease [Median, (IQR), months] | 7 (24) |
| Medical history (n/%) | |
| None | 56 (80%) |
| Present | 14 (20%) |
| Hypertension | 1 (1.4%) |
| Diabetes mellitus | 1 (1.4%) |
| Multiple diseases (HT, DM, Thyroid diseases, etc) | 12 (17.1%) |
| Drug use (n/%) | |
| None | 56 (80%) |
| Present | 14 (20%) |
| Anti-hypertensive drug | 1 (1.4%) |
| Oral anti-diabetic drug | 1 (1.4%) |
| Multiple medications (anti-hypertensive, anti-diabetic, anti-hyperlipidemic, etc) | 12 (17.1%) |
| Type of diagnosis: Definite/Probable (n/%) | |
| Definite diagnosis | 47 (67.1%) |
| Probable diagnosis | 23 (32.9%) |
| Clinical presentation for a probable diagnosis (n=23) | 19 (82.6%) |
| Widespread erythematous scaly plaques with an island of sparing | 15 (65.2%) |
| Follicular hyperkeratosis/nutmeg grater sign on erythematous scaly plaques on extensor surfaces of the extremities | |
| PRP subtypes (n/%) | |
| Type 1: Classical adult-onset | 51 (72.8%) |
| Type 3: classical juvenile-onset | 18 (25.8%) |
| Type 4: circumscribed juvenile-onset | 1 (1.4%) |
| Clinical course of PRP (n/%) | |
| Erythematous scaly plaques on extensor surfaces of the extremities | 48 (68.6%) |
| Widespread erythematous scaly plaques | 47 (67.1%) |
| Palmoplantar keratoderma | 21 (30%) |
| Intertriginous involvement | 4 (5.7%) |
| Erythroderma | 3 (4.3%) |
IQR: Interquartile range, PRP: Pityriasis rubra pilaris, SD: Standard deviation, Data were expressed as mean±SD, median, and IQR in continuous variables and n (%) in categorical variables.
Skin biopsies of all patients were included in the study. While a single biopsy was sufficient to diagnose 47 (67.1%) patients, two biopsies were required to definitively diagnose 16 (22.9%) patients, and three skin biopsies were required for 7 patients (10%). The most common preliminary diagnosis was psoriasis (81.4%), followed by mycosis fungoides (25.7%), contact dermatitis (18.6%), palmoplantar keratoderma (10%), and lichen planus (10%). When the biopsy reports were examined in detail, it was found that the most common epidermal changes were hyperkeratosis (78.6%), focal parakeratosis (75.7%), acanthosis (50%), spongiosis (37.1%) and hypergranulosis (34.3%), while the ‘checkerboard pattern,’ which is considered specific for the disease, was detected in only 16 (22.9%) cases. All the patients’ biopsies included perivascular lymphocytic infiltrate. Only eight biopsy samples (11.4%) had an eosinophilic infiltrate, one had neutrophils, and three biopsy specimens included plasma cells [Table 2].
| PRP group (n=70) | |
|---|---|
| Preliminary diagnoses of the skin biopsies (n/%) | |
| Psoriasis | 57 (81.4%) |
| Mycosis fungoides | 18 (25.7%) |
| Contact dermatitis | 13 (18.6%) |
| Palmoplantar keratoderma | 7 (10%) |
| Lichen planus | 7 (10%) |
| Parapsoriasis | 5 (7.1%) |
| Atopic dermatitis | 5 (7.1%) |
| Pityriasis rosea | 5 (7.1%) |
| Dermatophytosis | 5 (7.1%) |
| Maculopapular drug eruption | 4 (5.7%) |
| Pityriasis lichenoides chronic | 3 (4.3%) |
| Keratosis pilaris | 3 (4.3%) |
| Darier disease | 2 (2.9%) |
| Epidermal changes (n/%) | |
| Hyperkeratosis | 55 (78.6%) |
| Focal parakeratosis | 53 (75.7%) |
| Alternating vertical and horizontal parakeratosis and orthokeratosis (checkerboard pattern) | 16 (22.9%) |
| Follicular plug | 7 (10%) |
| Hypergranulosis | 24 (34.3%) |
| Spongiosis | 26 (37.1%) |
| Acanthosis | 35 (50%) |
| Irregular hyperplasia | 13 (18.6%) |
| Dermal changes (n/%) | |
| Perivascular lymphocytic inflammatory cell infiltrate | 70 (100%) |
| Band-like lymphocytic inflammatory cell infiltrate | 0 |
| Types/patterns of inflammatory cells (n/%) | |
| Lymphocytes | 70 (100%) |
| Eosinophils | 8 (11.4%) |
| Neutrophils | 1 (1.4%) |
| Plasma cells | 3 (4.3%) |
PRP: Pityriasis rubra pilaris, Data were expressed as n (%) in categorical variables.
The entire study group received topical therapies as a first choice. The most used topical therapy was corticosteroids, followed by emollients, vitamin D analogues, and retinoids. A total of 33 (47.1%) patients needed systemic therapy in addition to topical therapy. Out of these, the disease was treatment-resistant in eight (11.4%) patients. The treatment agents administered to the patients with PRP are presented in Table 3. Only four of these 33 patients were classified as Type 3, classical juvenile type. Twenty-nine of 33 patients requiring systemic therapy were classified as Type 1, classical adult type. The detailed analysis of PRP subtypes of patients receiving systemic treatment is given in Table 4. Oral retinoids were the first choice for systemic treatment. Twenty patients receiving systemic therapy improved with only acitretin, and three patients improved with only isotretinoin. One patient was not responsive to initial isotretinoin therapy and improved with oral acitretin. Five patients did not respond to initial oral retinoid therapy and improved with methotrexate. Two patients responded to narrow-band UVB therapy alone. Only two patients needed biologics (ustekinumab and infliximab) after receiving acitretin and methotrexate and still had an inadequate clinical response. Of the three patients with erythroderma, one improved with acitretin alone, one with methotrexate after receiving acitretin, and one erythrodermic patient was unresponsive to both conventional agents and improved with ustekinumab. The other patient who required biologic therapy was not erythrodermic but had a generalised and recalcitrant course.
| PRP group (n=70) | |
|---|---|
| Treatment history (n/%) | |
| Topical agents | 70 (100%) |
| Emollients | 43 (61.4%) |
| Topical corticosteroids | 67 (95.7%) |
| Topical vitamin D Analogues | 30 (42.9%) |
| Topical retinoids | 5 (7.1%) |
| Topical and systemic agents | 33 (47.14%) |
| Acitretin | 20 (28.6%) |
| Isotretinoin | 3 (4.3%) |
| Isotretinoin and acitretin | 1 (1.4%) |
| Isotretinoin/acitretin and methotrexate | 5 (7.1%) |
| Narrow-band UVB phototherapy | 2 (2.9%) |
| Methotrexate, acitretin and biological agents (Ustekinumab, Infliximab) | 2 (2.9%) |
PRP: Pityriasis rubra pilaris, Data were expressed as n (%) in categorical variables.
| PRP Subtypes (n/%) | |
|---|---|
| Treatment history (n/%) | |
| Systemic treatments in total, 33 (47.1%) |
Type 1: Classical adult-onset: 29 (41.4%) Type 3: classical juvenile-onset: 4 (5.7%) |
| Acitretin, 20 (28.6%) |
Type 1: Classical adult-onset: 19 (27.2%) Type 3: classical juvenile-onset: 1 (1.4%) |
| Isotretinoin, 3 (4.3%) |
Type 1: Classical adult-onset: 2 (2.9%) Type 3: classical juvenile-onset: 1 (1.4%) |
| Isotretinoin and acitretin, 1 (1.4%) | Type 1: Classical adult-onset: 1 (1.4%) |
| Isotretinoin/acitretin and methotrexate, 5 (7.1%) |
Type 1: Classical adult-onset: 4 (5.7%) Type 3: classical juvenile-onset: 1 (1.4%) |
| Narrow-band phototherapy, 2 (2.9%) |
Type 1: Classical adult-onset: 1 (1.4%) Type 3: classical juvenile-onset: 1 (1.4%) |
| Methotrexate, acitretin and biological agents (Ustekinumab, Infliximab), 2 (2.9%) | Type 1: Classical adult-onset: 2 (2.9%) |
Data were expressed as n (%) in categorical variables.
Discussion
This study presents a large series of patients diagnosed with PRP and provide details of our 20 years of experience. To the best of our knowledge, this is the largest case series, including 70 patients, from our country. When the current literature was reviewed, it was recognised that most publications on PRP consisted of case reports and review articles. Only three retrospective studies have been conducted so far.1,2,4 Due to the insufficient number of studies on this subject, we thought this study would add to the current literature significantly.
In the present study, female patients were predominant (57.1%). This finding differed from the existing data, revealing equal sex distributions.4,8,9 In line with the literature, the disease had a broad age distribution.4,8,9 The disease has two peaks of onset, the first in the first decade and the second in the sixth or seventh decade.4 Contrary to the previous data, this study did not detect the bimodal age-onset distribution pattern. Only one rare clinical subtype was detected and defined as Type 4, a circumscribed juvenile form among all patients. Type 4 PRP has a limited course, characterised by well-defined hyperkeratotic erythematous plaques localised to the elbows and knees with an accompanying palmoplantar keratoderma.10 The patient in the present study had a mild and limited course and responded well to topical corticosteroids and emollients. The average time required to establish a definitive diagnosis of the disease or reach a ‘probable diagnosis’ was 7 months. During this time, approximately 33% of patients required two or three skin biopsies. Not surprisingly, the most common preliminary diagnosis was psoriasis vulgaris, which has similar clinical signs and symptoms to the entity. The difficulties in diagnosing the disease were also emphasised in previous studies. In their retrospective study, Ross et al. categorised the diagnosis into four levels. According to this classification, level 1 was defined as the ‘definitive diagnosis’ together with clinical and histopathological findings, while different levels of a ‘possible diagnosis’ were grouped in levels 2, 3, and 4.4 This classification further supports the fact that a definitive diagnosis cannot always be reached. The diagnosis of PRP is made by evaluating clinical and histopathological findings together, and there is no serological or immunohistochemical marker for assessment, putting it in the group of diseases that are difficult to diagnose.11
Alternating checkerboard orthokeratosis and parakeratosis, psoriasiform hyperplasia, and focal or confluent hypergranulosis are specific histopathologic features of PRP.11 There is usually shoulder parakeratosis, which is the plugging of hair follicles, and parakeratosis adjacent to the plugged follicle. A mild lymphocytic infiltrate may be present, which can be seen as superficial, perivascular, or band-like.12 Acantholysis, which was speculated to be a distinguishing feature of the disease, is a finding in some case reports.12-14 Moreover, it was suggested in one case report that acantholysis could be a sign in diagnosing PRP, which may progress into erythroderma from isolated papulosquamous lesions.14 The presence of eosinophils was also revealed in some cases.1,14,15 In their systematic review of the literature, Sanchez et al. investigated the presence of eosinophils in eosinophil-poor dermatoses such as psoriasis, PRP, lichen sclerosus and dermatomyositis. Among these dermatoses, the percentage of specimens with increased eosinophils in PRP ranged from 22%-63%. The authors concluded that these reports in dermatologic conditions known to be deficient in eosinophils indicate a diagnostic pitfall and suggested that tissue eosinophilia alone should not be used to exclude the diagnosis of one of these conditions.15 In a recent multi-center cohort study, 28 of 142 (19.7%) patients with PRP were reported to have tissue (24/28) and/or peripheral eosinophilia (6/28). In addition, patients with eosinophilia were found to have more likely multi-site disease than patients without eosinophilia. They suggested that the presence of eosinophilia should not exclude PRP diagnosis.16 In this study, hyperkeratosis, focal parakeratosis, acanthosis, spongiosis, hypergranulosis, and checkerboard alternating para- and orthokeratosis were the most common histopathological features, respectively. While acantholysis was not detected in any of the patients, eosinophilia was detected in eight histopathological patients (11.4%) specimens. When the findings of our study and previous literature data are evaluated together, it is seen that diverse histopathological findings of the disease and the possible lack of specific findings in biopsy specimens increase the need for multiple biopsies and the need for correlation with clinical findings to reach diagnoses. Increasing the number of large-scale, multi-center, prospective, and retrospective studies on this subject will facilitate the daily practice of clinicians and prevent loss of time for both the involved parties.
Eighty percent of the patients in the present study did not have any co-morbidities, and the remaining had multiple co-morbidities such as hypertension, diabetes mellitus, hyperlipidemia, and thyroid diseases, which are common in the population. This finding was consistent with most of the reports in the literature, revealing no specific disease association1,2 Interestingly, Ross et al. found an increased incidence of hypothyroidism in their Level 1 (definite diagnosis) patients versus their general population.4 There are several studies in the literature revealing the co-existence of hypothyroidism. Moreover, some of them reported an improvement in PRP after correcting the hypothyroidism.17,18 Although the possible pathogenetic mechanisms between PRP and hypothyroidism are unclear at present, determining a definitive link between the two entities may be important in terms of creating treatment alternatives.
The disease responds to topical treatment, mostly in juvenile-onset forms, and its general course is good.19 This study’s finding that only 4 of 33 patients requiring systemic therapy were Type 3 (classical juvenile-onset) supporting the previous literature. However, in some cases, especially Type 1 classical adult forms, difficulties in treatment may be experienced. Oral retinoids, especially acitretin, are the most effective treatments for PRP.1,2,4,20 Consistent with the literature, acitretin was the most preferred and successful initial therapy in this study. Five patients who did not respond to initial retinoid therapy underwent methotrexate therapy. Methotrexate was reported to be an effective drug in the treatment of PRP.1,2,4,21Contrary to the existing data and our results, some studies reported that methotrexate was ineffective in treating PRP.3,21,22 In their systematic review, Kromer et al. evaluated 182 Pubmed and Cochrane Library studies, reporting any systemic treatments for PRP. They have stated that 42% of all patients who received retinoids responded excellently. Isotretinoin, with a 61.1% achievement was shown to be the most successful retinoid, followed by etretinate at 47% and acitretin at 24.7%. Methotrexate had a 33.1% excellent response rate.23 The authors suggested that the relatively high dose of isotretinoin in patients with PRP may have contributed to its reported effectiveness. Moreover, they revealed a prominently shorter washout phase for isotretinoin (5 weeks) than acitretin (3 years) putting isotretinoin at a more preferable point for women with childbearing potential.21 In a retrospective cohort study, Greenzaid et al.24 evaluated a series of patients treated with methotrexate and acitretin. Of the 24 patients, 12 received only methotrexate, 5 received only acitretin, and 17 underwent combination therapy (methotrexate+acitretin). Complete clearance rates of patients receiving monotherapy were similar. However, nearly half of the patients were unresponsive to methotrexate or acitretin and needed combination therapy. Interestingly, they reported 8 patients who were previously unresponsive to some biologics such as etanercept, ixekizumab, adalimumab and dupilumab and subsequently responded to methotrexate and/or acitretin. The authors revealed that methotrexate and acitretin are more cost-effective, and the high costs of biologics may retain their access.24 The collaborated results of our study and previous data recommend that conventional treatments take precedence over biologics in the treatment algorithm due to their advantages, such as providing high clinical response, achieving good response rates in disseminated and erythrodermic forms, being easily accessible, highly tolerable, cost-effective, and being able to be used orally.
Biologics were reported to be a promising option for PRP patients due to fewer dosing frequencies and a significant improvement in quality of life.25 Kromer et al. reported a 51% success rate with biologics. Among the biologics, ustekinumab was the most effective, followed by infliximab, etanercept and adalimumab. The authors concluded that biologics were more effective than retinoid therapy, with lower and more tolerable side effects.23 A recent retrospective, multicenter study reported that 71% of their cases were erythrodermic, 68% of the patients needed biologic therapy, and ustekinumab was most frequently used.2 In contrast, in our study, only 2 patients needed biologic agents, and only 3 patients were erythrodermic. Based on the findings of our study, it can be recommended that acitretin should be the first choice in systemic treatment while isotretinoin and narrow-band UV-B therapy are the options for women with childbearing potential. Methotrexate should be preferred in cases where acitretin is not successful and safe, and biological agents should be preferred as the third-line treatment. Our recommended algorithm for systemic drug therapy in PRP can be seen in Figure 4.
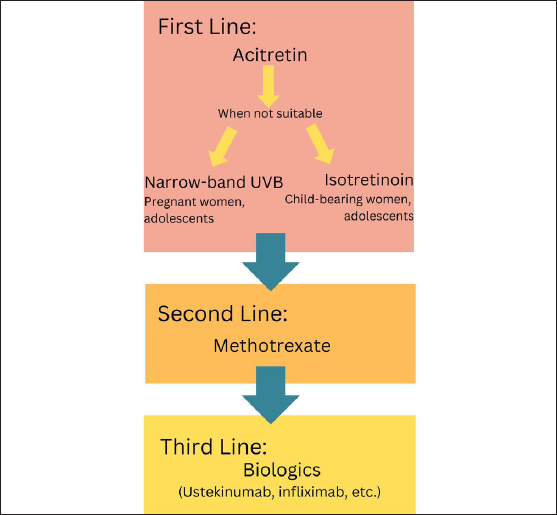
- Recommended algorithm for systemic drug therapy.
Limitation
There are some limitations to this study. Firstly, it was a retrospective study including a relatively limited number of patients. Secondly, due to deficiencies in records, the exact time for the length of the treatment and the detailed dosages of the drugs used could not be analysed. Prospective, comparative, multicenter studies with a larger number of patients are needed for future development.
Conclusion
In conclusion, PRP is a rare and difficult-to-manage disease. There are no definite diagnostic criteria, including clinical, histopathological, and serological findings. At the same time, there are no guidelines for treatment approaches after diagnosis. New studies are required to standardise and improve the diagnosis and treatment approaches for the disease.
Ethical approval
The research/study was approved by the Institutional Review Board at Ankara Training and Research Hospital Ethics Committee, number E-24-179, dated 07.08.2024.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
References
- Pityriasis rubra pilaris: Clinicopathological study of 32 cases from Lebanon. Int J Dermatol. 2014;53:434-9.
- [CrossRef] [PubMed] [Google Scholar]
- Pityriasis rubra pilaris: A multicentric case series of 65 spanish patients. Actas Dermosifiliogr. 2024;115:761-5.
- [CrossRef] [PubMed] [Google Scholar]
- Epidemiologic, clinicopathologic, diagnostic, and management challenges of pityriasis rubra pilaris: A case series of 100 patients. JAMA Dermatol. 2016;152:670-5.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Pityriasis rubra pilaris and human immunodeficiency virus infection--type 6 pityriasis rubra pilaris? Br J Dermatol. 1996;135:1008-9.
- [CrossRef] [PubMed] [Google Scholar]
- Histologic criteria for the diagnosis of pityriasis rubra pilaris. Am J Dermatopathol. 1986;8:277-83.
- [CrossRef] [PubMed] [Google Scholar]
- Pityriasis rubra pilaris as a systemic disease. Clin Dermatol. 2019;37:657-62.
- [CrossRef] [PubMed] [Google Scholar]
- A review on pityriasis rubra pilaris. Am J Clin Dermatol. 2018;19:377-90.
- [CrossRef] [PubMed] [Google Scholar]
- Pityriasis rubra pilaris: Algorithms for diagnosis and treatment. J Eur Acad Dermatol Venereol. 2018;32:889-98.
- [CrossRef] [PubMed] [Google Scholar]
- Circumscribed juvenile pityriasis rubra pilaris (type 4) koebnerising after a hot water burn: mild disease with maximum koebner response. Acta Dermatovenerol Croat. 2019;27:47-9.
- [PubMed] [Google Scholar]
- Early presentation of pityriasis rubra pilaris mimicking tinea corporis: Diagnostic challenges of a rare skin condition. Am J Case Rep. 2022;23:e936906.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Focal acantholytic dyskeratosis occurring in pityriasis rubra pilaris. Am J Dermatopathol. 1989;11:172-6.
- [CrossRef] [PubMed] [Google Scholar]
- Pityriasis rubra pilaris: The clinical context of acantholysis and other histologic features. Int J Dermatol. 2011;50:1480-5.
- [CrossRef] [PubMed] [Google Scholar]
- Eosinophils in traditionally noneosinophil-rich dermatoses. Am J Dermatopathol. 2023;45:820-1.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical features and eosinophilia in pityriasis rubra pilaris: A multicenter cohort. J Am Acad Dermatol. 2022;86:907-9.
- [CrossRef] [PubMed] [Google Scholar]
- Pityriasis Rubra Pilar and hypothyroidism. An Bras Dermatol. 2014;89:497-500.
- [CrossRef] [PubMed] [Google Scholar]
- Pityriasis rubra pilaris and hypothyroidism. Efficacy of thyroid hormone replacement therapy in skin recovery. Br J Dermatol. 2007;156:606-7.
- [CrossRef] [PubMed] [Google Scholar]
- Juvenile onset classical pityriasis rubra pilaris: Every patient may not require systemic therapy. J Coll Physicians Surg Pak. 2007;17:564-5.
- [PubMed] [Google Scholar]
- Pityriasis rubra pilaris: A review of diagnosis and treatment. Am J Clin Dermatol. 2010;11:157-70.
- [CrossRef] [PubMed] [Google Scholar]
- Methotrexate treatment for pityriasis rubra pilaris: A case series and literature review. Acta Derm Venereol. 2018;98:501-5.
- [CrossRef] [PubMed] [Google Scholar]
- Pityriasis rubra pilaris in children. J Am Acad Dermatol. 2002;47:386-9.
- [CrossRef] [PubMed] [Google Scholar]
- Systemic therapies of pityriasis rubra pilaris: A systematic review. J Dtsch Dermatol Ges. 2019;17:243-9.
- [CrossRef] [Google Scholar]
- Methotrexate and acitretin in pityriasis rubra pilaris: A retrospective cohort study. J Am Acad Dermatol. 2024;90:652-4.
- [CrossRef] [PubMed] [Google Scholar]
- Biologics for treatment of pityriasis rubra pilaris: A literature review. J Cutan Med Surg. 2024;28:269-5.
- [CrossRef] [PubMed] [Google Scholar]







