Translate this page into:
Co-existence of extramammary Paget's disease and Bowen's disease of vulva
2 Department of Pathology, Muzaffarnagar Medical College and Hospital, Uttar Pradesh, India
Correspondence Address:
Tarang Goyal
Department of Dermatology, Muzaffarnagar Medical College and Hospital, Muzaffarnagar - 251 201, Uttar Pradesh
India
| How to cite this article: Goyal T, Varshney A, Solanki R. Co-existence of extramammary Paget's disease and Bowen's disease of vulva . Indian J Dermatol Venereol Leprol 2014;80:530-533 |
Abstract
Extramammary Paget's disease and Bowen's disease are histologically similar and immunohistochemistry is often required to make the diagnosis. We present a case of vulval Paget's disease with Bowen's disease in an elderly female. Strong positivity for cytokeratin 7, anti CAM 5.2, carcinoembryonic antigen (CEA) and periodic acid-Schiff (PAS) stain in clitoral, left labial and interface regions of the vulvectomy specimen confirmed the diagnosis of Paget's disease (PD) while positive staining for p63 in the right labial and interface regions helped in establishing the diagnosis of concurrent Bowen's disease (BD).INTRODUCTION
Extramammary Paget′s disease of the vulva is a rare neoplasm accounting for 2% of all vulval malignancies. [1] Dubreuilh [2] described the first case of vulval extramammary Paget′s disease in 1901. Later, a report by Bowen [3] highlighted the histological resemblance between extramammary Paget′s disease and Bowen′s disease. The term ′pagetoid Bowen′s disease′, was first used by Williamson et al.[4] for a histologic variant of squamous cell carcinoma in situ showing nests of pale staining cytoplasmic cells with no immunohistochemical features of Paget′s disease.
It is currently believed that most cases of vulval extramammary Paget′s disease are primary, arising within the epidermis with a very small number associated with cutaneous sweat gland tumors. [5],[6] A new variant of "mammary like" cutaneous glands, which combine morphological features of mammary, eccrine and apocrine glands, are found predominantly in the interlabial sulcus on the vulva. These glands have also been proposed as the site of origin of sweat gland-derived tumors from which extramammary Paget′s disease arises. [7] The most common symptoms are pruritus followed by pain and oozing.
The association of Bowen′s disease of vulva with extramammary Paget′s disease is rare with only a few previous reports of the association.
CASE REPORT
A 57-year-old woman presented with a itchy, eroded plaque over the vulva for two years with a history of rapid progression for 4 months. On examination, there was a single, well defined, hyperpigmented, erythematous, lacy reticular, verrucous plaque on the right labia majora and minora extending to the clitoris and the left labia minora, introitus and perianal regions. There were brightly erythematous moist areas in the clitoral and left labial region [Figure - 1]a. On per speculum examination, the lesion was seen partially extending to the vaginal wall on the right side, but the cervix and left vaginal wall were uninvolved [Figure - 1]b. There was no inguinal adenopathy. Ultrasound and CT imaging of abdomen and pelvis, barium enema, intravenous pyelogram, chest X-ray, mammography, cystoscopy and colonoscopy did not reveal any underlying mass or abnormality. All routine blood investigations were within normal limits. ELISA (enzyme-linked immunosorbent assay) for human immunodeficiency virus (HIV) was non-reactive. A punch biopsy was performed from two sites, the hyperpigmented area and the erythematous area. Features of Bowen′s disease were noted in the hyperpigmented lesion while Pagetoid cells were noted in the erythematous moist area.
 |
| Figure 1: (a) A well defi ned, lacy reticular, hyperpigmented, verrucous looking plaque involving vulval and perianal regions. (b) Per-speculum examination showing involvement of lateral vaginal wall |
Subsequently, the patient was planned for surgical excision and vulvectomy was done under general anesthesia.
Histopathological examination of the entire vulvectomy specimen was undertaken. It showed acanthosis, full thickness cellular atypia, focal hyperkeratosis and parakeratosis with no Paget cells from the right-sided region comprising of labia majora, minora and adjacent areas [Figure - 2]a. On higher power, pleomorphic hyperchromatic keratinocytes typical of Bowen′s disease were noted. The interface between the two clinically different lesions showed basal Pagetoid cells with upper areas showing Bowen′s disease [Figure - 2]b. The left side of the lesion showed Paget′s cells [Figure - 2]c with abundant clear cytoplasm, atypia and nucleolar prominence.
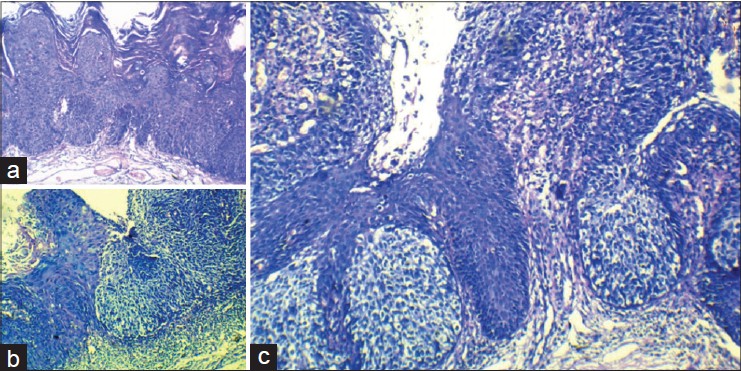 |
| Figure 2: (a) Bowen's disease with acanthosis, full thickness cellular atypia, focal hyperkeratosis and parakeratosis. (H and E, ×10) (b) Interface of Bowen's and Paget's disease (H and E, ×100). (c) Nests of Paget's cells in basal locations in erythematous clitoral and peri-clitoral areas (H and E, ×100) |
The results of immunohistochemical staining are shown in [Figure - 3] and [Figure - 4]. In the right labial regions, p63 and CK 7 were strongly positive in the neoplastic cells in a nuclear and membranous pattern respectively while CK5/6 was weakly positive in a membranous staining pattern. Overall this suggested Bowen′s disease. [Figure - 3] and [Figure - 4]. The immunohistochemistry on the clitoral and left labial regions showed positivity of cytokeratin 7 in a membranous pattern, anti CAM 5.2 and PAS positivity indicating presence of mucin. In addition, CEA was positive indicating that the cells were not Merkel or Toker cells. Thus a diagnosis of Bowen′s disease of vulva with extramammary Paget′s disease in the same lesion, from right to left, with a sharp demarcation was confirmed.
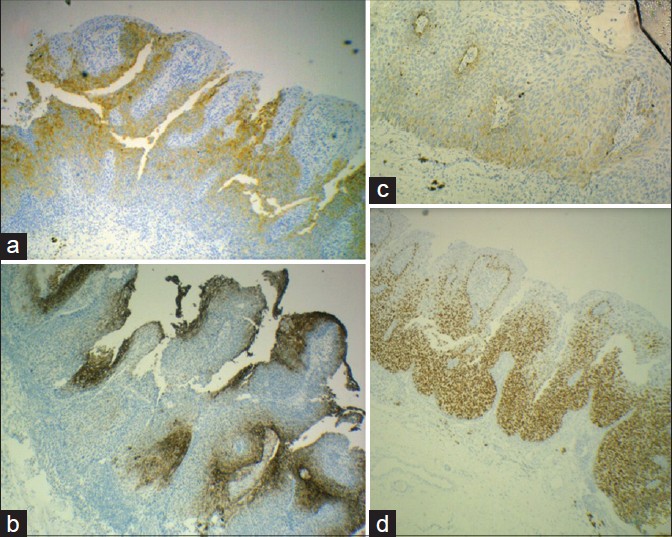 |
| Figure 3: (a) Strong positivity of CK 7 in cells of Bowen's and Paget's disease (×100). (b) Positive carcinoembryonic antigen staining in Paget cells (×100). (c) Weakly positive CK-5/6 in Bowen's disease (×100). (d) Strongly positive p 63 in Bowen's disease (×100) |
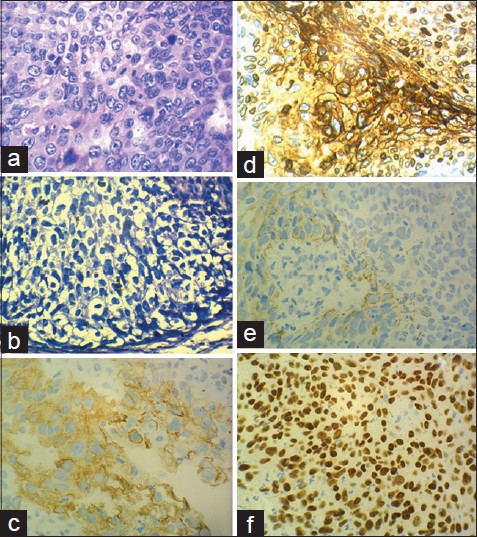 |
| Figure 4: (a) Typical pleomorphic hyperchromatic keratinocytes of Bowen's disease. (H and E, ×400) (b) Paget's cells showing abundant clear cytoplasm, atypia and nucleolar prominence (H and E, ×400). (c) Cytokeratin 7 positivity in a membranous pattern in cells of both Bowen's disease and Paget's disease (×400). (d) Carcinoembryonic positive Paget's cells (×400). (e) CK 5/6 weakly positive cells of Bowen's disease (×400). (F) p63 positive cells of Bowen's disease (×400) |
As there was no lymphadenopathy and imaging studies were normal, lymph nodes were not excised. She is currently under follow-up with no complications or recurrence 6 months after surgery.
DISCUSSION
Our case showed clinical features of Bowen′s disease but the biopsy revealed features of both Paget′s disease and Bowen′s disease. Subsequently, the vulvectomy specimen revealed changes of both these diseases in sharply demarcated areas.
We were able to find only a few previous reports of simultaneous occurrence of vulval extramammary Paget′s disease with Bowen′s disease. [8],[9],[10],[11] The histological findings in these diseases can be overlaping and in such cases, immunohistochemical markers are helpful in differentiating the disorders.
The clinical differential diagnoses of such lesions are tinea cruris, candidiasis, psoriasis, lichen planus, dermatitis, and melanoma. The histopathological differentials for the distinctive pattern of ′pagetoid spread′ throughout the epithelial layer include pagetoid Bowen′s disease, mycosis fungoides, Langerhans cell histiocytosis and Spitz nevus.
In general, Paget cells stain with PAS stain (and are diastase resistant), mucicarmine, Alcian blue at pH 2.5 and colloidal iron. The cells also stain strongly with the immunohistochemical stains, cytokeratin-7 and anticytokeratin CAM 5.2. However, Merkel cells and Toker cells may also show similar staining properties. The CEA, epithelial membrane antigen (EMA), Ber-EP4, BRST-2 and HER-2 neu which are positive for Paget cells but not for Merkel and Toker cells are helpful in differentiation. p63 marker is strongly positive in pagetoid Bowen′s disease but not in Paget′s disease. [12] Further, co-existent Paget′s and Bowen′s disease has to be differentiated also from Bowen′s disease showing the Borst-Jadassohn phenomenon. Lora et al.[13] found that the cells of Bowen′s disease which show clonality and the Borst Jadassohn phenomenon are positive for epidermal growth factor receptors (EGF-R), Ki-67, p53 and p63 and negative for cytokeratin 5/6 and cytokeratin 7. In our case, CK 7 was strongly positive ruling out the Borst Jadassohn phenomenon as the explanation for our findings.
Raju et al.[14] described 2 cases of pagetoid squamous cell carcinoma in situ and showed that CK 7 can be positive in such cases, but added that these would then be non-reactive for GCDFP-15, CEA, CAM-5.2 and c-erbB2. In our case, both CEA and anti CAM-5.2 were positive in the areas showing changes of Paget′s disease indicating the coexistence of these two entities. The proposed origin of Pagets cells in extramammary Paget′s disease from intraepidermal portions of eccrine/apocrine glands or from primitive basal cells within the epidermis is supported by Matsumoto et al. 11 who found that the cells showed CK8 and CK7 positivity.
The treatment of choice remains surgical excision. Radical vulvectomy, radical hemivulvectomy (if localized to one side), wide local excision and Moh′s micrographic surgical excision have all been tried with variable recurrence rates.
For those patients who decline or are unfit for surgery, alternative treatment options include topical chemotherapy and immunomodulators such as 5-fluorouracil (5-FU), bleomycin and imiquimod as adjunctive or primary modalities of treatment. There are some reports of the use of combinations of these topical chemotherapeutic agents with systemic chemotherapy agents including cisplatin, mitomycin C, epirubicin, vincristine and docetaxel. In general, a diagnosis of intraepidermal Paget′s disease with underlying adenocarcinoma, invasive Paget′s disease clitoral location or Paget′s disease with coexisting cancer predicted a poor survival rate. [15] The survival rate was higher with excision with a wide local margin.
ACKNOWLEGMENT
IADVL ACAD-E GROUP for helping with discussions and diagnosis of this case.
| 1. |
Shepherd V, Davidson EJ, Davies-Humphreys J. Extramammary Paget's disease. BJOG 2005;12:273-9.
[Google Scholar]
|
| 2. |
Dubreuilh W. Paget's Disease of the Vulva. Br J Dermatol 1901;13:407-13.
[Google Scholar]
|
| 3. |
Bowen JT. Precancerous dermatosis: A study of two cases of chronic atypical epithelial proliferation. J Cutan Dis 1912;30:241-255.
[Google Scholar]
|
| 4. |
Williamson JD, Colome MI, Sahin A, Ayala AG, Medeiros LJ. Pagetoid bowen disease: A report of 2 cases that express cytokeratin 7. Arch Pathol Lab Med. 2000;124:427-430.
[Google Scholar]
|
| 5. |
Curtin JP, Rubin SC, Jones WB, Hoskins WJ, Lewis JL Jr. Paget's disease of the vulva. Gynecol Oncol 1990;39:374-7.
[Google Scholar]
|
| 6. |
Goldblum JR, Hart WR. Vulvar Paget's disease: A clinicopathologic and immunohistochemical study of 19 cases. Am J Surg Pathol 1997;21:1178-87.
[Google Scholar]
|
| 7. |
van der Putte SC. Anogenital "sweat" glands. Histology and pathology of a gland that may mimic mammary glands. Am J Dermatopathol 1991;13:557-67.
[Google Scholar]
|
| 8. |
Kim SA, Kwon JI, Jung HR, Lee KS, Cho JW. Primary Extramammary Paget's Disease combined with Bowen's disease in vulva. Ann Dermatol 2011;23:S222-5.
[Google Scholar]
|
| 9. |
Parmley TH, Woodruff JD, Julian CG. Invasive vulvar Paget's disease. Obstet Gynecol 1975;46:341-6.
[Google Scholar]
|
| 10. |
Quinn AM, Sienko A, Basrawala Z, Campbell SC. Extramammary Paget disease of the scrotum with features of Bowen disease. Arch Pathol Lab Med 2004;128:84-6.
[Google Scholar]
|
| 11. |
Matsumoto M, Ishiguro M, Ikeno F, Ikeda M, Kamijima R, Hirata Y, et al. Combined Bowen disease and extramammary Paget disease. J Cutan Pathol 2007;34 Suppl 1:47-51.
[Google Scholar]
|
| 12. |
Memezawa A, Okuyama R, Tagami H, Aiba S. p63 constitutes a useful histochemical marker for differentiation of pagetoid Bowen's disease from extramammary Paget's disease. Acta Derm Venereol 2008;88:619-20.
[Google Scholar]
|
| 13. |
Lora V, Chouvet B, Kanitakis J. The "intraepidermal epithelioma" revisited: Immunohistochemical study of the borst-jadassohn phenomenon. Am J Dermatopathol 2011;33:492-7.
[Google Scholar]
|
| 14. |
Raju RR, Goldblum JR, Hart WR. Pagetoid squamous cell carcinoma in situ (pagetoid Bowen's disease) of the external genitalia. Int J Gynecol Pathol 2003;22:127-35.
[Google Scholar]
|
| 15. |
Parker LP, Parker JR, Bodurka-Bevers D, Deavers M, Bevers MW, Shen-Gunther J, et al. Paget's disease of the vulva: Pathology, pattern of involvement and prognosis. Gynecol Oncol 2000;77:183-9.
[Google Scholar]
|
Fulltext Views
2,144
PDF downloads
948





