Translate this page into:
Concomitant psoriasis and hidradenitis suppurativa responsive to adalimumab therapy: A case series
Corresponding author: Prof. Ching-Chi Chi, Department of Dermatology, Chang Gung Memorial Hospital, Linkou, 5, Fuxing St, Guishan Dist, Taoyuan 33305, Taiwan. chingchi@cgmh.org.tw, chingchichi@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Yen CF, Huang YH, Chi CC. Concomitant psoriasis and hidradenitis suppurativa responsive to adalimumab therapy: A case series. Indian J Dermatol Venereol Leprol 2021;87:223-6.
Abstract
Psoriasis and hidradenitis suppurativa are inflammatory dermatoses that have been associated with arthritis, metabolic syndrome, obesity, and smoking. They share common pathogenic mechanisms such as elevated levels of several proinflammatory cytokines including tumor necrosis factor (TNF), interleukin-17A, and impaired Notch pathway. Thus, treatments for both diseases are sometimes overlapping. Biological therapy such as adalimumab is effective for patients with hidradenitis suppurativa and psoriasis. Adalimumab is a monoclonal antibody that binds to TNF and inhibits the cytokine interaction with the TNF receptors, thus inhibiting the inflammatory cascade. Currently, data are lacking on the treatment for co-occurrence of psoriasis and hidradenitis suppurativa. This case series describes three patients with a diagnosis of concomitant psoriasis and hidradenitis suppurativa. In these cases, after 12 weeks of treatment with adalimumab 40 mg every other week, the average Psoriasis Area Severity Index score reduced from 21.4 to 2.9 for psoriasis, Hidradenitis Suppurativa-Physician’s Global Assessment from 3.3 to 0.7, and pain Visual Analog Scale for hidradenitis suppurativa from 4.6 to 2. The results suggest that adalimumab is a treatment of choice for patients with concomitant hidradenitis suppurativa and psoriasis.
Keywords
Adalimumab
hidradenitis suppurative
psoriasis
Introduction
Psoriasis and hidradenitis suppurativa are inflammatory dermatoses in which obesity and smoking have been suggested to play a common role in their pathogenesis.1,2 Also, elevated levels of tumor necrosis factor (TNF) have been found in the blood and lesional skin of psoriasis and hidradenitis suppurativa.1,2 Adalimumab, a biologic targeting TNF, has been approved for treating hidradenitis suppurativa and psoriasis, with different dosing regimens.3 Coexistence of psoriasis and hidradenitis suppurativa has been reported; nonetheless, there are no documented data on the treatment response of concurrent psoriasis and hidradenitis suppurativa to adalimumab therapy.4 Herein, we report three cases with concomitant hidradenitis suppurativa and psoriasis that responded well to adalimumab.
Case Report
Patient 1
A 66-year-old man presented with a history of chronic plaque-type psoriasis, proven by skin biopsy, for 40 years. He was a smoker for 40 years and had a body mass index (BMI) of 21.3. Various treatments were administered for psoriasis [Table 1]. However, the skin condition waxed and waned. In 2016, recurrent painful swollen erythematous nodules and sinuses over the hips developed with pus discharge and hidradenitis suppurativa was diagnosed by clinical examination [Figure 1a]. Oral cefadroxil monohydrate 500 mg and topical gentamicin cream twice daily provided only temporary relief. In 2017, flare up of psoriasis with extensive skin lesions was noticed and he was started treatment with adalimumab (Humira; AbbVie) without combining other conventional treatment. The dosing regimen received was 80 mg administered subcutaneously on day 1, followed by 40 mg on day 8, and then every 2 weeks. After 3 months of adalimumab treatment, the patient’s Psoriasis Area Severity Index (PASI) score was reduced from 28.9 to 5.6, Hidradenitis Suppurativa-Physician’s Global Assessment (HS-PGA) from 4 to 0, and pain Visual Analog Scale (VAS) for HS over hips from 5 to 1 [Figure 1b].
| Patient no./sex/age/BMI | Smoking duration (years) | Previous conventional treatments for psoriasis or psoriatic arthritis | Previous treatments for HS | PASI score before and after adalimumab | HS-PGA before and after adalimumab | Pain VAS for HS before and after adalimumab |
|---|---|---|---|---|---|---|
| 1/male/66/21.3 | 40 | Acitretin 25 mg/day Methotrexate 7.5 mg/week Cyclosporine 200 mg/day NB-UVB phototherapy |
Oral cefadroxil Topical gentamicin |
28.9→5.6 | 4→0 | 5→1 |
| 2/male/57/25.5 | 20 | Methotrexate 15 mg/week Sulfasalazine 1000 mg/day Celecoxib 200 mg/day |
Oral amoxicillin/clavulanate potassium | 23.4→1 | 3→1 | 5→3 |
| 3/male/46/30 | 10 | Leflunomide 20 mg/day Acitretin 10 mg/day Cyclosporine 300 mg/day NB-UVB phototherapy |
Oral cefadroxil | 11.8→2.2 | 3→1 | 4→2 |
BMI: body mass index; NB-UVB: narrow-band ultraviolet B; HS: hidradenitis suppurativa; PASI: Psoriasis Area and Severity Index; HS-PGA: HS-Physician’s Global Assessment; VAS: Visual Analog Scale

- Erythematous nodules with numerous opening of sinuses of hidradenitis suppurativa with heavy silvery scales for psoriasis over bilateral buttocks
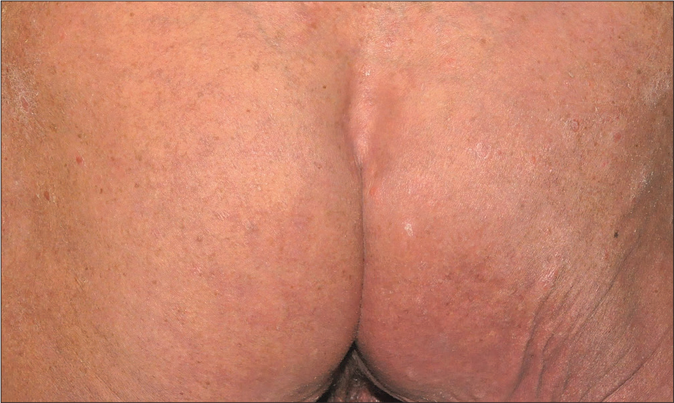
- Improved conditions of hidradenitis suppurativa and psoriasis after adalimumab treatment
Patient 2
A 57-year-old man reported with a history of psoriasis, proven by skin biopsy, for 20 years and psoriatic arthritis with nail onycholysis for 1 year. Moreover, recurrent hidradenitis suppurativa with purulent discharge and sinuses over bilateral hips, diagnosed by clinical examination, occurred over the past 6 months [Figure 2a]. Various treatments were prescribed, but psoriasis and hidradenitis suppurativa were not controlled well [Table 1]. Thus, the patient started to receive adalimumab treatment using the same regimen as case 1 without combining with other conventional treatment. His PASI score was then reduced from 23.4 to 1, tender joint count for psoriatic arthritis from 34 to 18, HS-PGA from 3 to 1, and pain VAS for HS over hips from 5 to 3 [Figure 2b].
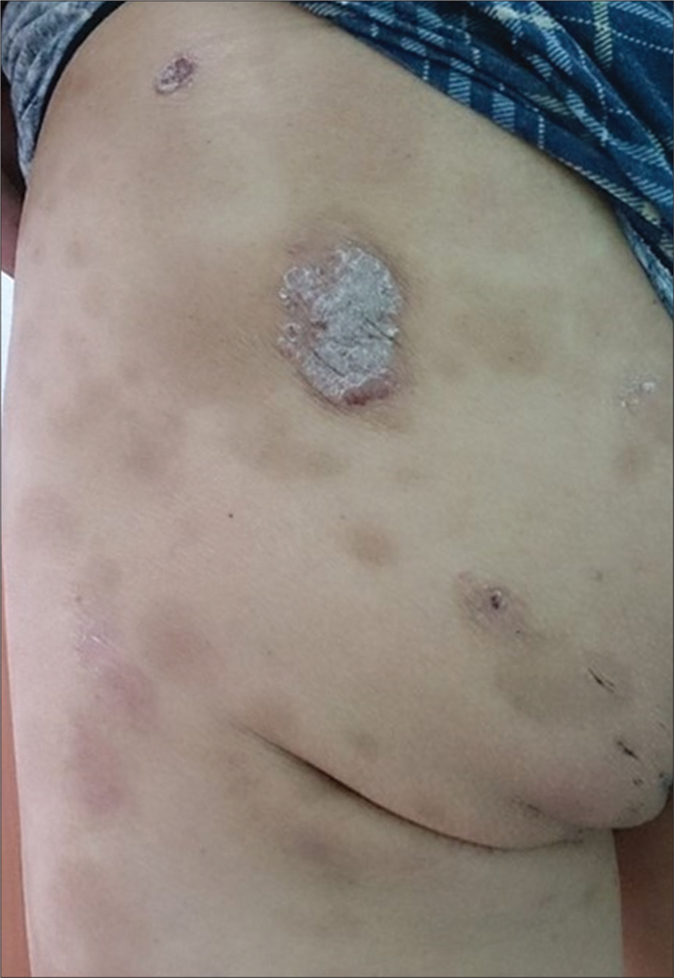
- Plaques covered with heavy silvery scales for psoriasis and numerous opening of sinuses for hidradenitis suppurativa over left buttock
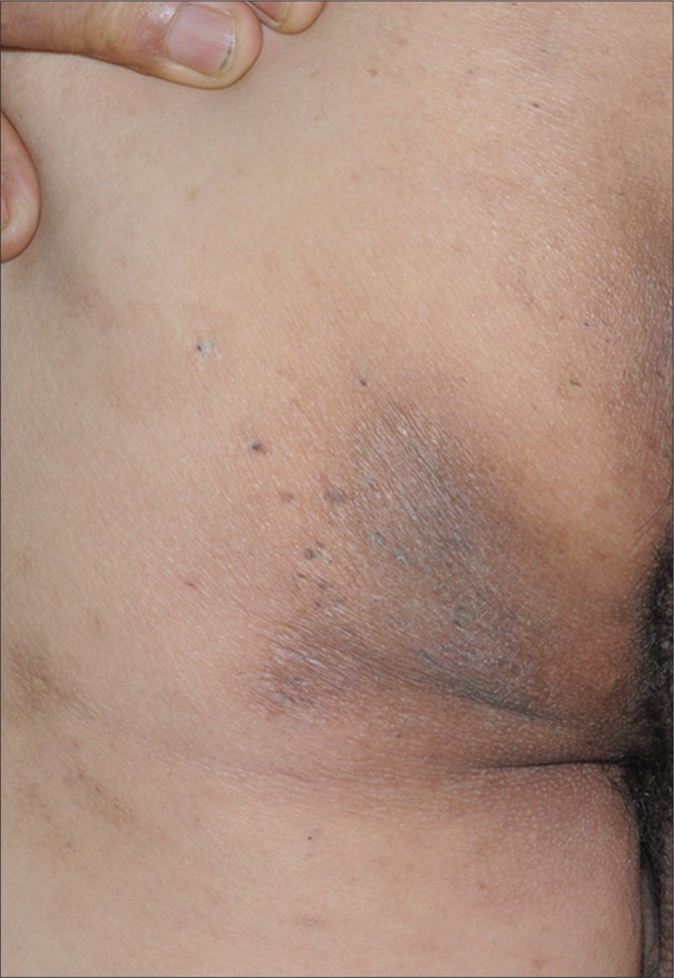
- Improved conditions of psoriasis and hidradenitis suppurativa after adalimumab treatment
Patient 3
A 46-year-old man reported with a history of psoriasis, proven by skin biopsy, for 30 years and psoriatic arthritis for 20 years. He was a smoker for 10 years and had a BMI of 30. Due to poor response to conventional systemic therapy, adalimumab was initiated and continued for 2 years. After adalimumab was discontinued due to reimbursement regulations for half a year, confluent red plaques on the lower limbs and scattered on the trunk were found. Besides, painful erythematous nodules with pus formation appeared over bilateral axillae and hidradenitis suppurativa was diagnosed by clinical examination. Therefore, adalimumab was restarted with the same regimen as case 1 without combining other conventional treatment. After 3 months of adalimumab treatment, the patient’s PASI score was reduced from 11.8 to 2.2, HS-PGA from 3 to 1, and pain VAS for hidradenitis suppurativa from 4 to 2.
Discussion
The prevalence of psoriasis in Caucasians is higher than in the East Asian populations.5 The male-to- female ratio for psoriasis was 1:1 in Caucasians, but was 1.6:1 in East Asians.5 As for hidradenitis suppurativa, women outnumbered men by nearly 3:1 in the United States.6 To date, concomitant psoriasis and hidradenitis suppurativa have been reported mostly in Caucasians.4
A US single-center study reported a female preponderance (>80%) of coexistent psoriasis and hidradenitis suppurtiva.4 Nonetheless, all subjects with concomitant hidradenitis suppurativa and psoriasis in our case series were male. The cause for the gender difference is unclear and may be due to the slightly higher prevalence of psoriasis and hidradenitis suppurativa in men than in women in our institution.
von Laffert et al. reported a higher prevalence and risk of hidradenitis suppurativa in patients with psoriasis than the general population, which may be attributed to their similar pathophysiology.7 In our case series, the prevalence of psoriasis vulgaris in our patients with hidradenitis suppurativa was reported to be 3 (3.8%) patients in our institution (Chang Gung Memorial Hospital Linkou Branch) which was similar to von Laffert et al.7 All our patients presented with psoriasis before the onset of hidradenitis suppurativas, suggesting that the flares of psoriasis with hyperkeratosis could induce follicular occlusion, which is considered the primary event in hidradenitis suppurativa.8
Due to shared pathogenic mechanisms, biological therapy such as adalimumab is effective for hidradenitis suppurativa and psoriasis.3,6 Adalimumab is a monoclonal antibody which binds to TNF and inhibits the cytokine interaction with the TNF receptors, thus aborting the inflammatory cascade. Furthermore, biological therapy, for example, TNF inhibitors, can induce expression changes in the Notch pathway which is suppressed in psoriasis and hidradenitis suppurativa.9 This explains the efficacy of adalimumab in treating psoriasis and hidradenitis suppurativa. Our patients reported great satisfaction with the treatment, with substantial improvement of PASI score from 21.4 to 2.9 for psoriasis, HS-PGA from 3.3 to 0.7, and pain VAS for HS from 4.6 to 2 after 12 weeks of adalimumab treatment.
In our case series, the dosing regimen of adalimumab is indicated for treating psoriasis, which is half of the recommended dosing regimen for HS.6 In a previous study, a greater proportion achieved reduction in hidradenitis suppurativa severity and pain with adalimumab 40 mg weekly dosing compared with every other week.10 However, in our cases, treatment with the dosing regimen for psoriasis achieved significant improvement in reducing hidradenitis suppurativa symptoms. It might be that our patients only had mild to moderate hidradenitis suppurativasymptoms rather than severe conditions. We were unable to find any previous reports of concomitant psoriasis and hidradenitis suppurativa successfully treated with adalimumab.
The limitation of this case series includes that we did not investigate for Crohn’s disease in our patients with perianal lesions.
Conclusion
Psoriasis and hidradenitis suppurativas are both inflammatory skin diseases that share common pathogenic mechanisms. Concurrence of psoriasis and hidradenitis suppurativa may be seen not only in Caucasians but also in Asians. Adalimumab may be the treatment of choice for concomitant psoriasis and hidradenitis suppurativa as we can kill two birds with one stone.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patients have given their consent for their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Psoriasis and smoking: A systematic review and meta-analysis. Br J Dermatol. 2014;170:304-14.
- [CrossRef] [PubMed] [Google Scholar]
- Pathophysiology of hidradenitis suppurativa: An update. J Am Acad Dermatol. 2015;73:S8-11.
- [CrossRef] [PubMed] [Google Scholar]
- British Association of Dermatologists guidelines for biologic therapy for psoriasis 2017. Br J Dermatol. 2017;177:628-36.
- [Google Scholar]
- Epidemiology of concomitant psoriasis and hidradenitis suppurativa (HS): Experience of a tertiary medical center. J Am Acad Dermatol. 2015;73:701-2.
- [CrossRef] [PubMed] [Google Scholar]
- Epidemiology and comorbidities of psoriasis patients in a national database in Taiwan. J Dermatol Sci. 2011;63:40-6.
- [CrossRef] [PubMed] [Google Scholar]
- Interventions for hidradenitis suppurativa: Updated summary of an original cochrane review. JAMA Dermatol. 2017;153:458-9.
- [CrossRef] [PubMed] [Google Scholar]
- Hidradenitis suppurativa/acne inversa: Bilocated epithelial hyperplasia with very different sequelae. Br J Dermatol. 2011;164:367-71.
- [CrossRef] [PubMed] [Google Scholar]
- Hidradenitis suppurativa: A disease of follicular epithelium, rather than apocrine glands. Br J Dermatol. 1990;122:763-9.
- [CrossRef] [PubMed] [Google Scholar]
- Biological therapy induces expression changes in Notch pathway in psoriasis. Arch Dermatol Res. 2015;307:863-73.
- [CrossRef] [PubMed] [Google Scholar]
- Adalimumab treatment in women with moderate-to-severe hidradenitis suppurativa from the placebo-controlled portion of a phase 2, randomized, double-blind study. J Drugs Dermatol. 2016;15:1192-6.
- [Google Scholar]






