Translate this page into:
Cutaneous seeding of melanoma by complete lymph node dissection
Corresponding author: Dr. Jee-Woong Choi, Department of Dermatology, Ajou University Hospital, Ajou University School of Medicine, Suwon, South Korea. dermaboy@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Choi JW, Yoon J, Kim. Cutaneous seeding of melanoma by complete lymph node dissection. Indian J Dermatol Venereol Leprol 2021;87:685-7.
Sir,
The surgical management of regional metastasis to lymph nodes is critical in the survival of patients with melanoma. However, there is a theoretical risk of tumor seeding that can occur as a complication of the surgical management of regional metastasis to the lymph nodes. Here, we present a patient who developed cutaneous seeding of melanoma by complete lymph node dissection.
A 57-year-old female came to our clinic for pigmentation on the linear scar line of her right groin. Physical examination revealed several well-demarcated, black macules along the linear scar line [Figure 1]. Dermoscopic examination of the pigmented macules was performed [Figure 1; black arrow] revealing dark brown irregular streaks confined to the linear scar line [Figure 2, ×10]. One year previously, she had undergone total excision of the nail unit followed by a full-thickness skin graft of her right big toe for acral lentiginous melanoma with a Breslow thickness of 2.4 mm, lymphovascular invasion and ulceration [Figure 3]. A sentinel lymph node biopsy (SLNB) was performed at the ipsilateral groin and three lymph nodes were positive. As the sentinel lymph node was positive, complete lymph node dissection (CLND) was done. The biopsy of the pigmented macule on the scar, developed after CLND, revealed melanoma [Figure 4].
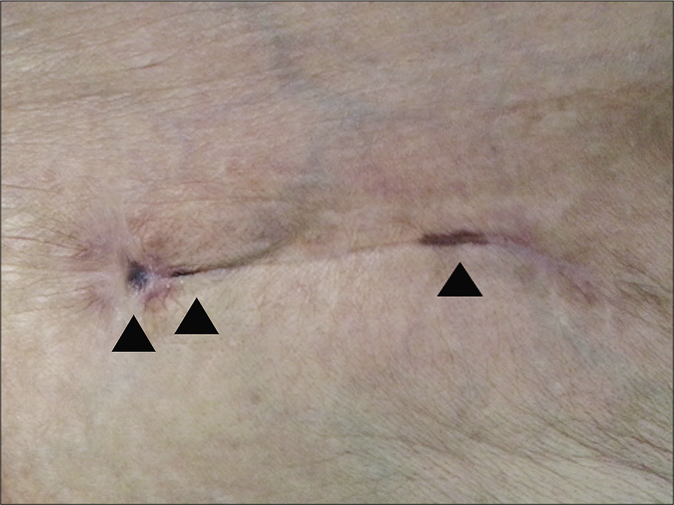
- Physical examination revealed several well-demarcated, black macules along the linear scar line
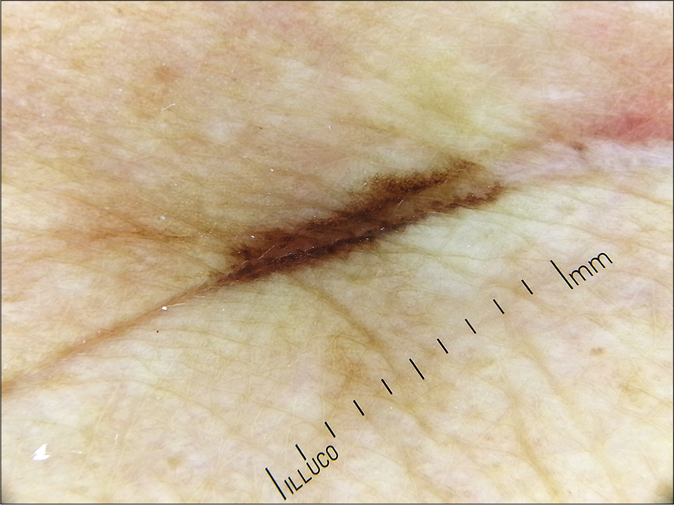
- Dark brown irregular streaks confined to the linear scar line (original magnification, ×10)
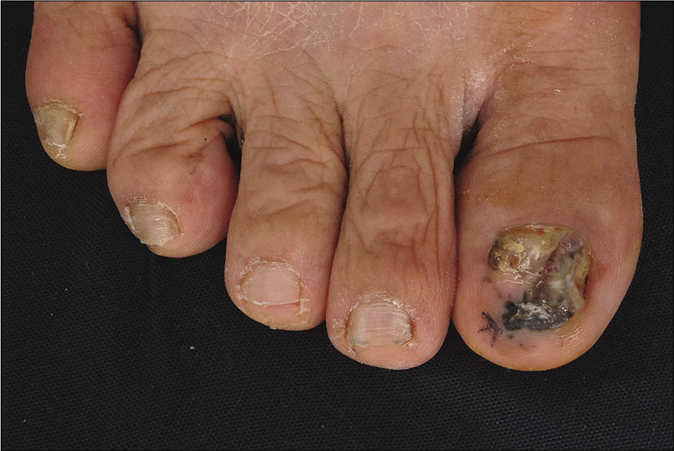
- The acral lentiginous melanoma on the right big toe
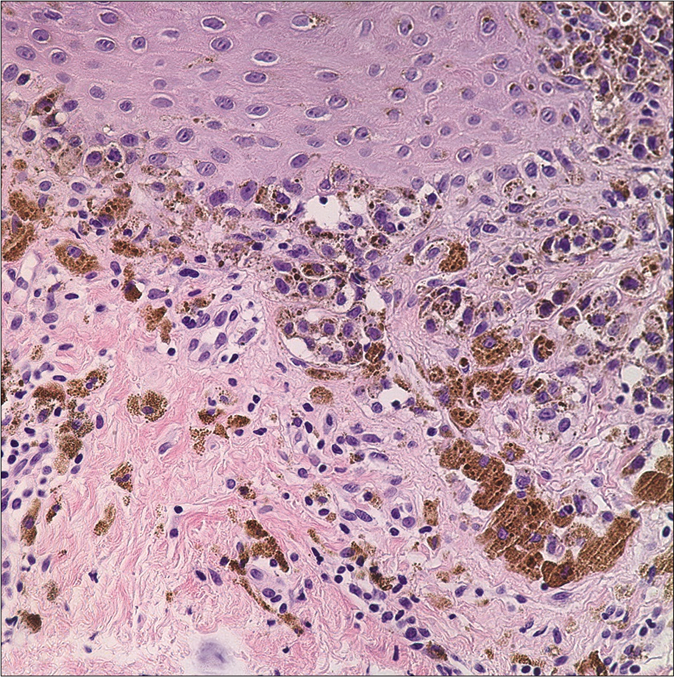
- The biopsy on the scar revealed a lentiginous array of atypical melanocytes (hematoxylin and eosin, ×400)
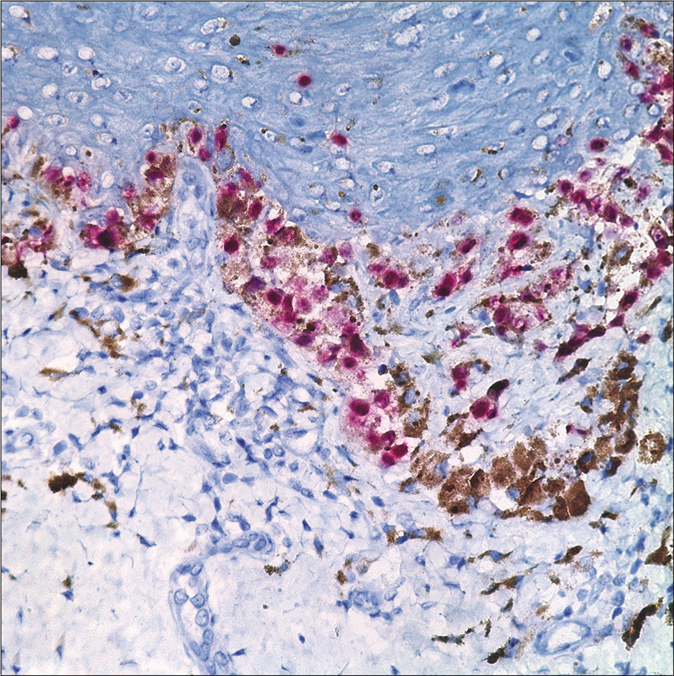
- Atypical cells showing HMB45 stain positivity (HMB-45, ×400)
The whole-body positron emission tomography showed no evidence of distant metastasis. We concluded that this patient had melanoma tumor seeding as a result of CLND. She is undergoing adjuvant immunotherapy with pembrolizumab.
Over the past 30 years, surgeons have witnessed a shift from elective lymph node dissection to CLND after positive SLNB and now toward SLNB alone.1 The previous studies sought to address whether CLND is therapeutic or only provides prognostic information. The multicenter selective lymphadenectomy trial two was an international, randomized, Phase three clinical trial comparing immediate CLND against observation in melanoma patients positive for sentinel node metastasis.2 There were no significant differences in either mean three-year melanoma-specific survival or distant metastasis-free survival between patients who underwent CLND or observation after lymph node metastasis was detected. Therefore, the therapeutic value of CLND for all patients remains doubtful.
We assume that the melanoma on the CLND scar in our case is attributed to tumor seeding for the following reasons. First, the lesions were located on both ends of linear scar line where the incision line was extended for better visibility during CLND and this could be easily identified with a dermoscope [Figure 2]. Second, the lymphatic spread is unlikely in that no lesion was detected on the ipsilateral limb of primary lesion except the linear scar line. Finally, our patient had low possibility of hematogenous metastasis in that she had metastasis to the skin rather than metastasis of visceral organs.
Robinson and Brown reported a case of fine-needle aspiration associated tumor seeding of a malignant melanoma within the head-and-neck region.3 We think that this report might be the first case of tumor seeding due to CLND in melanoma. Therefore, in managing melanoma, clinicians should perform CLND selectively while assessing risk versus benefit and be cautious about the cutaneous seeding during CLND.
Declaration of patient consent
The patient's consent is not required as the patient's identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Sentinel lymph node biopsy and management of regional lymph nodes in melanoma: American society of clinical oncology and society of surgical oncology clinical practice guideline update summary. J Oncol Pract. 2018;14:242-5.
- [CrossRef] [Google Scholar]
- Completion dissection or observation for sentinel-node metastasis in melanoma. N Engl J Med. 2017;376:2211-22.
- [CrossRef] [Google Scholar]
- Tumour seeding in a case of malignant melanoma due to fine needle aspiration of a lymph node. J Plast Reconstr Aesthet Surg. 2010;63:e571-2.
- [CrossRef] [Google Scholar]





