Translate this page into:
Dermatoscopy of multiple piloleiomyomas with disseminated and segmental distribution
2 Faculty of Medicine, Institute of Pathology, University of Belgrade, Belgrade, Serbia
Correspondence Address:
Mirjana Popadić
Pasterova 2, 11000 Belgrade
Serbia
| How to cite this article: Popadić M, Brasanac D, Milinković M, Milčić D. Dermatoscopy of multiple piloleiomyomas with disseminated and segmental distribution. Indian J Dermatol Venereol Leprol 2018;84:726-729 |
Sir,
A 42-year-old female presented with disseminated, painless lesions, which she has had for nine years and which gradually increased in number. Her medical history included myomectomy five years earlier.
Clinical examination revealed clustered, firm, reddish-brown papules, fixed to the skin, covering the forehead, cheeks [Figure - 1]a and [Figure - 1]b, left side of the neck [Figure - 1]a, gluteal region (disseminated distribution) and the right scapular region (segmental distribution) [Figure - 1]c.
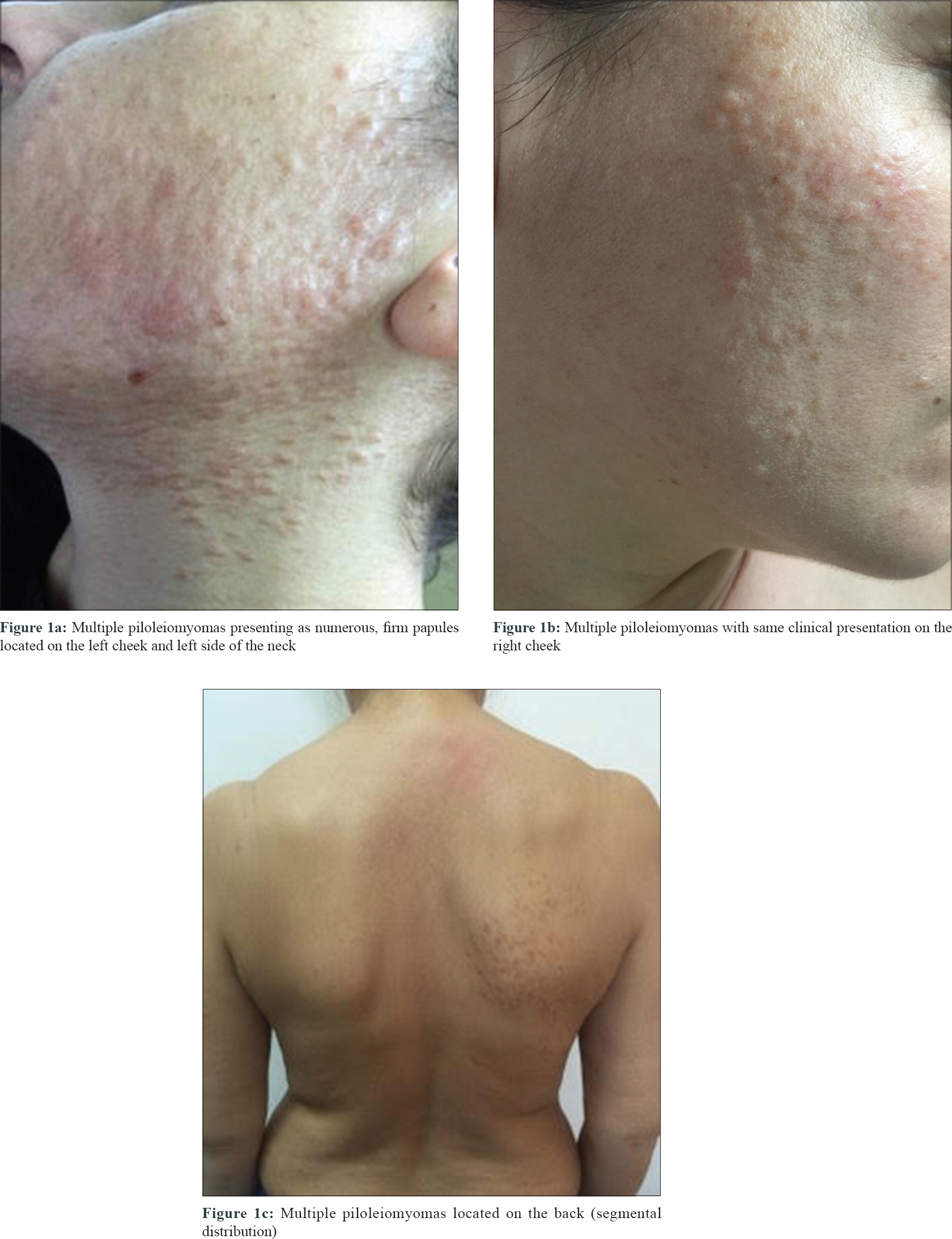 |
| Figure 1: |
The papules located on the head and neck region dermatoscopically showed whitish clods, scattered and partially clustered throughout the lesion, varying in size and number, with delicate brown reticular lines between them [Figure - 2]a and [Figure - 2]b. The papules localized on the back mainly showed only brown, reticular, parallel or irregular lines [Figure - 2]c and [Figure - 2]d.
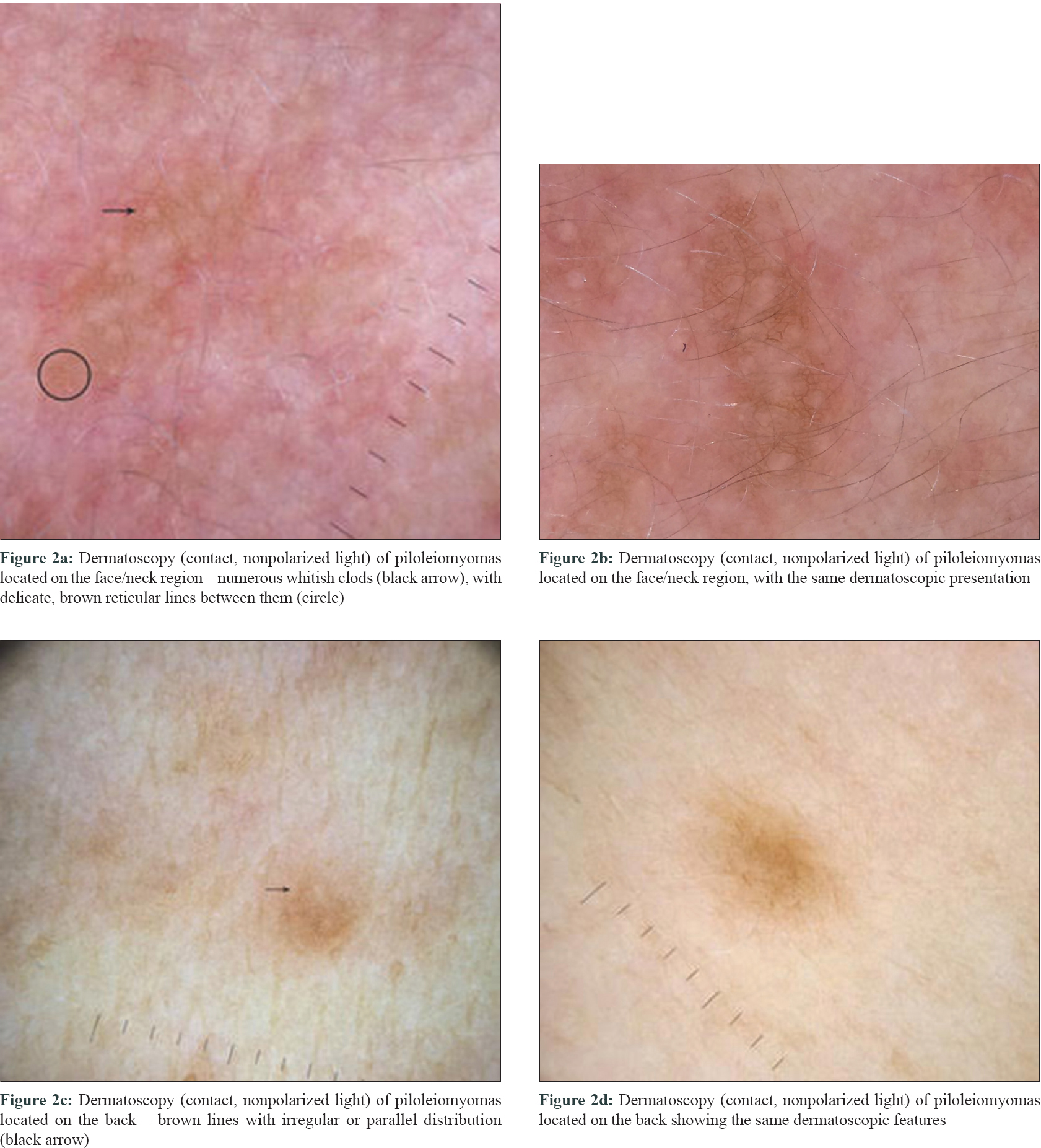 |
| Figure 2: |
Two incisional biopsies were performed, one from the jawline and the second from the back. Histopathological examination of both biopsies showed bundles of eosinophilic, elongated cells with cigar-shaped nuclei [Figure - 3]a, immunohistochemically positive for desmin [Figure - 3]b.
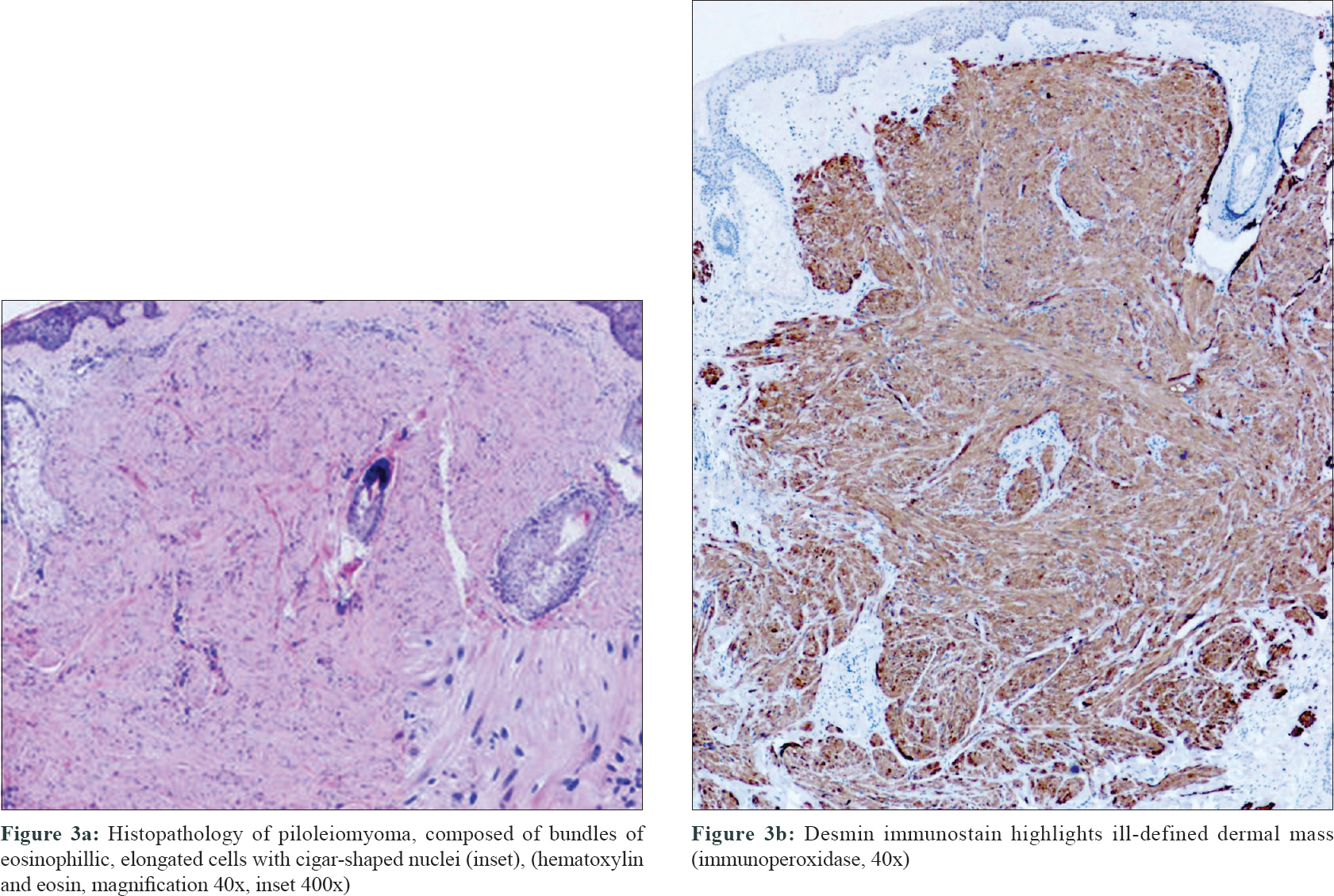 |
| Figure 3: |
Gynecological examination revealed uterine leiomyomatosis confirmed by ultrasound pelvis examination.
Cutaneous leiomyomas, first described in 1854 by Rudolf Virchow, are uncommon, painful neoplasms arising from smooth muscle cells.[1] They comprise ~5% of all leiomyomas and 75% of extrauterine leiomyomas.[2] The precise etiology of this disease is still unknown but the genetic alterations of the fumarate hydratase gene has been suggested, with reports of autosomal dominant inheritance in familial occurrence of multiple piloleiomyomas.[2],[3],[4] According to the origin, they are classified into three groups – piloleiomyomas (the most common type), genital (dartoic) leiomyomas and angioleiomyomas.[2],[3]
Piloleiomyoma may appear as solitary, or more often as multiple lesions. Segmental (zosteriform) distribution represents a rare pattern, with only few cases reported in the literature.[2] However, we were unable to find any previous report of simultaneous occurrence of segmental and disseminated piloleiomyomas, which represent type 2 segmental mosaicism as it was in our case. In this type of mosaicism, individuals carrying a germline-dominant mutation that affects all cells of the body acquire a second postyzgotic alteration of the other allele of the same gene, resulting in loss of heterozygosity affecting only a part of the cells. The consequence of this type of genetic event is a clinical presentation in segmental areas superimposed on the milder diffuse cutaneous involvement.[5]
Multiple piloleiomyomas could be accompanied by uterine leiomyomatosis, as it was the case in our patient, or rarely by renal cell carcinoma (multiple cutaneous and uterine leiomyomatosis syndrome, hereditary leiomyomatosis and renal cell cancer).[1],[4]
Piloleiomyomas are frequently unrecognized by clinicians during examination. Despite widespread use of dermatoscopy, a small number of patients with piloleiomyomas have been dermatoscopically evaluated.[1],[3],[4],[6] In several reports, both solitary[1],[3] and multiple piloleiomyomas[2],[4],[6] have been described. All authors reported that the dermatoscopic findings of piloleiomyoma showed characteristics similar to dermatofibroma. Histopathologically, the central white patch reflects the proliferation of smooth muscle cells and collagenous fibers in the dermis, whereas the delicate pigment network reflects reactive epidermal basal pigmentation.[1]
The tumors located on the head and neck region in our patient also showed discrete brown reticular lines, but mixed with numerous whitish clods of variable size and number throughout the whole lesions. However, piloleiomyomas on the back predominantly showed only discrete, brown lines of irregular or parallel distribution, resembling more the dermatoscopic finding of flat seborrheic keratosis or solar lentigo.
Different dermatoscopic presentations suggest interdependence on tumor location. The explanation for such a difference could be the presence of a higher number of follicular ostia, which are also more prominent, on the face/neck region compared to the skin of the back.
However, oval and/or elongated hyperpigmented structures within the central area, reported by Paschoal,[6] and central masses of “white-like cloud” areas without translucent streak reported by Diluvio[4] were not found in our case. Although prominent telangiectases[6] and dotted vessels[1] were observed by some authors, in our case no vascular structures were found.
The main clinical and dermatoscopic differential diagnosis particularly in solitary lesions should include dermatofibroma, flat seborrheic keratosis/solar lentigo and neurofibromas. In cases where piloleiomyoma has characteristics similar to dermatofibroma, the negative “button sign” should be suggestive for considering the biopsy. On the other hand, neurofibromas often display only a delicate brownish network at the periphery of the lesions which may sometimes be confused with piloleiomyomas, however, the softness of the neurofibromas is the major differential clue from both piloleiomyoma and dermatofibroma.[7] However, in dermatoscopic presentation of piloleiomyoma similar to flat seborrheic keratosis/solar lentigo, they could be indistinguishable, especially in multiple flat lesions of seborrheic keratosis/solar lentigo localized on the back.
The correct diagnosis of multiple piloleiomyomas is of high importance, particularly in female patients where detailed gynecological and renal investigations should be done, with regular annual follow-ups for risk-assessment of leiomyosarcoma and renal carcinoma.
In conclusion, we report a case of type 2 segmental mosaicism of piloleiomyomas, which showed various dermatoscopic presentations at different locations. In addition, we report a new dermatoscopic presentation of piloleiomyoma, resembling flat seborrheic keratosis/solar lentigo more than dermatofibroma.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patient has given her consent for her images and other clinical information to be reported in the journal. The patient understands that name and initials will not be published and due efforts will be made to conceal identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. |
Suzuki R, Fukuda H, Mukai H. Dermoscopic findings of piloleiomyoma with hypertrichosis. Eur J Dermatol 2015;25:362-4.
[Google Scholar]
|
| 2. |
Albuquerque MM, Rocha CF, Costa IS, Maia Rda R, Branco FJ, Gonçalves Hde S, et al. Piloleiomyoma with segmental distribution – Case report. An Bras Dermatol 2015;90:178-80.
[Google Scholar]
|
| 3. |
Kim GW, Park HJ, Kim HS, Kim SH, Ko HC, Kim BS, et al. Giant piloleiomyoma of the forehead. Ann Dermatol 2011;23:S144-6.
[Google Scholar]
|
| 4. |
Diluvio L, Torti C, Terrinoni A, Candi E, Piancatelli R, Piccione E, et al. Dermoscopy as an adjuvant tool for detecting skin leiomyomas in patient with uterine fibroids and cerebral cavernomas. BMC Dermatol 2014;14:7.
[Google Scholar]
|
| 5. |
Goldberg I, Sprecher E. Patterned disorders in dermatology. Clin Dermatol 2011;29:498-503.
[Google Scholar]
|
| 6. |
Paschoal FM, Rezze GG. Dermoscopic findings in a patient with multiple piloleiomyomas. Dermatol Pract Concept 2012;2:204a06.
[Google Scholar]
|
| 7. |
Duman N, Elmas M. Dermoscopy of cutaneous neurofibromas associated with neurofibromatosis type 1. J Am Acad Dermatol 2015;73:529-31.
[Google Scholar]
|
Fulltext Views
2,185
PDF downloads
441





