Translate this page into:
Epidermodysplasia verruciformis: An unusual malignant transformation
Correspondence Address:
Prachi G Agrawal
Department of Skin & V.D., Opd No. 117, GSMC & KEM Hospital, Parel, Mumbai - 400 012
India
| How to cite this article: Agrawal PG, Mahajan SA, Khopkar US, Kharkar VD. Epidermodysplasia verruciformis: An unusual malignant transformation. Indian J Dermatol Venereol Leprol 2013;79:97-99 |
Abstract
Epidermodysplasia verruciformis (EV) is a rare, life-long heritable disease caused due to a unique susceptibility to human papilloma virus. The disseminated verrucous lesions and pityriasis versicolor-like lesions persist from early childhood and can transform into a cutaneous malignancy in a fourth of patients. Malignant transformation into syringoid eccrine carcinoma (SEC) has been reported only once so far. SEC is an extremely invasive, rare, locally destructive, slowly growing adnexal tumor. We hereby report the association of EV with SEC in a 29-year-old male.Introduction
Epidermodysplasia verruciformis (EV) is a rare geno-dermatosis characterized by a unique susceptibility to cutaneous infection by human papilloma viruses (HPVs). The defect is usually inherited as an autosomal recessive trait and presents clinically with plane warts, pityriasis versicolor-like lesions, and reddish verrucous plaques. Dysplastic and malignant changes may occur in the form of actinic keratoses, Bowen′s disease, squamous cell carcinoma (SCC), basal cell carcinoma, or rarely sweat apparatus carcinoma. [1] We hereby report a case of EV with syringoid eccrine carcinoma (SEC). SEC is an extremely invasive, rare, locally destructive, slowly growing adnexal tumor, derived from eccrine sweat glands. [2] Only a single case of SEC in EV patient has been reported till date. [3]
Case Report
A 29-year-old male born of non-consanguineous marriage presented with multiple asymptomatic dark colored and light colored lesions over the face, trunk, and extremities since the age of 8 years. These lesions had appeared over a period of time and progressively increased in number and size. Two years back, patient developed a large nodule over the scalp which was excised twice and finally turned into a non-healing ulcer. About 1.5 years ago, the patient developed a peanut-sized outgrowth near the lateral canthus of the right eye which was excised with histopathology report suggestive of some malignancy (details not available). On clinical examination of the patient, multiple mildly erythematous, scaly, hypopigmented, pityriasis versicolor-like lesions were present over the trunk and extremities and multiple brownish-black hyperkeratotic verrucous papules and plaques (actinic keratoses) were present over the extremities, neck, and face [Figure - 1]. A large non-healing ulcer was present on the scalp measuring 8 × 5.5 cm, non-tender, hard, fixed, with rolled out margins and bleeding on touch [Figure - 2]. The lymph nodes were not enlarged and the examination of hair, nail, and mucous membrane was normal. The systemic examination of the patient revealed no abnormality. On investigating, complete blood counts, serum biochemistry, HIV serology, and urinalysis were normal. X-ray of skull showed destruction of the calvarial bones overlying the parietal scalp [Figure - 3]a. Magnetic resonance imaging (MRI) of brain showed a mass lesion measuring 9 × 6.3 × 3.7 cm in right parietal and occipital region involving the calvarium and subgaleal space extending into the epidural space with intracranial extension [Figure - 3]b. There was no other clinico-investigative evidence of distant metastasis. Haematoxylin and eosin (H and E) stained section of a cutaneous biopsy from verrucous papule [Figure - 4] showed hyperkeratotic and acanthotic epidermis and multiple vacuolated cells in the upper stratum malpighii and granular layer with basophilic cytoplasm and numerous round, basophilic keratohyalin granules. Some nuclei were pyknotic; others appeared large, round, and empty. A few dyskeratotic cells were present in the lower epidermis. H and E stained section of a cutaneous biposy from the scalp lesion [Figure - 5] showed nests, cords, and tubules of neoplastic cytologically uniform, cuboidal, or squamous cells randomly dispersed in a dense desmoplastic collagenous stroma. Few nests resembled "tadpoles" or "commas" and few cell aggregates showed lumen, some of which contained eosinophilic secretory material. There was an extension of the neoplastic cells into the subcutaneous tissue. Based on the history, clinical features, and investigations, the final diagnosis was kept as EV with SEC. The patient was given genetic counseling and sunscreens for photoprotection. He was advised life-long observation for early diagnosis of malignant/premalignant lesions. Radiotherapy and chemotherapy (five cycles of injection cisplatin, 30 mg/ m 2 over 5 weeks) were administered for the scalp lesion.
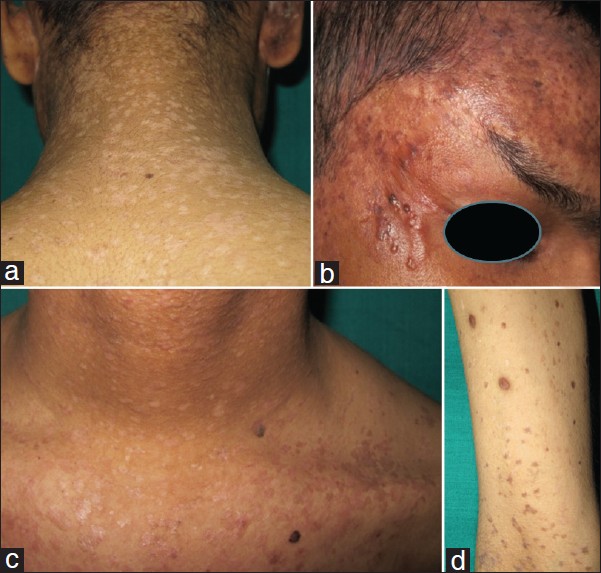 |
| Figure 1: Multiple pityriasis versicolor-like lesions with few brownish-black hyperkeratotic verrucous papules and plaques present over the (a) back, (b) face, (c) chest, and (d) forearm |
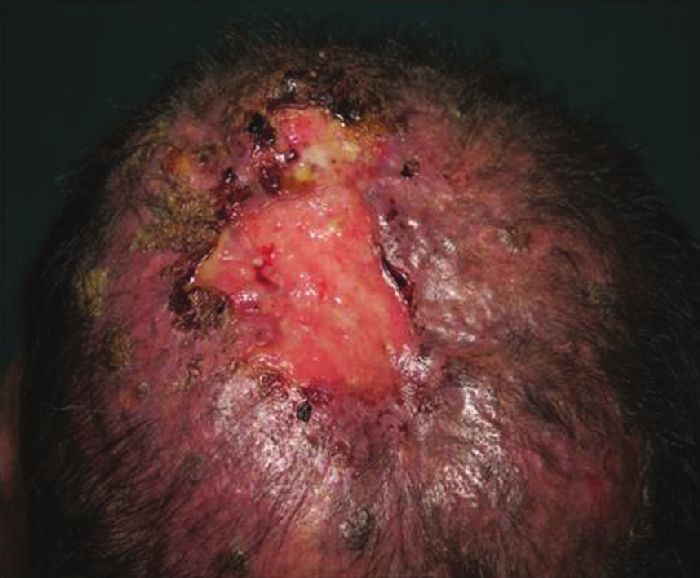 |
| Figure 2: A large non-healing ulcer with rolled out margins on the scalp |
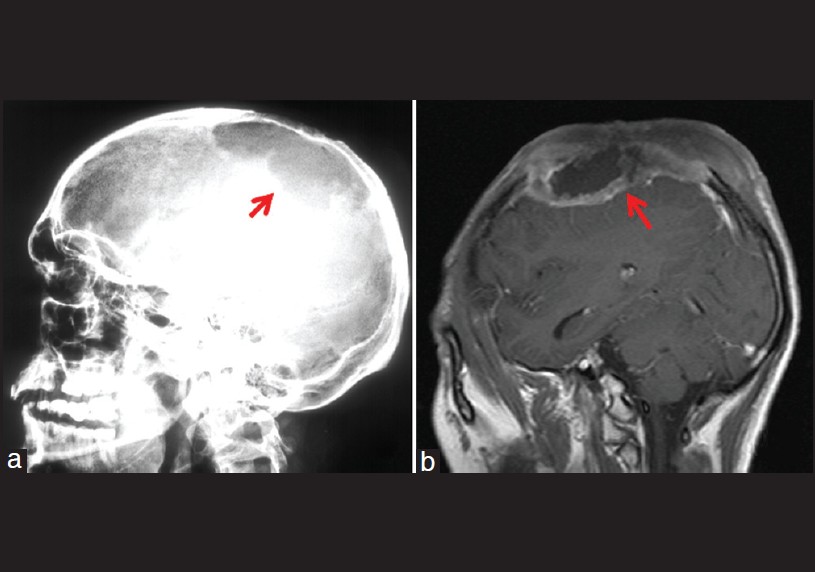 |
| Figure 3: (a) X-ray of skull, lateral view showing destruction of calvarial bones. (b) MRI brain showing mass involving the calvarium and subgaleal space |
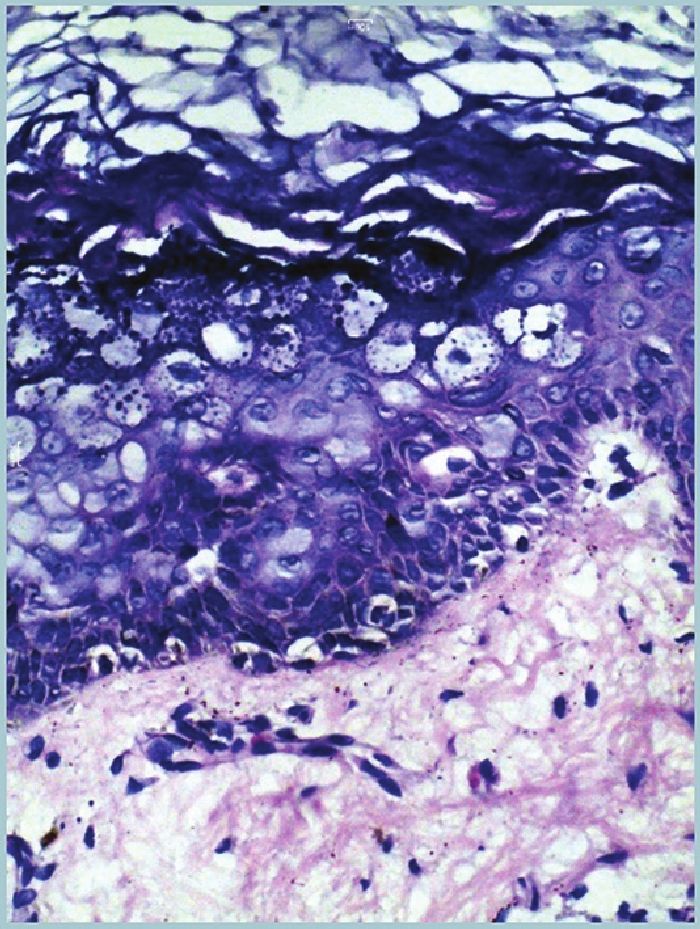 |
| Figure 4: Biopsy from verrucous papule showing hyperkeratotic and acanthotic epidermis and multiple basophilic vacuolated cells in granular layer (H and E, ×40) |
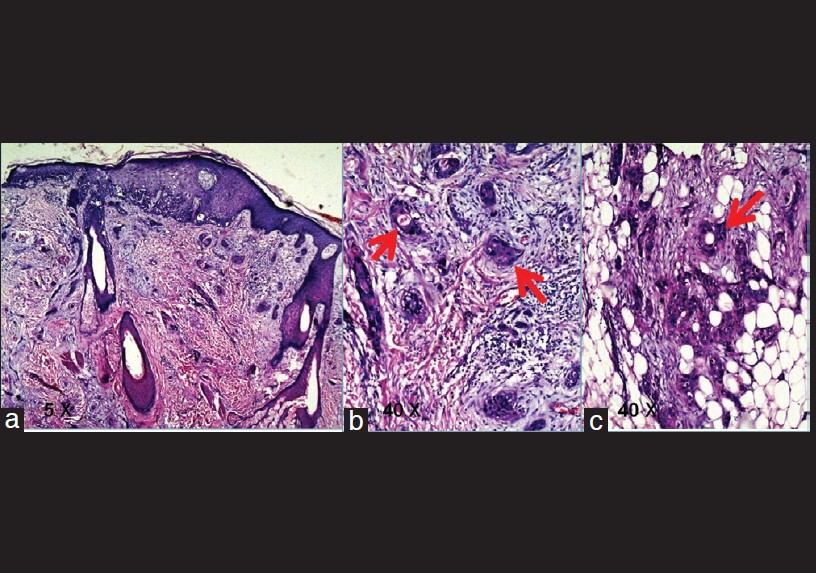 |
| Figure 5: Biopsy from scalp lesion showing nests, cords, and tubules of neoplastic cells dispersed in desmoplastic stroma. Few nests resemble "tadpoles" or "commas" and few cell aggregates show lumen [(a) H and E, ×10 (b and c) H and E, ×40] |
Discussion
EV is a rare genodermatosis representing a widespread and persistent infection with HPVs. Mostly HPV 3, 5, 8, 9, 10, 12, 14, 17, 19-25, 28, and 29 are related, out of which HPV 5 and 8 are the main types incriminated for malignant transformation. [1] It is usually transmitted as an autosomal recessive disorder with mutations in EVER1/TMC6 and EVER2/TMC8 genes located on chromosome 17q 25. [4] A HPV-specific defect of cell-mediated immunity is present. There is no gender predisposition. [1] Its frequency is 11% in Europe and the USA and 40% among Japanese, but it has been reported infrequently from India. [5] It usually begins in childhood and continues throughout the patient′s lifetime. It presents clinically with characteristic combination of pityriasis versicolor-like lesions, reddish verruca-like and seborrheic keratosis-like plaques. The lesions are preferentially located on sun-exposed sites. It is often associated with IgM deficiency and HIV infection. [6] Dysplastic and malignant changes may occur in the form of actinic keratosis, Bowen′s disease, SCC, basal cell carcinoma, or rarely sweat apparatus carcinoma. Malignant transformation into SCC occurs in 30-70% cases. Actinic damage is perhaps the pathogenic mechanism involved in malignant transformation. [1] The diagnosis is mainly clinico-pathological, while DNA amplification studies may be needed for identification of HPVs′ types (could not be done in our patient due to financial constraints). Stringent sun protection and life-long observation for early diagnosis of malignant/premalignant lesions are imperative for improved survival. Acitretin (0.5- 1 mg/ day) is the effective and palliative drug of choice. [5] Chemotherapy with or without radiotherapy may be useful to achieve regression of the tumor/lymph nodes before surgery.
SEC is an extremely invasive, rare, locally destructive, slowly growing adnexal tumor, derived from eccrine sweat glands. It has no gender predilection and occurs in the middle aged and elderly. It is mostly situated on the head and neck. It has high chances of recurrence due to extensive perineural invasion. The chances of regional or distant metastases are rare. It extends deep into the dermis, often into subcutaneous fat and skeletal muscle. A correct histological diagnosis is of importance because of the localy aggressive nature of the tumor. [2],[3] The histological diagnosis can be confirmed by immunohistochemical analysis: positivity for anti-epithelial membrane antigen (EMA), anti- carcinoembryonic antigen (CEA), and S100 protein is consistent with adnexal tumor with sweat gland differentiation (could not be done in our patient due to financial constraints). The optimal treatment consists of a complete microscopically controlled surgical excision with clear surgical margins. [2]
The co-existence of SEC and EV has been described only once in the literature to the best of our knowledge.
| 1. |
Gül U, Kiliç A, Gönül M, Cakmak SK, Bayis SS. Clinical aspects of epidermodysplasia verruciformis and review of the literature. Int J Dermatol 2007;46:1069-72.
[Google Scholar]
|
| 2. |
Hoppenreijs VP, Reuser TT, Mooy CM, De Keizer RJ, Mourits MP. Syringomatous carcinoma of the eyelid and orbit: A clinical and histopathological challenge. Br J Ophthalmol 1997;81:668-72.
[Google Scholar]
|
| 3. |
Beaulieu P, Blanchet-Bardon C, Breitburd F, Verola O, Favre M, Dubertret L. Epidermodysplasia verruciformis and eccrine syringoid carcinoma. Ann Dermatol Venereol 1993;120:833-5.
[Google Scholar]
|
| 4. |
Aochi S, Nakanishi G, Suzuki N, Setsu N, Suzuki D, Aya K, et al. A novel homozygous mutation of the EVER1/TMC6 gene in a Japanese patient with epidermodysplasia verruciformis. Br J Dermatol 2007;157:1265-6.
[Google Scholar]
|
| 5. |
Sehgal VN, Luthra A, Bajaj P. Epidermodysplasia verruciformis: 14 members of a pedigree with an intriguing squamous cell carcinoma transformation. Int J Dermatol 2002;41:500-3.
[Google Scholar]
|
| 6. |
Vohra S, Sharma NL, Shanker V, Mahajan VK, Jindal N. Autosomal dominant epidermodysplasia verruciformis: A clinicotherapeutic experience in two cases. Indian J Dermatol Venereol Leprol 2010;76:557-61.
[Google Scholar]
|
Fulltext Views
2,651
PDF downloads
727





