Translate this page into:
Ethionamide-induced pellagroid dermatitis resembling lichen simplex chronicus: A report of two cases
Correspondence Address:
Uday Khopkar
Department of Dermatology, Seth GS Medical College and KEM Hospital, Parel, Mumbai - 400 012, Maharashtra
India
| How to cite this article: Garg G, Khopkar U. Ethionamide-induced pellagroid dermatitis resembling lichen simplex chronicus: A report of two cases. Indian J Dermatol Venereol Leprol 2011;77:534 |
Abstract
Pellagra is a niacin deficiency disorder characterized clinically by diarrhea, dermatitis, and dementia. However, few drugs also cause pellagroid dermatitis. Recently, we encountered two cases of pellagroid dermatitis; both were on second line of antituberculosis drugs. Case 1 was of multidrug-resistant pulmonary tuberculosis. Patient was on ethionamide since one year before developing pellagroid dermatitis. Case 2 was of central nervous system tuberculoma and was on second line of antitubercular drugs. This patient was on ethionamide and isoniazid (INH) since six months before developing pellagroid dermatitis. This patient had previously taken first line of antituberculous therapy, inclusive of INH, for 1 year without any dermatitis. The skin lesions in both patients were symmetric hyperpigmented thickened plaques with prominent skin markings resembling lichen simplex chronicus. Nicotinamide 300 mg in three divided doses healed the lesions completely within 4 weeks and 3 weeks in first and second patient, respectively.Introduction
The traditionally remembered classical triad of symptoms of pellagra, i.e., 3 d′s; dermatitis, diarrhea, and dementia, is very occasionally seen in today′s era. Most cases are less typical and borderline in nature. [1],[2],[3] Pellagra is caused by deficiency of niacin or its precursor tryptophan. It mainly occurs in undernourished patients with history of alcohol abuse. [1] Uncommonly, it occurs secondary to drugs, especially anitubercular drugs such as isoniazid (mainly) and ethionamide (rarely) and phenytoin, chloramphenicol, azathioprine, 5-fluorouracil, and phenobarbital. [2],[3] It is characterized both by dermatologic and systemic manifestations.
Case Reports
Case 1
A 32-year-old house wife presented with well-defined, hyperpigmented, hyperkeratotic, symmetrical, thick scaly plaques surrounded by erythema over dorsal aspect of both hands and first three toes of both feet since 15 to 20 days [Figure - 1]. Lesions were increasing gradually in size. She also complained of mild to moderate burning and itching over lesions while going out in sun. Mild erythema of lips and tongue were present in the patient. No history of diarrhea or affection of mental faculties was present. There was no lesion on "V" area of neck. One and half years back, she was diagnosed as pulmonary tuberculosis and was started on four drug regimen of isoniazid, rifampicin, ethambutol, and pyrazinamide. After four months of treatment with the above four drugs, she developed cold abscess in the sternal region, which was drained and the pus culture was suggestive of multidrug-resistant tuberculosis, resistant to all four drugs of first-line antituberculous regimen. She was started on second-line antitubercular regimen consisting of ethionamide 250 mg bid, PAS granules one measure (3.5 gm) bid, and ofloxacin 400 mg bid, 1 year back. Injection kanamycin 500 mg intramuscular alternate day and clofazimine 100 mg od were also taken for initial six months of second-line antitubercular regimen. She denied any exposure to chemical agents and contact allergens. A differential diagnosis of pellagroid dermatitis, zinc deficiency, acanthosis nigricans, marginal keratoderma, and lichen simplex chronicus was kept.
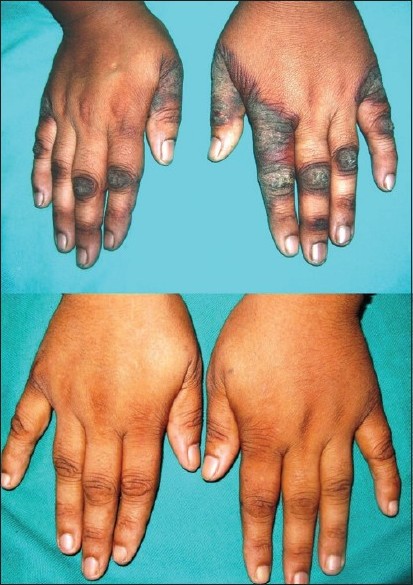 |
| Figure 1: Pre- and post-treatment photographs of patient one. Hyperpigmented, hyperkeratotic, well-defined plaques surrounded with dusky erythema were present over dorsum of both hands mimicking lichen simplex chronicus. Lesions cleared completely after four weeks of nicotinamide therapy |
Her blood investigations were: Complete blood count - 11 000/mm 3 , erythrocyte sedimentation rate - 36.0 mm at the end of one hour, blood urea nitrogen - 7.0 mg/dl, and serum electrolyte levels - Na + 136.0 mEq/l, K + 4.2 mEq/l, Cl - 101.0 mEq/l which were within normal limits. Serum zinc levels were 88.9 μg/dl and total proteins were 7.6 mg/dl (albumin, 3.6 mg/dl) which were also within normal limits. Skin biopsy on scanner view showed hyperplastic epidermis with moderate thickening of the granular and corneal layer. Sparse superficial perivascular lymphocytic and neutrophilic infiltrate was present [Figure - 2]. On higher magnification, ballooning of upper spinous and granular layer with eosinophil staining of lower stratum corneum with focal parakeratosis was present [Figure - 2].
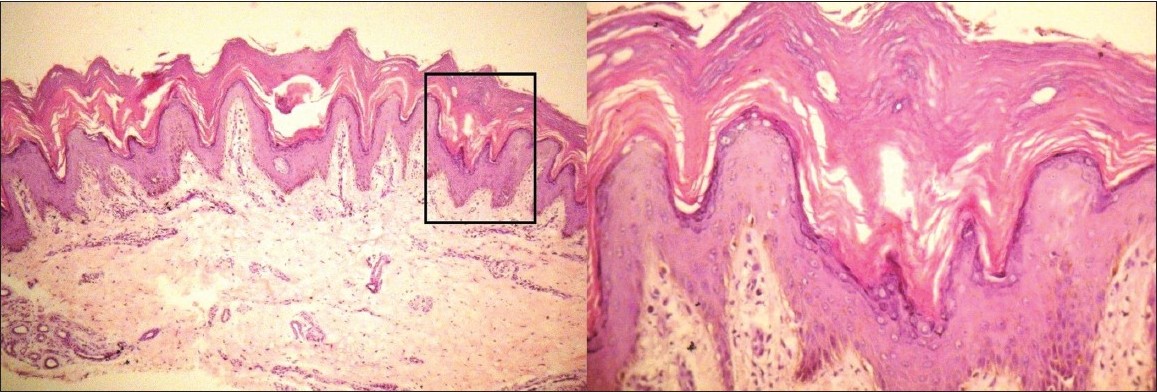 |
| Figure 2: Biopsy sample from margin of hyperpigmented, hyperkeratotic plaque of patient one. Scanner view shows hyperplastic epidermis with moderate thickening of the granular and corneal layer with sparse superficial perivascular lymphocytic and neutrophilic infiltrate (H and E, ×40). Higher magnification shows ballooning of upper spinous and granular layer with eosinophil staining of lower stratum corneum. Focal parakeratosis is present (H and E, ×400) |
Based on history, physical examination, and investigations, a diagnosis of drug-induced pellagra-like dermatitis was established. Patient was started on nicotinamide (300 mg/day) in three dividing doses with multivitamin B complex while continuing her multidrug-resistant antitubercular regimen. Her lesions healed completely within 4 weeks of nicotinamide therapy [Figure - 1]. Proper nutrition with high protein diet and sun screen creams was also added to her treatment protocol. She was advised to continue nicotinamide supplements 100 mg/day till the completion of the course of antitubercular therapy.
Case 2
A 21-year-old female presented with multiple, symmetric, hyperpigmented, hyperkeratotic, well-defined plaques surrounded by dusky discoloration over dorsum of both hands, more on middle and terminal phalanges of fingers, gradually progressive in size and thickness since three months. Similar lesions were also present over medial and lateral aspect of both feet [Figure - 3]. No history of itching and burning was present over lesions while going out in sun. No history of diarrhea and mental confusion was present. There was no cheilitis or glossitis. Also, no lesion was present over "V" area of neck. Five years back, patient was diagnosed as pulmonary tuberculosis and was started on four drug regimen of isoniazid, rifampicin, ethambutol, and pyrazinamide. After three months of regular treatment, patient discontinued the treatment on its own. Three years back, she was diagnosed as tubercular meningitis and started on first-line antitubercular drugs for one year. She successfully completed her course. No side effect was noted during this period. After one year of completion of course of antitubercular drugs, the patient again got convulsions and her brain MRI was suggestive of multiple tuberculomas. Patient was started on second line of antitubercular drugs nine months back, including ethionamide 250 mg bid, isoniazid 300 mg od, rifampicin 600 mg od, levofloxacin 750 mg od, injection kanamycin 0.5 ml intramuscular weekly, carbamazepine 200 mg bid, and clobazam 50 mg od. After 6 months of second-line antitubercular therapy, she started developing hyperpigmented, hyperkeratotic, and mildly scaly plaques over dorsum of both hands and medial and lateral aspect of both feet. She denied any exposure to chemical agents and contact allergens. A differential diagnosis of pellagroid dermatitis, acanthosis nigricans, psoriasis, lichen simplex chronicus, marginal keratoderma, and zinc deficiency was kept.
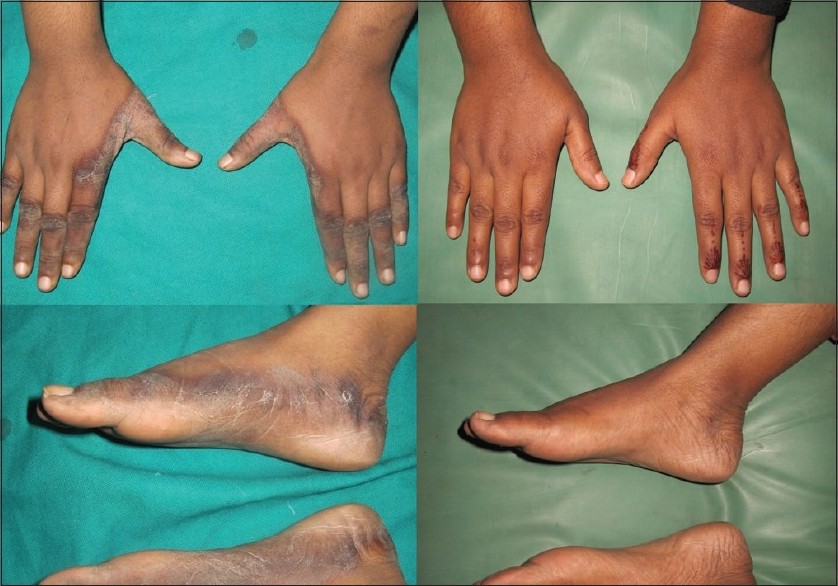 |
| Figure 3: Pre- and post-treatment photographs of patient two showing symmetric, hyperpigmented, hyperkeratotic, well-defined plaques surrounded by dusky discoloration over medial and lateral aspect of both hands and feet. Lesions healed completely after three weeks of nicotinamide therapy |
Her blood investigations were: Complete blood count - 6 000/ml, erythrocyte sedimentation rate - 23.0 mm at the end of one hour, blood urea nitrogen - 6.0 mg%, serum creatinine - 0.6 mg%, total protein - 6.6 mg% (albumin, 3.0 mg%), SGOT - 69 U/l, SGPT - 30 U/l, and serum electrolyte levels - Na + 137.0 mEq/l, K + 3.3 mEq/l, Cl - 101 mEq/l. Serum zinc levels - 86.4 μg/dl and serum B 12 levels - 1048 pg/ml were within normal limits. Her skin biopsy from the periphery of the hyperpigmented plaque showed hyperkeratosis, focal parakeratosis, acanthosis, and sparse superficial perivascular lymphocytic and neutrophilic infiltrate.
Based on history, physical examination, and investigations, a diagnosis of drug-induced pellagra-like dermatitis was established. She was started on nicotinamide (300 mg/day) in three divided doses with multivitamin B complex. Proper nutrition with high protein diet and sun screen creams was also advised. Her lesions healed completely within three weeks of nicotinamide therapy [Figure - 3]. She was advised to continue nicotinamide supplements 100 mg/day until the course of her antitubercular medicines complete.
Discussion
Pellagra is a systemic disease resulting from deficiency of niacin or vitamin B 3 . The dietary tryptophan produces niacin in the presence of vitamins B 2 and B 6 . [2] Niacin is an essential component of coenzyme 1 (oxidized form of nicotinamide adenine dinucleotide [NAD]) and coenzyme 2 (oxidized form of nicotinamide adenine dinucleotide phosphate [NADP]) which are required for adequate cellular functioning and various metabolic reactions in our body. [1],[2] Historically, pellagra is a disease of a maize-eating population. It has also been reported in India in some jowar (Sorghum vulgare)-eating populations. [4] Amino-acid imbalance caused by an excess of leucine is the cause of pellagra in both jowar and maize eaters. Excess of leucine appears to interfere in the conversion of tryptophan to niacin. [4]
Malabsorption syndromes (jejunoileitis, gastroenterostomy, subtotal gastrectomy, Crohn′s disease), chronic alcoholism, [1] metabolic derangements (Hartnup disease, [5],[6] carcinoid syndrome [7],[8] ), and drugs [2] (INH, 6-mercaptopurine, 5-fluorouracil, pyrazinamide, phenytoin, chloramphenicol, azathioprine, phenobarbitone, ethionamide) are the causes of pellagra. In chronic alcoholics, pellagra occurs as a result of nutritionally poor diet and malabsorption. The hepatic cell also inefficiently utilizes nicotinic acid in chronic alcoholics. [1] In Hartnup disease, due to the defect in the neutral brush border system, malabsorption of amino acids as tryptophan occurs. [5],[6] However, in carcinoid syndrome, there is increased conversion of tryptophan to serotonin, leading to decreased production of niacin. [7],[8]
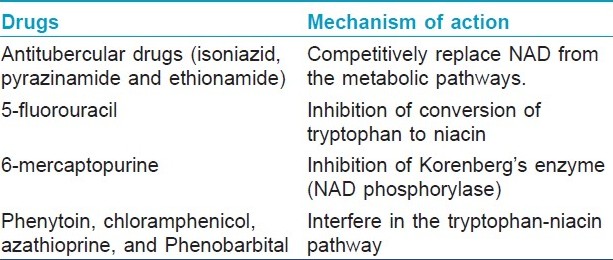
Antitubercular drugs, isoniazid, pyrazinamide, and ethionamide, are structurally similar with NAD. [9] All these substances have a ring of five carbon atoms and one of nitrogen in position 4. [9] These antitubercular drugs competitively replace NAD from the metabolic pathways. Hence, tissues with high energy requirements, like brain, or with high turnover rate like skin and gut, get affected in pellagra. [10] Moreover, these drugs are also hepatotoxic and this further adds to the decreased utilization of absorbed niacin and tryptophan. [2]
Inhibition of conversion of tryptophan to niacin by 5-fluorouracil leads to pellagra, and 6-mercaptopurine causes pellagra-like dermatitis by inhibiting Korenberg′s enzyme (NAD phosphorylase). [2],[10] Other drugs like phenytoin, chloramphenicol, azathioprine, and phenobarbital interfere in the tryptophan-niacin pathway and lead to pellagroid dermatitis [Table - 1]. The exquisite photosensitivity seen in pellagra may result from a deficiency of urocanic acid and/or cutaneous accumulation of kynurenic acid that may induce a phototoxic reaction. [6],[10]
Both of our patients did not have any lesion over V area of neck. Also, in contrast to classical pellagra, diarrhea was not a complaint in both of the patients. No affection of mental faculties was present in both the patients. Both the patients had hyperpigmented, hyperkeratotic plaques over dorsum of both hands and feet only. Cheilitis and glossitis were present only in the first patient.
Both of our patients were on antitubercular drugs. First patient was on ethionamide, while second patient was on ethionamide and isoniazid. Second patient had earlier taken isoniazid for one year without any adverse effects. In second patient, the symptoms of pellagroid dermatitis developed only after introduction of ethionamide. Ethionamide is more toxic than isoniazid and because of more toxicity, ethionamide is placed in second line of antitubercular drugs. [9] In second patient, probably the ethionamide had exacerbated the symptoms of pellagroid dermatitis.
Both of our patients responded well to nicotinamide supplementation, which confirms our diagnoses.
Unfortunately, pellagra is a diagnosis based on clinical picture because measurement of nicotinic acid level is a time-consuming and expensive procedure. However, treatment with nicotinic acid rapidly resolves diarrhea and confusion, with a gradual improvement in skin.
Isoniazid is the first-line drug for antitubercular therapy. When this drug was introduced, a lot of cases of pellagroid dermatitis came to light. Despite being coadministered with pyridoxine, there are case reports of development of pellagroid dermatitis in patients on isoniazid therapy. [11] Ethionamide is used only as a second-line drug for the management of tuberculosis. With the increase in multidrug-resistant tuberculosis patients especially on the backdrop of HIV infection, ethionamide-induced pellagroid dermatitis may be observed more frequently in the near future.
Nicotinamide is preferred over niacin in the treatment of pellagroid dermatitis because it does not cause vasomotor disturbances (flushing, itching, or burning). Nicotinamide is administered orally 100 to 300 mg/day in three divided doses [12] with vitamin B complex supplementation and high protein diet. A dose of 100 mg three times a day can be given by intramuscular route. In patients of encephalopathy, 1000 mg of nicotinamide is recommended orally in addition to 100 to 200 mg parenterally. Neurological symptoms disappear within 24 to 48 hours, but skin lesions may take 3 to 4 weeks to resolve. [13] Maintenance dose with nicotinamide 50 to 100 mg/day is recommended.
These cases highlight lichen simplex chronicus-like presentation of ethionamide-induced pellagroid dermatitis.
| 1. |
Stratigos JD, Katsambas A. Pellagra: A still existing disease. Br J Dermatol 1977;96:99-106.
[Google Scholar]
|
| 2. |
Karthikeyan K, Thappa DM. Pellagra and Skin. Int J Dermatol 2002;41:476-81.
[Google Scholar]
|
| 3. |
Hegyi J, Schwartz RA, Hegyi V. Pellagra: Dermatitis, dementia, and diarrhea. Int J Dermatol 2004;43:1-5.
[Google Scholar]
|
| 4. |
Park K. Nutrition and health. In: Park K, editor. Park's Textbook of Preventive and Social Medicine. 20 th ed. Jabalpur, India: M/S Banarsidas Bhanot Publishers; 2009. p. 526-80.
th ed. Jabalpur, India: M/S Banarsidas Bhanot Publishers; 2009. p. 526-80.'>[Google Scholar]
|
| 5. |
Milne MD, Crawford MA, Girao CB, Loughridge LW. The metabolic disorder in Hartnup disease. Q J Med 1959;28:407-21.
[Google Scholar]
|
| 6. |
Dogliotti M, Liebowitz M, Downing DT, Strauss JS. Nutritional influences of pellagra on sebum composition. Br J Dermatol 1977;97:25-8.
[Google Scholar]
|
| 7. |
Castiello RJ, Lynch PJ. Pellagra and carcinoid syndrome. Arch Dermatol 1972;105:574-7.
[Google Scholar]
|
| 8. |
Sjoerdsma A, Weissbach H, Udenfriend S. A clinical, physiologic and biochemical study of patients with malignant carcinoid (argentaffinoma). Am J Med 1956;20:520-32.
[Google Scholar]
|
| 9. |
Sweetman SC. Nutritional agents and vitamins. In: Sweetman SC, editor. Martindale the complete drug reference. 36 th ed. London: RPS Publishing; 2009. p. 1957-9.
[Google Scholar]
|
| 10. |
Hendricks WM. Pellagra and pellagra like dermatoses. Etiology, differential diagnosis, dermatopathology and treatment. Semin Dermatol 1991;10:282-92.
[Google Scholar]
|
| 11. |
Darvay A, Basarab T, McGregor JM, Russell-Jones R. Isoniazid induced pellagra despite pyridoxine supplementation. Clin Exp Dermatol 1999;24:167-9.
[Google Scholar]
|
| 12. |
Soni BP, McLaren DS, Sherertz MF. Cutaneous changes in nutritional disease. In: Freedberg IM, Eisen AZ, Wolf K, Austen KF, Goldsmith LA, Katz SL, editors. Dermatology in General Medicine. 5 th ed. New York: McGraw-Hill; 1999. p. 1725-37.
[Google Scholar]
|
| 13. |
Gopalan C, Jaya Rao KS. Nutritional deficiencies in man: Niacin. In: Recheigl J, editor. Section E. Nutritional disorders. West Palm beach, FL: CRC press; 1978. p. 23-31.
[Google Scholar]
|
Fulltext Views
3,345
PDF downloads
1,485





