Translate this page into:
Experimental study and clinical observations of autologous hair follicle cell transplants to treat stable vitiligo
2 Department of Dermatology, The First Affiliated Hospital of Bengbu Medical College, Anhui, China
Correspondence Address:
Ru-Zhi Zhang
The Third Affiliated Hospital of Soochow University, Changzhou 213003
China
| How to cite this article: Shi HX, Zhang RZ, Xu B, Xu CX, Li D, Wang L, Xiao L. Experimental study and clinical observations of autologous hair follicle cell transplants to treat stable vitiligo. Indian J Dermatol Venereol Leprol 2020;86:124-133 |
Abstract
Background: Vitiligo is characterized by the loss and/or dysfunction of melanocytes in the skin and has a profound impact on the social interactions of patients. Although there are many treatment options for vitiligo, the outcome is frequently unsatisfactory, especially for patients with stable vitiligo.
Objectives: To study the biological properties of melanocytes derived from human hair follicles and to observe the efficacy of using transplants of autologous hair follicle cells to treat patients with stable vitiligo.
Methods: From February 2014 to March 2017, 26 patients with stable vitiligo, who were refractory to all current routine therapy, underwent treatment with transplanted autologous hair follicle cells. The skin graft from each patient's occipital region was trimmed to remove excess adipose tissue and some of the upper part of the dermis. The remaining tissue, including hair follicles and dermal papillae, was cut into pieces and incubated in collagenase type IV and then in trypsin-ethylenediaminetetraacetic acid solutions. The cells were recovered, resuspended in the patient's own serum and then applied to the recipient area. Clinical observations continued for 6 months to 1 year. Laboratory experiments were also performed during this time on scalp specimens obtained from normal human volunteers. Cells migrating from the outer root sheath and the dermal papillae at various times of culture were observed using a microscope.
Results: Most of the repigmentation in the vitiligo areas appeared within 8 weeks of transplantation of autologous hair follicle cells. Early skin repigmentation was not uniform and appeared more repigmented than the surrounding normal skin. As time went by, the repigmentation became more obvious and matched the color of the skin around the lesion. Most of the pigmentation presented as a diffuse pattern and was not localized around the hair follicles. Among the 26 patients, 9 (34.6%) achieved excellent repigmentation, while 13 (50.0%) had good, 3 (11.5%) fair and 1 (3.9%) poor repigmentation. During the follow-up visit at 1 year, no excess hair growth was observed in the recipient areas and there was no scarring or ulcer formation in the donor or recipient areas. In the experimental part of the study, many keratinocytes, melanocytes and fibroblasts migrated from the adherent outer root sheath. In later subcultures using a specialized medium, pure melanocytes were obtained that had a strong proliferative capacity and had bipolar or poly-dendritic shapes. On the other hand, cells from the dermal papillae grew radially in primary culture and were almost fibroblast-like. However, a few bipolar melanocytes appeared in the later stage of culture.
Conclusions: The results of our study show that transplantation of autologous hair follicle cells is a simple and effective method to treat patients with stable vitiligo. Hair follicles (especially the outer root sheath) harbor many melanocytes with potential proliferative ability.
Limitations: There are a few limitations of the present study: a small sample size, a short follow-up period, no cell counting or viability testing.
Introduction
Vitiligo is characterized by depigmented macules that result from an absence or reduction in the number of epidermal melanocytes in the skin and/or mucous membranes.[1] According to the revised classification/nomenclature of vitiligo, the disorder can be classified as nonsegmental vitiligo, segmental vitiligo, mixed or unclassified.[2] Treatment efficacy depends on the activity of the disease and the extent of the affected areas.[3] Various medical treatment modalities are available, such as combinations of ultraviolet A light therapy and a psoralen medication, narrowband ultraviolet B phototherapy, topical and systemic steroids, topical calcineurin inhibitors, etc.[4]
In recent years, surgical techniques have proven to be effective in treating patients with stable vitiligo. Generally, grafting procedures can be divided into tissue grafting (full-thickness punch grafts, thin dermoepidermal grafts and suction-blister grafts) and cellular grafting (cultured pure melanocytes, noncultured melanocytes and keratinocytes).[5] Noncultured cellular grafting was introduced in 1992 and since then, various modifications to the original technique have been described. The advantages of noncultured cellular grafting include a higher repigmentation rate with a larger recipient-to-donor surface area ratio, an improved textural outcome and a faster turnaround time without the need for cell cultivation.[6]
After hair follicle morphogenesis, various types of stem cells are maintained in the follicle stem cell niches, such as epithelial stem cells (with CD34 and CD49f-positive markers) in the bulge region of the permanent portion, neural crest-derived melanocyte precursors in the bulge and sub-bulge regions of the follicular permanent region, and multipotent mesenchymal precursors in the dermal papilla.[7] The presence of a melanocyte reservoir appears to be the essential factor determining the success of medical interventions for vitiligo. The main reservoir of melanocytes exists within hair follicles, particularly in the outer root sheath.[8] Based on that fact, a method using an autologous noncultured outer root sheath hair follicle cell suspension was recently introduced as a novel cellular graft technique for the treatment of vitiligo.[9] Moreover, the dermal papilla and hair matrix contain many types of stem cells and cytokines that are beneficial to the survival and proliferation of the transplanted cells. So, the purpose of our study was to evaluate the therapeutic efficacy of autologous hair follicle cell transplants to treat stable and stubborn vitiligo.
Methods
Patient selection
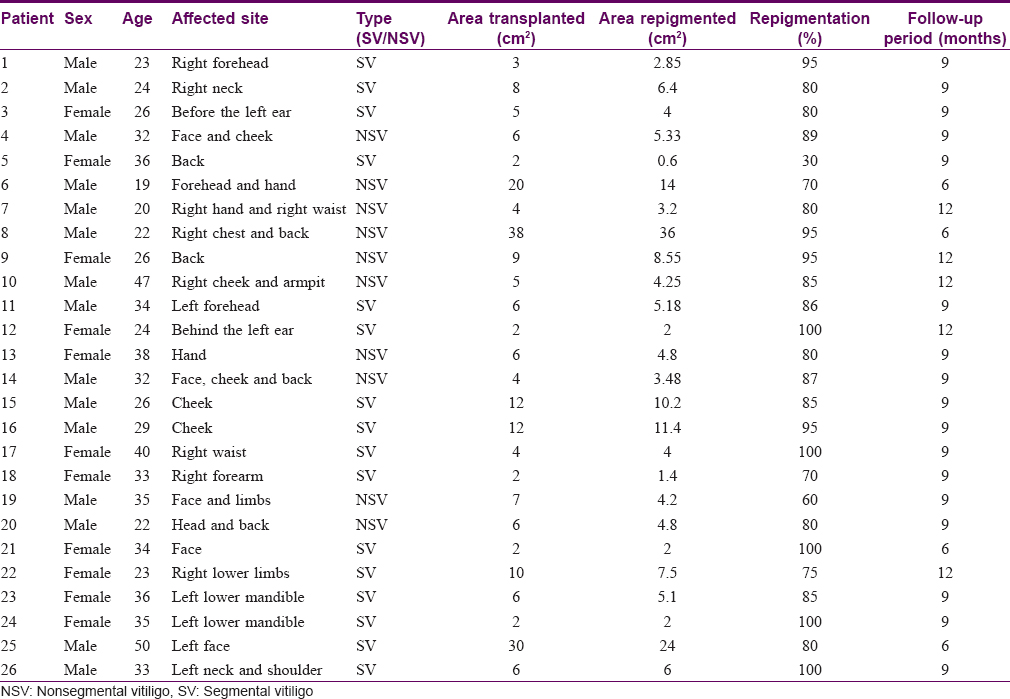
The study was conducted in an outpatient clinic of the Department of Dermatology at the Third Affiliated Hospital of Soochow University in Changzhou, China and was approved by the Hospital Ethics Review Board. From February 2014 to March 2017, 26 patients with stable vitiligo were selected according to the inclusion and exclusion criteria noted below and received autologous hair follicle cell transplants. Patient inclusion criteria were as follows: (1) all patients volunteering to participate in the study (2) age of the patients between 18 to 50 years (3) patients with stable vitiligo (i.e. no new lesions and no existing lesions which had progressed in the past 1 year, and no Koebner phenomenon), who were refractory to all routine treatment (topical and systemic steroids, topical calcineurin inhibitors and narrowband ultraviolet B phototherapy) and (4) patients who had not received any form of treatment over the past 6 months. Patient exclusion criteria were as follows: aged <18 years or >50 years, those with progressing lesions, a history of Koebner phenomenon, keloidal tendency, a history of bleeding diathesis, patients with unrealistic expectations, and pregnant patients. All subjects received only autologous hair follicle cell transplants and no topical or systemic treatments were given after surgery. Clinical photographs were taken at each visit. A total of 26 patients with stable vitiligo participated and completed the study. Among them, there were 15 males (57.7%) and 11 (42.3%) females. The average age was 30 ± 7.8 years (ranging from 19 to 50 years). Of 26 cases, 16 suffered from segmental vitiligo, which accounted for the majority (61.5%), with the other 10 cases were having nonsegmental vitiligo. The demographic characteristics and the responses of these patients are given in [Table - 1].
Preparation of skin grafts from the occipital area
In each patient, the occipital area was chosen as the donor site, with the size of the specimen depending on the clinical need. A scalp specimen containing 15–25 hair follicles is sufficient to cover an approximately 20–25 cm2 area to be transplanted. The donor area was shaved and sterilized three times with povidone–iodine, and then was anesthetized with a 2% lignocaine solution [Figure - 1]a. With the help of ophthalmic scissors and tweezers, the skin graft was obtained, placed in a sterile container containing phosphate-buffered saline and transferred to the laboratory. The scalp incisions were then sutured and bandaged [Figure - 1]b.
 |
| Figure 1: |
Preparation of autologous hair follicle cell suspensions
All the following steps were performed on a clean bench. Each scalp specimen containing hair follicles was removed from the sterile container and was placed into another container containing povidone–iodine solution for 5 min, and then was thoroughly washed with phosphate buffer saline containing 400 U/mL penicillin and 400 μg/mL streptomycin. After removing the excess adipose tissue and some of the upper part of the dermis, the remaining tissue, which contained the hair follicles and dermal papilla, was cut into pieces and incubated at 37 degrees celsius, and 5% CO2 in 0.25% collagenase type IV solution for approximately 2 h to free the hair follicles. Those hair follicles were subsequently incubated with 0.25% trypsin–0.05% ethylenediaminetetraacetic acid at 37 degrees Celsius and, 5% CO2 for 30 min to prepare single-cell suspensions. Phosphate buffer saline supplemented with each patient's own serum was used to terminate the digestion process. The mixture was filtered through a 200-μm filter to obtain the cell suspension, which was then centrifuged at 1000 rpm for 5 min. Ultimately, each cell pellet was resuspended in the patient's own serum and was transferred to our operating theater for transplantation.
Cell suspension transplantation
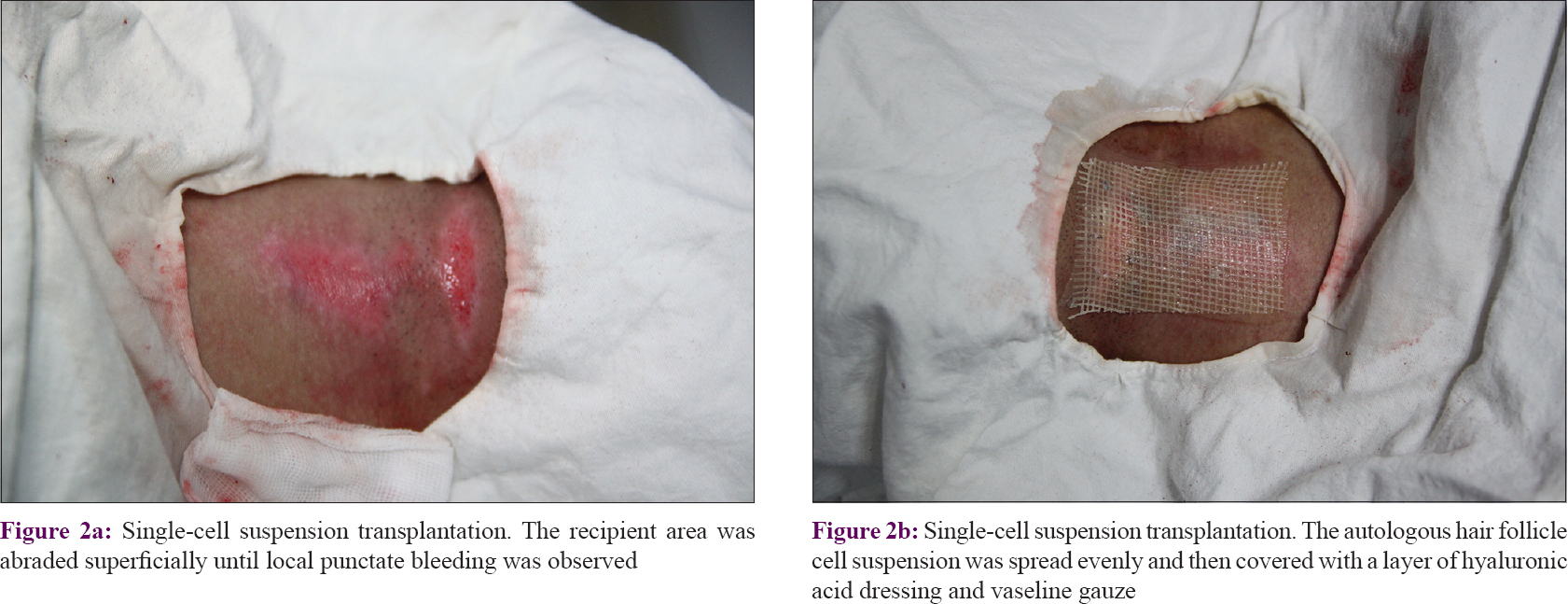
Each recipient area was shaved, sterilized with povidone–iodine, anesthetized with 2% lignocaine, and then abraded superficially using a motorized dermabrader or a CO2 laser until local punctate bleeding was observed [Figure - 2]a. With the help of a micropipette, the prepared single cell suspension was applied evenly onto the abraded vitiliginous area. A layer of hyaluronic acid dressing was gently spread on the surface and covered with a layer of vaseline-treated gauze [Figure - 2]b. Finally, a local pressure dressing was applied to prevent the cell suspension from leaving the recipient area. It is noteworthy that autologous serum was added to the dressing in order to increase the viability of the transplanted cells. After resting for 60 min, the patient was allowed to go home and was advised to avoid strenuous exercise. Eight days later, each patient returned to the clinic for removal of the dressings and for observations.
 |
| Figure 2: |
Follow-up and assessment
After the transplantation procedure, regular follow-up visits were carried out, usually on the 8th day, at 4, 8 and 12 weeks, and at 6 months and 1 year. The repigmentation of vitiligo lesions in all patients was observed and photographed. The degree and pattern of repigmentation and the color matching to the perilesional skin was evaluated. The response to transplantation was graded as follows: <50%, poor repigmentation; 50–74%, fair repigmentation; 75–89%, good repigmentation and 90–100%, excellent repigmentation.[10] The pattern of repigmentation was classified as: diffuse, perifollicular or dotted. The matching of the repigmented color with the perilesional surrounding area was described as: same, slightly darker or slightly lighter.
Experimental part
Treatment of specimens
Normal scalp specimens from regular plastic surgery procedures in our hospital were used for the preparation of hair follicles and their cells with prior consent of each patient. These specimens were trimmed regularly along the direction of hair growth. Excessive hairs, subcutaneous fascia tissues and adipose tissues were removed manually with scissors. Care was taken as much as possible to avoid damage to the hair bulbs during the actual operation.
Culture of human hair follicles and their cells
Each scalp specimen was immersed in povidone–iodine solution for 5 min and was then washed by phosphate buffer saline supplemented with 400 U/mL penicillin and 400 μg/mL streptomycin, and then transferred into Dulbecco's modified Eagle medium containing 10% fetal bovine serum. After carefully removing subcutaneous adipose tissue and some of the upper dermis, the residual tissue was trimmed into thin and long strips, was incubated at 37 degrees Celsius and, 5% CO2 in a 0.25% collagenase type IV solution for approximately 2 h and then in 0.25% dispase II solution for 0.5 h. Hair follicles were then pulled out or squeezed gently with eye scissors along the direction perpendicular to the hair follicles. Care was taken not to damage the dermal papilla or outer root sheath during the procedure. Free hair follicles were transferred into six-well plates coated with fetal bovine serum, and a small amount of medium was added into those plates 2 h later. After culture at 37 degrees Celsius and 5% CO2 for 3 days, the medium was changed for the first time and then was changed every 3 days. About 2 weeks later, the cells had become confluent and were passaged.
Results
Subjective description of the patients
The follow-up period ranged from 6 to 12 months, with an average of 9 months. All patients stated that they followed the doctor's advice and tried to keep the transplanted areas as inactive as possible, and that they did not feel any discomfort, such as local pain or itching, at the first follow-up visit at day 8. The dressing could be easily removed without any local bleeding or erosion. Most patients reported that they saw some repigmentation appearing in their original “white area” within 8 weeks of transplantation. Three patients observed repigmentation within 4 weeks, and two patients saw repigmentation within 12 weeks. The early repigmentation was not even and presented as hyperpigmentation in the vitiligo area compared with the perilesional skin color.
Areas which were not covered with the cell suspension did not show any repigmentation. As time went by, the repigmentation became more obvious and even matched the nearby perilesional skin color. According to the survey data, most patients reported high levels of satisfaction with this therapy.
Objective evaluation by photographs
In most subjects, repigmentation occurred within 2 months after transplantation of the cells. The age of the patient and the location of the lesion(s) affected the timing and extent of the repigmentation. Patients whose age was <30 years seemed more likely to obtain a good response, although we did not do a statistical analysis of that. The most obvious factor that affected repigmentation was the location of the lesion. Compared with other affected areas, repigmentation of lesions on the face and neck appeared earlier and was more obvious. Moreover, the repigmentation in facial lesions was better than those in the neck.
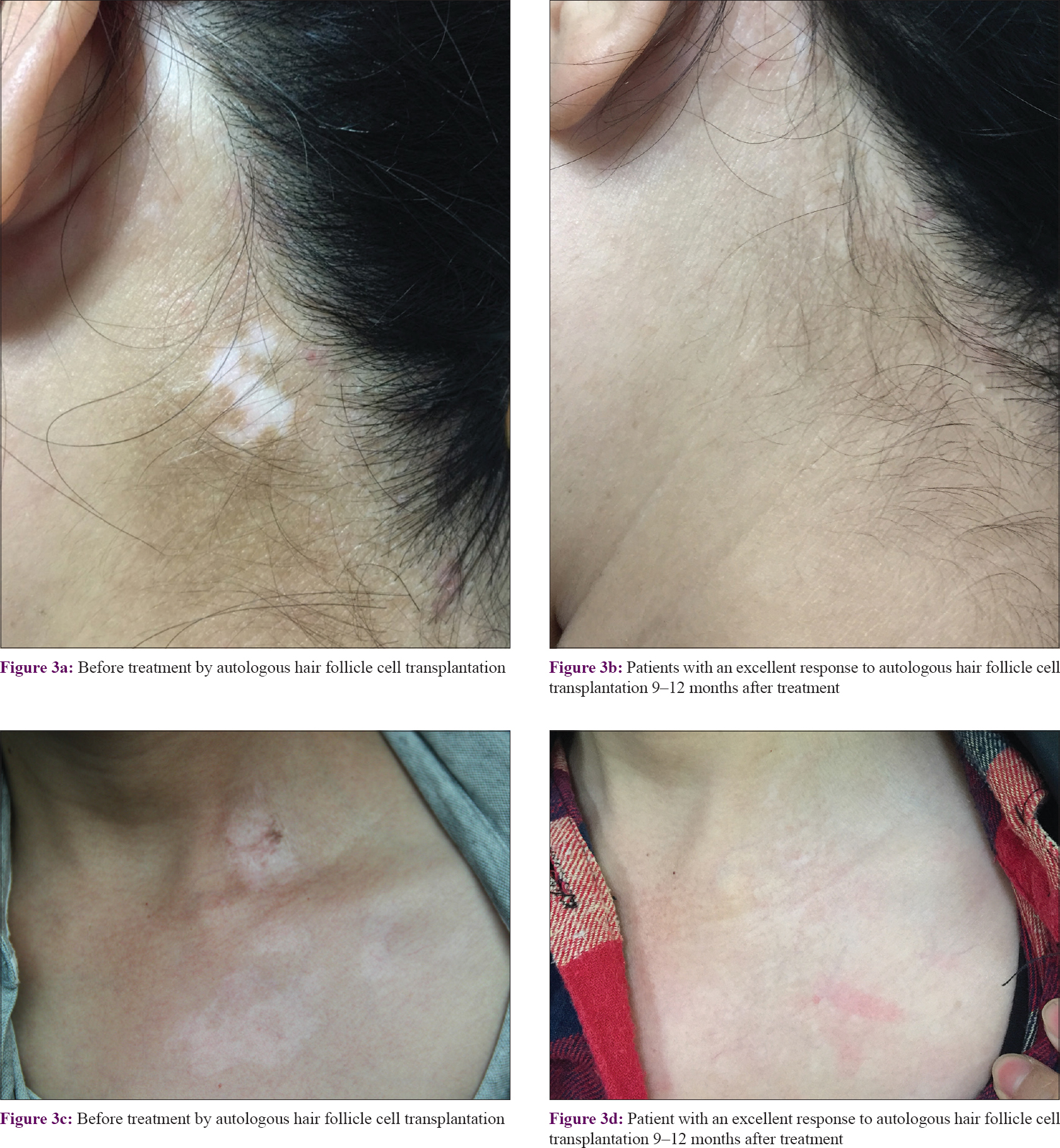
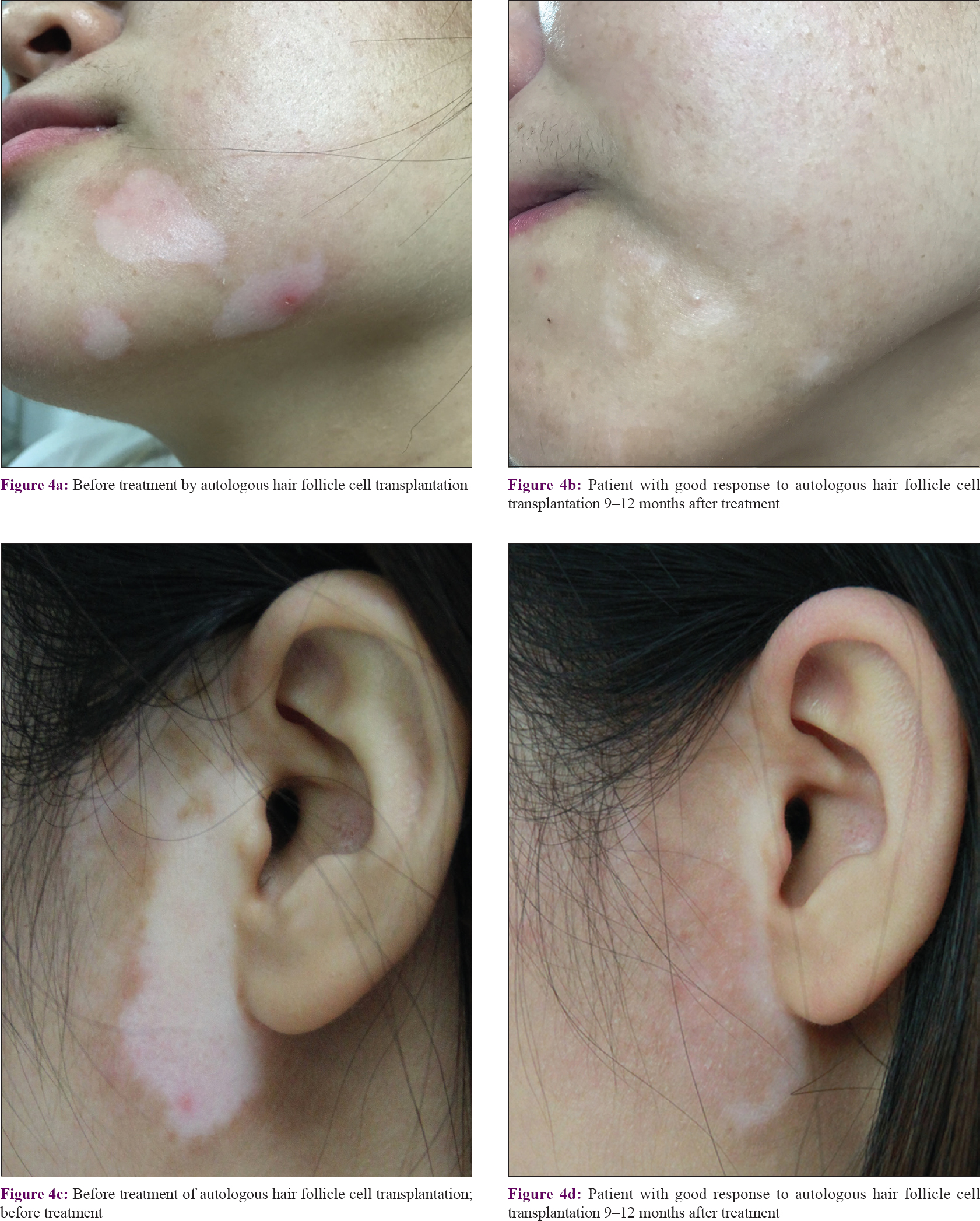
Among the 26 patients, 9 (34.6%) had an excellent response (90–100% repigmentation) [Figure - 3], 13 (50.0%) had a good response (75–89% repigmentation) [Figure - 4], 3 (11.5%) had a fair response (50–74% repigmentation) and 1 (3.9%) had a poor response (<50% repigmentation). Most of the pigmentation presented as a diffuse pattern and was not localized around the hair follicles.
 |
| Figure 3: |
 |
| Figure 4: |
At the 1-year follow-up visit, no excess hair growth on the recipient areas was observed. No scars or ulcer formation in the donor or recipient areas were observed. During the entire follow-up period, none of 26 patients had new lesions of depigmentation that occurred at a repigmented area.
Experimental part
Culture of hair follicles
Six-well plates coated with a small volume of fetal bovine serum were used for the culture of hair follicles and dermal papilla. A small amount of medium was added gradually to avoid the floatation of hair follicles. The attached hair follicles were roughly composed of the hair shaft, the inner root sheath, the outer root sheath and the hair bulb. The bulge region was recognized in the outer root sheath of upper hair follicles, where more cells were migrating. Those cells mainly included keratinocytes, fibroblasts and melanocytes (bipolar melanocytes and poly-dendritic melanocytes). Among them, keratinocytes accounted for the majority of cells and showed a cobblestone-like morphology [Figure - 5]a. On the other hand, some bipolar melanocytes were arranged in a fish-school appearance among the keratinocytes [Figure - 5]b. These findings indicated that these melanocytes came from the same ancestor and had a strong proliferative capacity. Compared with epidermal poly-dendritic melanocytes, they were hypopigmented and immature, appearing as amelanotic melanocytes. Meanwhile, a few pigmented melanocytes with three or more dendrites were seen.
 |
| Figure 5: |
Each dermal papilla appeared as a microsphere from which many fibroblast-like cells grew radially. The cells were tiny and compact in the center and relatively sparse and large at the edge zone of each dermal papilla [Figure - 6]a. In later cultures, some melanocytes were seen in the edge area. Because it is known that no melanocytes or their ancestors exist in the dermal papilla, those presumably came from the hair matrix, which is immediately adjacent to the dermal papilla [Figure - 6]b.
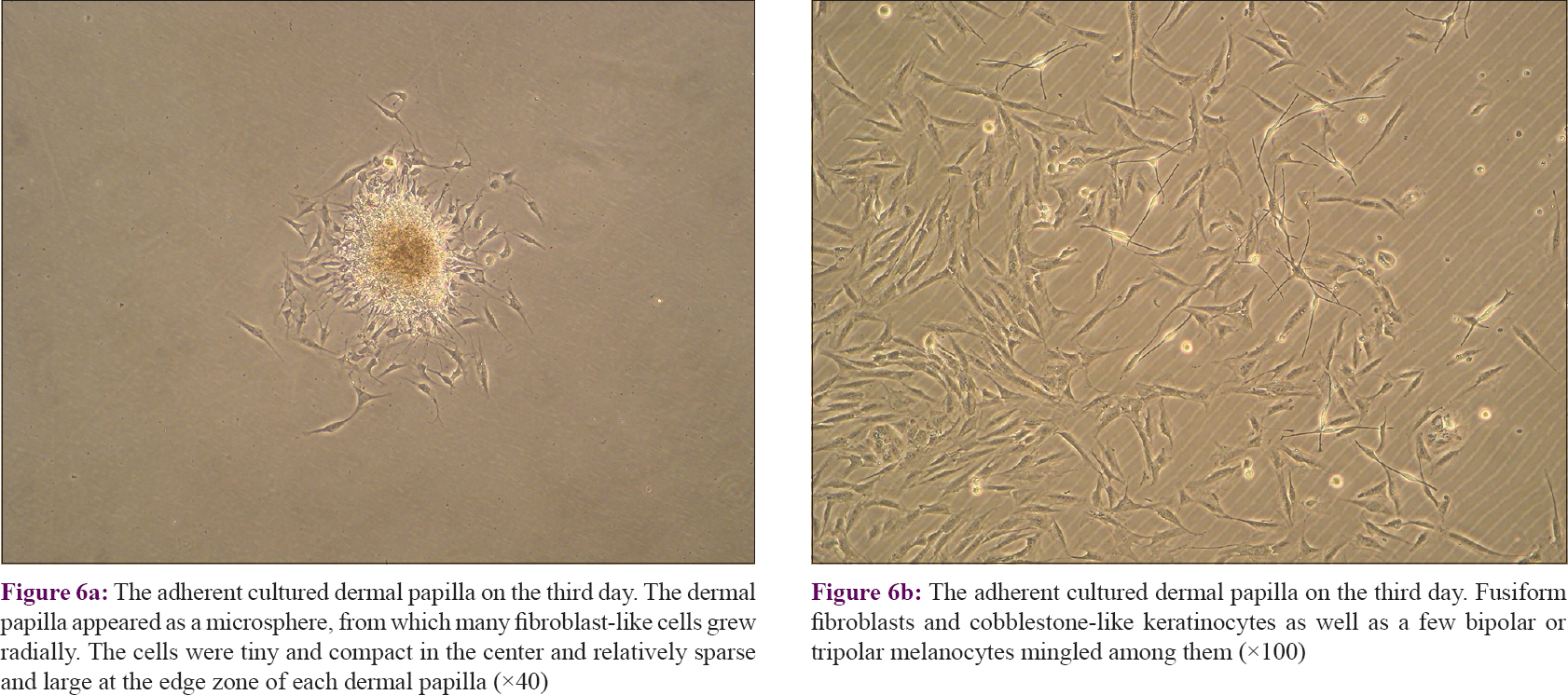 |
| Figure 6: |
Subculture of hair follicle cells
When cells from the hair follicles reached 80–90% confluence, a digestion solution containing 0.05% trypsin–0.02% ethylenediaminetetraacetic acid was used to dissociate the cells for the next passaging. The individual cells were incubated in a commercially available melanocyte medium, M254 supplemented with 2.5% fetal bovine serum, 2.5 ng/mL epidermal growth factor and 25 μg/mL bovine pituitary extract. To eliminate contaminating fibroblasts, 100 μg/mL geneticin (G418 sulfate) was added. When the second generation of cells (mainly consisting of keratinocytes and melanocytes) reached confluence, differential trypsinization was used to remove keratinocytes as follows: The cells were treated with 0.05% trypsin–0.02% ethylenediaminetetraacetic acid for 4 min. The dendrites of melanocytes could be observed to shrink and some of them began to break away from the bottom of the culture plate, while the keratinocytes were still tightly adhered to the wells. At that time, phosphate-buffered saline containing 10% fetal bovine serum was added to terminate the digestion and the melanocytes in suspension were collected. The remaining melanocytes were further cultured for 7 days in the above medium. Cells in the third passage presented with smaller cell bodies with bipolar or three dendrites, namely amelanotic melanocytes [Figure - 7].
 |
| Figure 7: The purified melanocytes had bipolar or tripolar dendrites, a slight discrepancy in size, a smaller cell body and strong light refraction of the cytoplasm (×100) |
Discussion
Cellular transplantation methods for treatment of stable vitiligo have become more efficient of late. At present, transplantation of cultured and noncultured epidermal cell suspensions is most commonly used. However, the transplantation of autologous noncultured outer root sheath hair follicle cell suspension to treat stable vitiligo has recently attracted the attention of dermatologists due to its unique advantages.
Melanocytes in human skin reside in hair follicles and in the epidermis. Epidermal melanocytes are moderately pigmented and are poly-dendritic. However, melanocytes in hair follicles are morphologically and antigenically distinct, and mainly comprise amelanotic melanocytes and intensely pigmented melanocytes.[11] Amelanotic melanocytes usually exist in the outer root sheath and in the hair matrix, representing the melanocyte reservoir in the skin. Large pigmented melanocytes in the hair bulbs are responsible for the hair color.[12] In our study, many cells, including keratinocytes, fibroblasts and melanocytes, migrated from the attached hair follicles, especially from the bulge area of the outer root sheath. This finding indicates that cells with multiple potentials (including stem cells) exist within the bulge area. Amoh and Hoffman have demonstrated that hair follicle-associated-pluripotent stem cells located in the bulge area of the hair follicles can differentiate into a variety of cells, including melanocytes and keratinocytes.[13]
It is noteworthy that amelanotic melanocytes were typically seen in the primary cultures. Further subcultures revealed that these cells had a strong proliferative capacity. Under certain conditions, these cells can be activated to proliferate and migrate and play an important role in the repigmentation of vitiligo. In fact, Vanscheidt and Hunziker demonstrated that melanocytes and/or stem cells residing in the outer root sheath of plucked anagen scalp hair follicles can be used to treat depigmented skin.[14]
The dermal papilla not only regulates hair follicle development and growth but is also thought to be a reservoir of multipotent stem cells.[15] Our experimental study showed that the dermal papilla contains many highly active mesenchymal cells. Surprisingly, some melanocytes similar to amelanotic melanocytes in the outer root sheath were seen in primary cultures of the dermal papilla. Presumably, those melanocytes came from the hair matrix immediately adjacent to the dermal papilla. From another perspective, the environment of the dermal papilla is beneficial to the growth of melanocytes.
Over the past few years, many studies have been conducted on the treatment of vitiligo with transplants of hair follicle cell suspensions. It has been found that hair follicle cell suspensions contain more CD200-positive cells (a marker for hair follicle bulge stem cells), including melanocyte stem cells, keratinocyte stem cells and mesenchymal stem cells. As early as 2011, Mohanty et al. attempted to treat vitiligo with an autologous noncultured outer root sheath cell suspension of extracted hair follicles and they achieved good results.[16] They found that 9 of their 14 patients achieved >75% repigmentation, of which 8 achieved >90% repigmentation. In our procedure, we used skin grafts containing intact hair follicles as the source of melanocytes and those cell suspensions also included melanocytes, hair matrix components, dermal papilla, keratinocytes and many other types of skin cells. The hair matrix may contain amelanotic melanocytes, which has been confirmed by numerous studies. The dermal papilla harbors many types of stem cells which could secrete cytokines that are beneficial to the survival and proliferation of the transplanted cells. It is known that keratinocytes provide a large number of paracrine factors and adhesion molecules for melanocytes and play a dominant role in regulating melanocyte survival, proliferation and differentiation.[17] In addition to keratinocytes, many other types of skin cells, such as fibroblasts, adipocytes, endothelial cells, inflammatory cells and dermal mast cells, have been shown to support optimal melanocyte function, and have a role in vitiligo repigmentation.[18] As a result, 22 of our 26 patients achieved >75% repigmentation, and of those 9 achieved >90% repigmentation.
At present, the conventional epidermal cell suspension technique has already been optimized and is widely used. Although the epidermal cell suspension transplantation technique may be more economical, it requires the operator to master the skills of separating the epidermis from the dermis. In contrast, autologous hair follicle cell suspensions can be easily acquired using trypsin, and only a very small area of the scalp needs to be used to prepare more cell suspension, so it can be used to cover larger depigmented lesions.
Compared with the noncultured outer root sheath hair follicle cell suspension transplant technique, our autologous hair follicle cell transplant procedure seems to be a bit more complicated, because it requires the use of two enzymes and several digestion steps rather than only trypsin. Both techniques have advantages but the autologous hair follicle cell procedure has the specific advantage that it provides a good yield of melanocytes, melanocyte stem cells and other types of stem cells (keratinocyte stem cells and mesenchymal stem cells in the surrounding dermis and dermal papilla).[10] Thus, the autologous hair follicle cell transplant mixture not only contains melanocytes but also many other types of skin cells that may exert synergistic actions in the repigmentation process. Besides, the entire autologous hair follicle cell transplantation process can be completed by a dermatologist within 3–4 h and patient satisfaction toward the entire treatment process and outcome is fairly high.
In addition, our procedure also has other novelties and advantages. In contrast to traditional procedures, a layer of hyaluronic acid dressing, rather than a collagen dressing, is spread evenly over the surface of the abraded vitiliginous lesion. Hyaluronic acid is an extremely long and linear carbohydrate polymer that is ubiquitously present in the extracellular matrix, being a major component of the connective tissue.[19] The use of a hyaluronic acid dressing is beneficial to cell colonization, survival and proliferation. During preparation of the autologous hair follicle cell suspension, the cells obtained are resuspended in autologous serum to avoid rejection and to protect cell viability. Other aspects to be considered in patients treated with the autologous hair follicle cell suspension transplantation technique are scarring and patient satisfaction. No apparent scars were observed in any of our patients. Because the procedure is a minimally invasive and effective technique, the satisfaction of the patients and their compliance is good.
This study of the autologous hair follicle cell transplantation technique, although safe and effective, does have a few limitations. Cell counting and viability testing were not performed in our study. Although the proportion of viable cells and CD200+ cells have been demonstrated in previous studies, the autologous hair follicle cell suspension needs to be examined to see what cells are present, test their viability and quantify them to make the study more comprehensive and convincing. In addition, a small sample size and a short follow-up period are also shortcomings of the present study. The follow-up period ranged from 6 to 12 months, due to differences in the compliance of patients. However, we also observed that repigmentation of some participants' continued over the next few months. Hence, a larger clinical trial and a longer follow-up period are needed for further evaluation of its efficacy.
To summarize, the autologous hair follicle cell transplantation technique is safe and effective. Our study has demonstrated that the autologous hair follicle cell transplantation technique is a successful in the treatment of patients with stable vitiligo. To improve existing methods, we also designed our own innovations. Those innovations include the procedure of cell suspension preparation and the use of autologous serum and hyaluronic acid, which can simplify and reduce the time required to maintain biological activities and promote the survival and proliferation of melanocytes. Our procedure is an effective modification of existing methods to treat patients with stable vitiligo.
Acknowledgment
The authors are very grateful to Professor V.J. Hearing for help with the English-language editing.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patients have given their consent for their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
The authors are very grateful to Professor V.J. Hearing for help with the English-language editing. The work was supported by the National Natural Science Foundation of China (Grant No. 81673078) and Jiangsu Provincial Special Program of Medical Science (BL2014036).
Conflicts of interest
There are no conflicts of interest.
| 1. |
Verma R, Grewal RS, Chatterjee M, Pragasam V, Vasudevan B, Mitra D. A comparative study of efficacy of cultured versus non cultured melanocyte transfer in the management of stable vitiligo. Med J Armed Forces India 2014;70:26-31.
[Google Scholar]
|
| 2. |
Shokeen D. Management of vitiligo patients with surgical interventions. Cutis 2016;97:E27-9.
[Google Scholar]
|
| 3. |
Janowska A, Dini V, Panduri S, Macchia M, Oranges T, Romanelli M. Epidermal skin grafting in vitiligo: A pilot study. Int Wound J 2016;13 Suppl 3:47-51.
[Google Scholar]
|
| 4. |
Yao L, Liu Y, Song Y, Zhong S, Li S. Successful treatment of stable vitiligo by low-density cultured autologous melanocyte transplantation combined with narrowband ultraviolet B therapy. Dermatol Surg 2017;43:1281-7.
[Google Scholar]
|
| 5. |
Silpa-Archa N, Griffith JL, Huggins RH, Henderson MD, Kerr HA, Jacobsen G, et al. Long-term follow-up of patients undergoing autologous noncultured melanocyte-keratinocyte transplantation for vitiligo and other leukodermas. J Am Acad Dermatol 2017;77:318-27.
[Google Scholar]
|
| 6. |
Gan EY, Kong YL, Tan WD, Thng ST, Goh BK. Twelve-month and sixty-month outcomes of noncultured cellular grafting for vitiligo. J Am Acad Dermatol 2016;75:564-71.
[Google Scholar]
|
| 7. |
Asakawa K, Toyoshima KE, Ishibashi N, Tobe H, Iwadate A, Kanayama T, et al. Hair organ regeneration via the bioengineered hair follicular unit transplantation. Sci Rep 2012;2:424.
[Google Scholar]
|
| 8. |
Mulekar SV, Isedeh P. Surgical interventions for vitiligo: An evidence-based review. Br J Dermatol 2013;169 Suppl 3:57-66.
[Google Scholar]
|
| 9. |
Vinay K, Dogra S, Parsad D, Kanwar AJ, Kumar R, Minz RW. Clinical and treatment characteristics determining therapeutic outcome in patients undergoing autologous non-cultured outer root sheath hair follicle cell suspension for treatment of stable vitiligo. J Eur Acad Dermatol Venereol 2015;29:31-7.
[Google Scholar]
|
| 10. |
Shah AN, Marfatia RK, Saikia SS. A study of noncultured extracted hair follicle outer root sheath cell suspension for transplantation in vitiligo. Int J Trichology 2016;8:67-72.
[Google Scholar]
|
| 11. |
Ma HJ, Yue XZ, Wang DG, Li CR, Zhu WY. A modified method for purifying amelanotic melanocytes from human hair follicles. J Dermatol 2006;33:239-48.
[Google Scholar]
|
| 12. |
Anbar TS, Abdel-Raouf H, Awad SS, Ragaie MH, Abdel-Rahman AT. The hair follicle melanocytes in vitiligo in relation to disease duration. J Eur Acad Dermatol Venereol 2009;23:934-9.
[Google Scholar]
|
| 13. |
Amoh Y, Hoffman RM. Hair follicle-associated-pluripotent (HAP) stem cells. Cell Cycle 2017;16:2169-75.
[Google Scholar]
|
| 14. |
Vanscheidt W, Hunziker T. Repigmentation by outer-root-sheath-derived melanocytes: Proof of concept in vitiligo and leucoderma. Dermatology 2009;218:342-3.
[Google Scholar]
|
| 15. |
Driskell RR, Clavel C, Rendl M, Watt FM. Hair follicle dermal papilla cells at a glance. J Cell Sci 2011;124:1179-82.
[Google Scholar]
|
| 16. |
Mohanty S, Kumar A, Dhawan J, Sreenivas V, Gupta S. Noncultured extracted hair follicle outer root sheath cell suspension for transplantation in vitiligo. Br J Dermatol 2011;164:1241-6.
[Google Scholar]
|
| 17. |
Osawa M. Melanocyte stem cells. Stem Book. Cambridge (MA): Harvard Stem Cell Institute; 2008-2009.
[Google Scholar]
|
| 18. |
Birlea SA, Costin GE, Roop DR, Norris DA. Trends in regenerative medicine: Repigmentation in vitiligo through melanocyte stem cell mobilization. Med Res Rev 2017;37:907-35.
[Google Scholar]
|
| 19. |
Castanedo-Cázares JP, Cortés-García JD, Fuentes-Ahumada C, Martinez-Rosales K, Torres-Álvarez B. Repigmentation patterns induced by NB-UVB and their relationship with melanocytic migration and proliferation in vitiligo. Photodermatol Photoimmunol Photomed 2016;32:269-75.
[Google Scholar]
|
Fulltext Views
7,650
PDF downloads
4,871





