Translate this page into:
Gram-negative diplococci in vaginal smear mistaken for child sexual abuse
2 Dermatology and Venereology, All India Institute of Medical Sciences, New Delhi, India
Correspondence Address:
Seema Sood
Department of Microbiology, All India Institute of Medical Sciences (AIIMS), Ansari Nagar, New Delhi - 110 029
India
| How to cite this article: Sood S, Verma R, Mukherjee A, Mahajan N, Das BK, Kapil A, Gupta S, Sharma VK. Gram-negative diplococci in vaginal smear mistaken for child sexual abuse. Indian J Dermatol Venereol Leprol 2014;80:260-262 |
Sir,
Sexual abuse in children is associated with not only physical but also immense psychological trauma. A microbiological misdiagnosis of gonorrhoea in such cases may have additional social and legal implications.
A seven year old girl presented with complaints of vaginal discharge and redness over the genitalia for the last four years. She also complained of itching causing her to rub her genitalia against hard objects like the arm of a chair. She had an otherwise unremarkable medical history. The child lived with her parents and an older brother. Sexual abuse was denied by the parents and the child herself. On physical examination, mild inflammation was noted over the labia majora and labia minora. There was no frank vaginal discharge. Systemic examination was unremarkable. Vaginal swabs were collected in triplicate, for microscopy, culture and polymerase chain reaction (PCR). A Gram-stained smear from the first swab revealed numerous pus cells with both intracellular and extracellular Gram negative diplococci [Figure - 1]. Based on a clinical suspicion of gonorrhoea, the child was given cefixime 200 mg and azithromycin 500 mg as part of syndromic management for STD patients. She was asked to report after two weeks.
Culture on modified Thayer-Martin medium and chocolate agar grew smooth, glistening colonies which were oxidase positive but superoxal test negative. Rapid carbohydrate utilization test (RCUT) was put up to confirm the species of isolate. [1] Simultaneously, DNA extracted from the third swab sample and the clinical isolate was amplified by PCR targeting opa gene of N. gonorrhoeae. [1] Interestingly, PCR was negative for N. gonorrhoeae while RCUT results confirmed that the isolate was that of N. meningitidis [Figure - 2]. Inhibition in the patient sample was ruled out by β-globin PCR. [1] PCR targeting 230 bp crgA gene of N. meningitidis was performed on the swab sample and isolate. [2] A positive result was obtained from both, confirming the presence of N. meningitidis [Figure - 3]. Antimicrobial susceptibility testing was put up as per CLSI guidelines [3] and the isolate was found to be susceptible to ciprofloxacin, cotrimaxazole, chloramphenicol, azithromycin, rifampicin, penicillin, ceftriaxone, cefotaxime and meropenem.
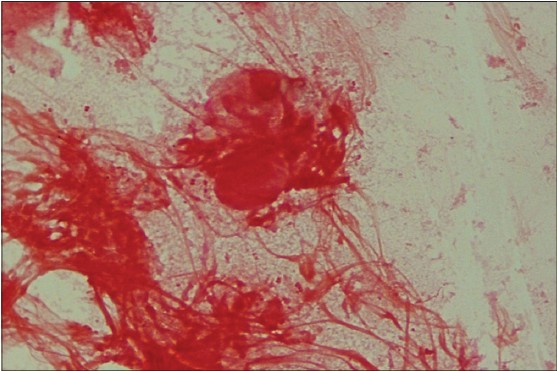 |
| Figure 1: Gram-stained smear of vaginal swab showing Gramnegative diplococci |
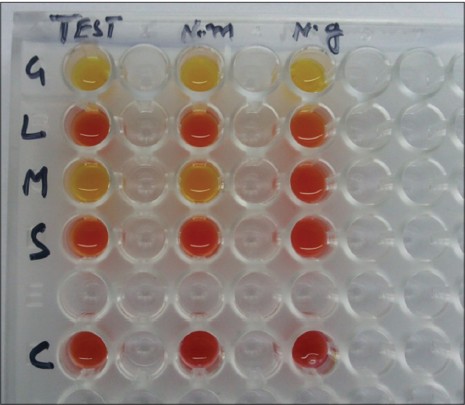 |
| Figure 2: RCUT result of the culture isolate (test) from vaginal swab in comparison to N. meningitidis (ATCC 13077) and N. gonorrhoeae (ATCC 49226) |
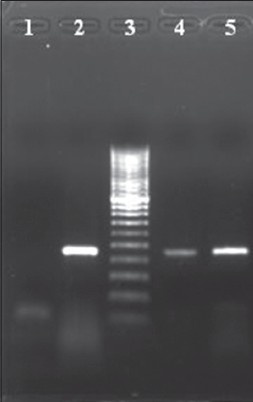 |
| Figure 3: crgA-gene based PCR result. Lane 1: Negative control, Lane 2: N. meningitidis (control strain with a band of 230 bps), Lane 3: 50 bps Ladder, Lane 4: DNA from vaginal swab, Lane 5: DNA from clinical isolate |
Additionally, during a follow-up visit, pharyngeal swabs were obtained in triplicate from parents, brother and the patient to check for colonization and processed as above. Microscopic examination revealed Gram negative diplococci in pharyngeal swabs from the patient, brother and mother while culture was negative in all. N. meningitidis was demonstrated in the pharyngeal swab of her brother by PCR [Figure - 4].
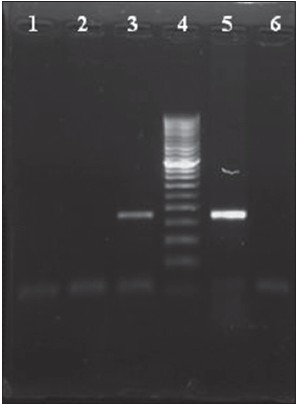 |
| Figure 4: crgA-gene based PCR result. Lane 1: Negative control, Lane 2: DNA extracted from pharyngeal swabs of father Lane 3: brother, Lane 4: 50 bps Ladder, Lane 5: DNA from N. meningitidis (control strain), Lane 6: DNA from pharyngeal swab of mother |
There was no significant improvement in the patient when she reported again after two weeks and she was prescribed a single intramuscular injection of ceftriaxone (125 mg). Other relevant investigations such as wet mount examination for Trichomonas vaginalis and Candida sp., VDRL test for syphilis and ELISA/rapid test for HIV were all negative. Thereafter, she was lost to follow-up and could not be contacted.
N. meningitidis is known to colonise the nasopharynx, especially during epidemics. [4] It has also been isolated from genitourinary sites [5] where it may cause infections clinically indistinguishable from those caused by N. gonorrhoeae including acute purulent urethritis, cervicitis, salpingitis and proctitis. This makes laboratory tests mandatory for accurate diagnosis and treatment. There are published reports of 84 patients from whom N. meningitidis was isolated from the cervix, urethra or anal canal. [5] Further, more severe symptoms have been described in women although the reason for this is not known. In this published case series, all female children presented with symptoms of vaginitis, similar to our patient, which is perhaps the first documented case from India.
Although an association between orogenital sex and acquisition of N. meningitidis has been suggested, we believe sexual abuse was unlikely in our patient. Meningococcal colonisation was detected in a throat swab from her sibling which points towards the availability of a reservoir. It is also possible that the child herself was a coloniser and developed vaginitis by auto-infection, though pharyngeal cultures were negative probably due to prior antibiotic therapy. Follow-up of such patients for extended periods may provide more definite information about the likely route of transmission.
Although antimicrobial resistance in N. meningitidis is not as serious an issue as in N. gonorrhoeae, the treatment for genital meningococcus is not established. There have been reports of spontaneous improvement, too. [5] The findings of the present case emphasize the importance of combining culture techniques and molecular diagnostic tests for confirmation of sexual abuse in children and are also helpful in situations where low burden of organism or prior antibiotic therapy precludes culture positivity. Overall, an interdisciplinary approach appears to be a valuable tool in evaluating such children.
| 1. |
Verma R, Sood S, Bala M, Mahajan N, Kapil A, Sharma VK, et al. Evaluation of an opa gene-based nucleic acid amplification test for detection of Neisseria gonorrhoeae in urogenital samples in North India. Epidemiol Infect 2012;104:2110-6.
[Google Scholar]
|
| 2. |
Taha MK. Simultaneous approach for nonculture PCR-based identification and sero group prediction of Neisseria meningitidis. J Clin Microbiol 2000;38:855-7.
[Google Scholar]
|
| 3. |
Clinical and Laboratory Standards Institute (CLSI). Performance standards for antimicrobial susceptibility testing: Twentieth informational supplement (Update) June 2010. Document M100-S20-U. Wayne, Pennsylvania: CLSI; 2010.
[Google Scholar]
|
| 4. |
Yazdankhah SP, Caugant DA. Neisseria meningitidis: An overview of the carriage state. J Med Microbiol 2004;53:821-32.
[Google Scholar]
|
| 5. |
Givan KF, Thomas BW, Johnston AG. Isolation of Neisseria meningitidis from the urethra, cervix, and anal canal: Further observations. Br J Vener Dis 1977;53:109-12.
[Google Scholar]
|
Fulltext Views
7,195
PDF downloads
895





