Translate this page into:
Gut microbiome in dermatology – A narrative review
Corresponding author: Dr. Varadraj Vasant Pai, Department of Dermatology, Goa Medical College, Bambolim, India. docpai@rediffmail.com
-
Received: ,
Accepted: ,
How to cite this article: Pai VV, Sarath AP, Kerkar Z. Gut microbiome in dermatology – A narrative review. Indian J Dermatol Venereol Leprol. doi: 10.25259/IJDVL_1094_2024
Abstract
The gut microbiome and human body have co-evolved in a synergistic host-microbial relationship. The ideal composition of human gut microbiota is an elusive concept, but every individual has a unique gut microbiota profile with regional differences. Newer diagnostic techniques have helped identify different bacteria and their roles in health and disease. The gut microbiome composition is affected by various factors like age, diet, immune system, environmental factors, exercise, and drugs. The microbiome has varied roles in metabolism, immune response, immune tolerance and antimicrobial protection. Diet plays an important role in maintaining the gut microbial diversity. Loss of homoeostasis in the microbiome results in dysbiosis. Dysbiosis plays a role in many dermatological diseases like atopic dermatitis, psoriasis, acne, rosacea, hidradenitis suppurativa, connective tissue disorders and many other systemic conditions like obesity, diabetes, neurological disease and malignancy. Reconstitution of the gut microbiome ecology in the form of bacteriotherapy with the reintegration of certain strains of microbiota has a beneficial role in many of these disorders.
Keywords
gut microbiome
dermatology
bacteriotherapy
Science has long been intrigued by human microbiota. During evolution, microbial colonisation in animals, including humans, has resulted in a complex host-microbial relationship, influencing their genotype and phenotype. This consequently has generated a wide range of interdependent physiologic activities for their well-being.1 The term microbiota has been used interchangeably with microbiome.2–4 The microbiome in humans is composed of bacteria, yeast and viruses that can impact both health and diseases.5
The gastrointestinal tract accounts for the largest portion of the microbiome with 100 trillion microbial cells, which is thrice the number of human cells. The host-environment interface of the gastrointestinal tract has a surface area of 30 m2, followed by the skin interface accounting for 25 m2.6,7
The dynamics of the microbiome of the skin and the gastrointestinal tract appear to be interlinked. This relationship has been described by the ancient Indians and the Greeks. The Indian system of Ayurveda recognises the primary location of vata dosha in the colon. The father of modern medicine, Hippocrates, famously asserted that the gut is the basis of overall health.8
In 1930, dermatologists Stokes and Pillsbury first hypothesised regarding the communication of the skin and gut with the brain. They theorised that emotional states might alter the normal gut flora and contribute to systemic inflammation and diseases like acne. These findings have been recently validated and have formed the basis of the gut-skin-brain axis.9,10
This review provides an overview on the gut microbiome ecology, factors affecting the gut microbiome leading to dysbiosis and its role in many dermatological conditions.
Human gut ecology
One of the major challenges for scientists with respect to the gut microbiome was the inability to culture these microorganisms. However, advances in identifying techniques like The Human Microbiome Project of 2007, which used DNA based sequencing methods, expanded our knowledge of the human microbiome.1 The areas of the healthy microbiome in humans include the gastrointestinal tract, skin, oral cavity, respiratory tract and vagina. Each of these sites has a unique microbial ecosystem that interacts dynamically with the host.4,11,12
The gut microbiome has been found to have more than 35,000 species of bacteria. These trillions of bacteria in the human colon form an extremely complex microbial ecosystem. The presence of extensive cellular and neural networks in the gut and their interactions with the other organs through chemical mediators is considered by many as the ‘second brain’.13–17
Gut microbiota variations
There is no ‘ideal and optimal’ gut microbiome composition, but every human has a fingerprint-like distinctive gut microbiome profile with functions including maintenance of the gut mucosal barrier, immune tolerance, antimicrobial actions and formation of metabolites required for host nutrient metabolism.16 The large intestine accounts for almost 70% of all the microbes in the human body.15,18 The predominant gut phyla are Bacteroidetes, Firmicutes, Proteobacteria, Actinobacteria and more. Out of these, Firmicutes and Bacteroidetes represent 90% of the gut microbiome.19 The Firmicutes phylum includes Lactobacillus, Staphylococcus and Streptococcus among others. Common pathogenic bacteria like Escherichia coli, Shigella and Helicobacteria belong to the Proteobacteria phylum, and Bifidobacterium and Bacterioides belong to the Actinobacteria and Bacteroidetes phylum, respectively.+
The distribution of microbiome in different parts of the gut is given in Table 1.20,21 In addition to the spatial difference, there is an axial change in microbes from the lumen to the mucosa of the intestine [Figure 1].15,22
| Stomach and duodenum | Fermicutes species like Lactobacillus, Staphylococcus, Streptococcus. Enterobacteriaceae, Helicobacter Pylori (proteobacteria phyla) |
| Jejunum and ileum | Lactobacillus, Streptococcus, Enterobacteriaceae, Bifidobacterium, Bacterioides |
| Colon | Lactobacillus, Streptococcus, Enterobacteriaceae, Bifidobacterium, Bacterioides, Clostridium, Fusobacteria |
| Skin |
Moist areas: Staphylococcus, (Actinobacteria) Corynebacterium and Proteobacteria Oily areas: Corynebacterium, Staphylococcus Cutibacterium Dry skin: Proteobacteria |
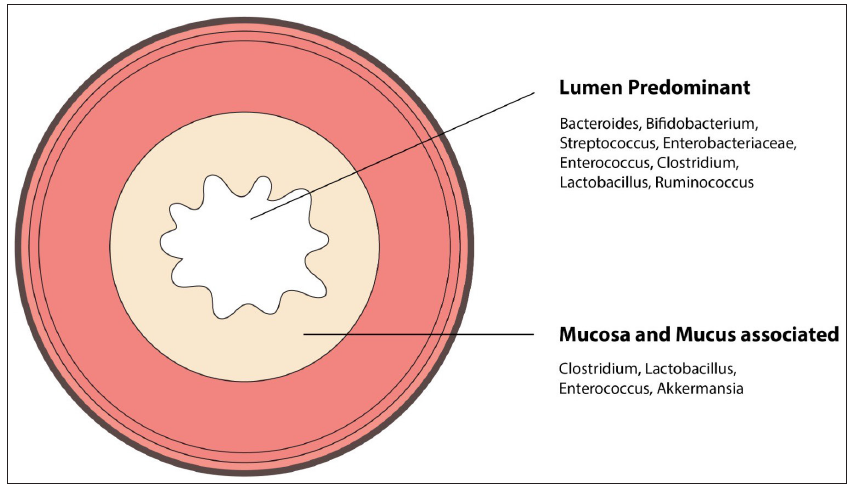
- Luminal and mucosal microbiome.
Factors affecting the gut microbiome composition
The entire gut microbiome can be divided into a stable microbiome that is highly constant over time, and an opportunistic microbiome which varies and can affect the immune system. The composition of the gut microbiome is affected by various factors like age, diet, immune system, environmental factors and exercise [Figure 2].16,23,24
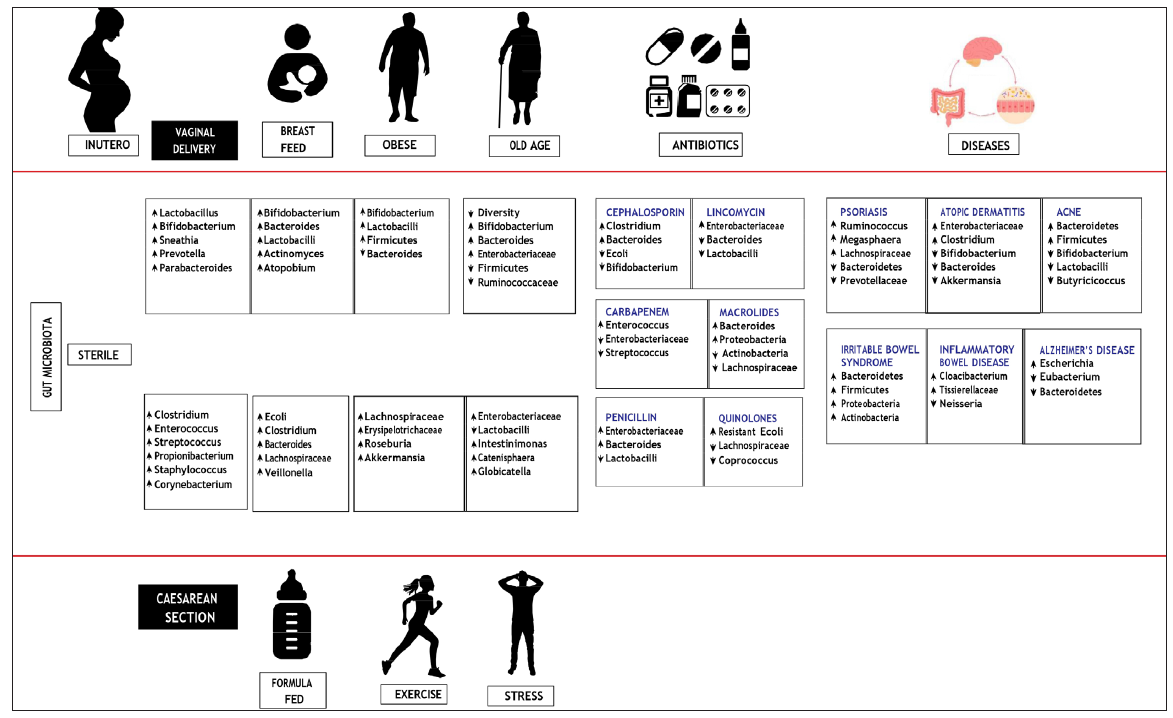
- Factors affecting the gut microbiome composition.
Functions of Gut microbiome
Metabolome
The larger number of genes of the gut microbiome as compared to the human genome results in immense functional superiority and versatility.25,26 The dietary carbohydrates nourish the gut microbiome with nutrients, the gut microbiome, in turn, ferments the complex carbohydrates present in non-digestible dietary fibres and intestinal mucus. This fermentation results in the formation of short chain fatty acids like butyrate, which are energy sources for the intestinal and liver epithelial cells.27 The various metabolomes associated with the gut microbiome are given in Figure 3.15,18,28–30
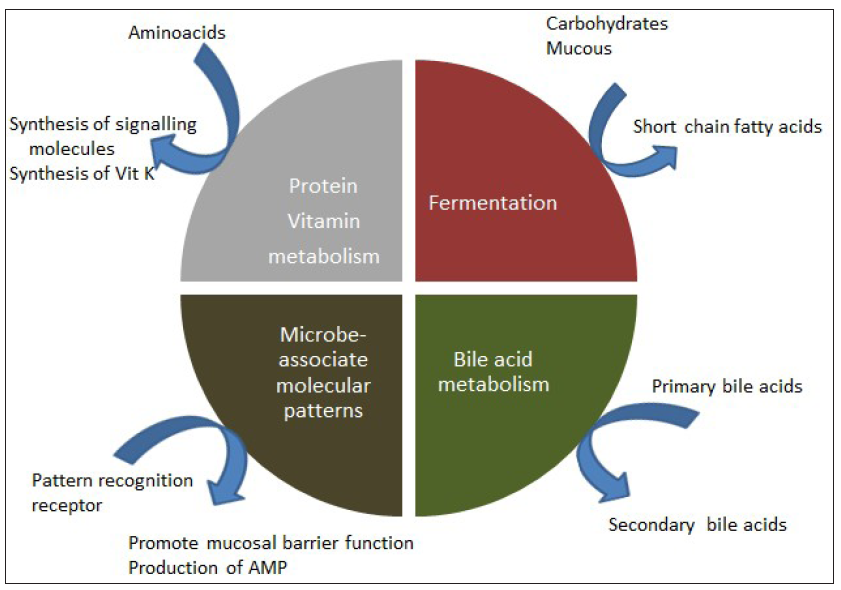
- Metabolomes of the gut microbiome.
Antimicrobial protection
The gut microbiome plays an important role in the education and improvement of the immune system.23 Via organism-specific microbe-associated molecular patterns such as peptidoglycan, lipopolysaccharides and bacterial nucleic acids, the gut microbiome constituents activate the pattern recognition receptor-mediated mechanisms (e.g. toll-like receptor) in the Paneth cells, which results in the synthesis of antimicrobial peptides (AMP) such as defensins, cathelicidin, calprotectin and so on. However, the gut homoeostasis can be disrupted due to overactive toll-like receptor (TLR) stimulation, leading to cytokine responses which can increase the risk of inflammatory and autoimmune disorders.15,31,32
Gut microbiome and diet
The food humans consume is a driving factor in the overall design and function of the gut microbiome. This microbial community interacts with the gut epithelium and immune system and maintains intestinal homoeostasis in a healthy state.31
Various dietary factors which affect the gut microbiome are given in Table 2.31,33–39 There are regional variations in diet and therefore in the gut microbiome. The data from studies comparing the gut microbiome of tribes from Papua New Guinea to samples isolated from the developed areas of the United States showed higher biodiversity and lower rate for autoimmune diseases with a higher average number of bacterial taxonomic units in the two tribes when compared with the samples from United States.17,35 Among the developed countries, the primary factor for decrease in beneficial microbes which help with fibre breakdown, is a diet rich in fat and protein and low in fibre. This can lead to an increase in pathogenic bacteria which can damage and disrupt the mucosal barrier causing systemic inflammation.34,37
| Dietary fibre | Dietary fibre is the main nutrient for maintaining the diversity of the gut microbiome. Most dietary fibres are fermentable (e.g. non-digestible oligosaccharides and polysaccharides, resistant starches and chemically synthesised carbohydrates). The sources of these fibres include plant-based foods such as fruits, vegetable, nuts, cereals, legumes, nuts and tubers broadly called prebiotics.34,35 The dietary fibres are not processed enzymatically in the small intestine but through fermentation by the gut microbiome in the large intestine to provide energy to the host. It maintains the integrity of the mucus barrier and eliminates the risk of pathogenic infection.36–38 |
| Sugar | A high sugar intake induces an increase in the mucus-degrading bacteria, which decreases the expression of tight junction proteins leading to increased gut permeability and inflammation.34 |
| Fats | A high-fat diet leads to reduced diversity of the gut microbiome in humans with an increased ratio of Firmicutes/Bacteroidetes. Fat-soluble vitamin D plays an important role in the regulation of gut microbiome and immune responses and has a protective role in the inflammatory bowel disease.36 |
| Proteins |
The quantity and source of dietary protein determines the amount and profile of bacterial metabolites. A high-protein diet shifts gut bacteria metabolism to protein fermentation and can disturb the gut mucosal homoeostasis. Plant protein has been reported to increase gut-commensal Bifidobacterium and Lactobacillus, while they additionally decrease the pathogenic Bacteroides fragilis and Clostridium perfringens and also produce useful metabolites like short chain fatty acids. High animal protein intake is associated with an increased risk of inflammatory bowel disease. Processed meat contains high amounts of sulphated amino acids with increased levels of trimethylamine-N-oxide, a proatherogenic compound that increases risk of cardiovascular disease.36,39–42 |
Gut-skin axis
The gut and skin share many functionally analogous features in terms of immunological, neuroendocrinal and metabolic properties.37 The gut-skin axis represents the sum total of the close association of the gut microbiome with skin. The concept was further expanded by Arck et al.34 in 2016, who defined the concept of the gut-skin-brain axis, wherein the three organs share complex signals which modulate various interactions between the gut microbiota, emotional states and systemic and skin inflammation. Disturbances in the gut-skin-brain axis homeostasis can contribute to the development of various dermatologic, gastrointestinal and neuro-psychiatric disorders.4,34,38 This intricate relationship requires an optimal and delicately balanced host response to prevent over utilisation of host resources by commensals while maintaining immune tolerance.40 Any pathologic alteration in gut microbial diversity results in ‘Dysbiosis’. This can make the host susceptible to many autoimmune and inflammatory dermatoses (e.g. acne, atopic dermatitis, psoriasis, hidradenitis suppurativa, rosacea, connective tissue disorders etc.) [Figure 4].41
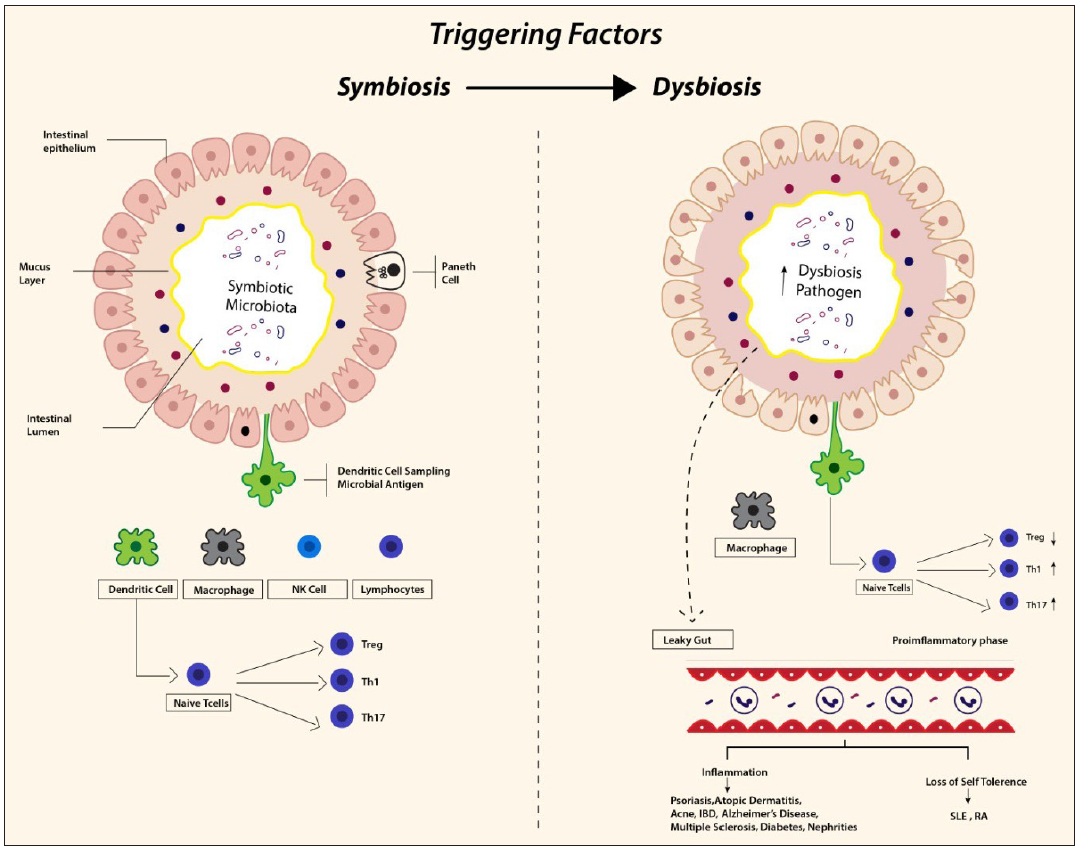
- Immunology of symbiosis and dysbiosis. NK: natural killer cells, SLE: systemic lupus erythematosus, RA: Rheumatoid arthritis.
Consequences of intestinal dysbiosis
-
1.
Inflammation – The inflammatory response of the gut epithelial membrane is systematically regulated by the epithelial cells, mucus layer, dendritic cells, natural killer cells, T-cells and macrophages [Figure 4]. Treg cells, through cytokines, like interleukin 10, transforming growth factor beta and interleukin 35, help in developing immune tolerance to self antigens and preventing autoimmune and inflammatory diseases. A balance in T helper type 1 and 2 cells activity is required for homoeostasis and preventing both chronic inflammation and allergic responses. Similarly, the T helper type17 cells, normally maintain the barrier function of the gastrointestinal tract, but when dysregulated, they contribute to the pathogenesis of diseases like psoriasis via interleukin 17 and interleukin 22.42–44
-
2.
Leaky gut – Another hypothesis is that gut inflammation can increase intestinal permeability and migration of bacteria or their metabolic products from the gut into the circulation. The effect on the skin can be in the form of reduced keratin synthesis and deranged epidermal differentiation. Studies have shown the presence of deoxyribonucleic acid of the gut bacteria in the bloodstream in patients with chronic skin diseases.9,38
-
3.
Neuroendocrine – The gut microbiome acts on neural pathways through the production of a variety of metabolomes, cortisol and neurotransmitters like gamma-amino butyric acid, serotonin and more. These molecules stimulate keratinocyte production of pro-inflammatory cytokines and enhance Langerhans cells immune responses, as seen in dermatoses like atopic dermatitis, psoriasis and rosacea.38,45
-
4.
Immunological – The gut microbial ecosystem has a wide range of diverse antigens coded by more than nine million genes as compared to the human genome of around 23,000 genes. In a healthy individual, this may programme the immune system to be tolerant towards innocuous stimuli and self-antigens. However, dysbiosis may provide a fertile ground for molecular mimicry between the self and bacterial antigens, which can activate the effector immune cells to attack self-antigens, resulting in an autoimmune response.38,46–49
Gut microbiome and dermatoses
Recent data shows an ever-increasing association between gut microbiome and various diseases like diabetes, obesity, neurological illness, malignancy and autoimmune diseases and also many inflammatory dermatoses like psoriasis, atopic dermatitis among others.50–55
Atopic dermatitis (AD) – The role of the gut microbiome has been widely studied in AD. While the ‘hygiene hypothesis’ in AD suggests that decreased diversity and microbial exposure in early life, results in loss of immunological tolerance and an increased prevalence of allergic diseases, it is the gut microbes that form the most important component of microbial exposures.56 The age of onset, phenotype, severity and remission of AD is associated with gut microbiome composition in the early life of an individual.57–60 Studies have suggested that patients with AD exhibit a decreased microbial diversity and alterations in both beneficial and harmful bacteria compared to healthy subjects [Figure 2].61–64
Psoriasis
Psoriasis is characterised by epidermal hyperproliferation and a dermal immune response in the form of an overactive tumour necrosis factor-alpha, interleukin-17 and interleukin-22 activity. The dysbiosis in psoriasis is akin to that seen in inflammatory bowel disease with an altered Firmicutes/Bacteroidetes ratio and depletion of beneficial bacteria in psoriasis patients.65,66
In patients with moderate-to-severe psoriasis, markers of intestinal barrier damage such as intestinal fatty acid-binding protein are significantly elevated which may explain the underlying metabolic abnormalities in these patients.67,68
A recent study by Li et al.that used the Mendelian randomisation strategy revealed that eight specific bacterial taxa at the genus level were suggestively associated with the risk of psoriasis.69
Acne
The exact mechanism by which the gut microbiome is involved in the pathogenesis of acne is not established, but an immune mechanism similar to psoriasis and AD may play a role. One of the possible mechanisms may be the mammalian target of rapamycin signalling pathway. Mammalian target of rapamycin is a nutrition sensitive regulator of lipogenesis and adipogenesis. Studies show an increased expression of mammalian target of rapamycin in patients with acne as compared to healthy controls. Also, a diet high in glycaemic index was associated with greater mammalian target of rapamycin signalling, resulting in increased sebaceous activity that may trigger acne.70–73
The gut-skin-brain axis may also play a role due in the stress-induced aggravation of acne due to increased production of neurotransmitters by bacteria.73–75
Rosacea
Though the exact role of Gut microbiome (GM) in rosacea is not known, overexpression of microbe-associated molecular patterns, TLR, AMP and mammalian target of rapamycin play an important role in human innate immune protection against bacteria is seen.41 A study evaluating the role of small intestinal bacterial overgrowth in rosacea revealed that treatment with rifaximin in these patients resulted in clinical remission of rosacea.41,76
Hidradenitis suppurativa (HS)
The role of gut microbiome in HS is suggested due to its shared pathogenetic mechanisms with inflammatory bowel disease (IBD) and also by the response of IBD to anti-tumour necrosis factor alpha therapies,76,77 Different studies have implicated varied species in HS which included an increase in Escherichia, Shigella and depletion of Faecalibacterium prausnitzii.78 HS symptoms can be aggravated by dairy products, brewer’s yeast, high sugar intake, carbonated drinks and saturated fat. A plant-based or the Mediterranean diet is one of the therapeutic approaches to mitigate inflammation in HS.79
Connective tissue diseases
Gut dysbiosis is noted in connective tissue disorders like systemic lupus erythematosus and systemic sclerosis. The pathogenesis involves crosstalk between the commensal microbiota, immune system and metabolites. Systemic lupus erythematosus patients show a reduced Firmicutes/Bacteroidetes ratio while reduced levels of Clostridiaceae are seen in patients with scleroderma. These microbes cause an increased production of pro-inflammatory free fatty acids and a reduction in short chain fatty acids, which are anti-inflammatory.80–82
Vitiligo
Recent studies have shown that an altered gut microbiome may contribute to vitiligo pathogenesis. Bzioueche et al. compared vitiligo patients with healthy controls and found gut dysbiosis via reduced richness and bacterial species distributions in patients with vitiligo.83,84
Vulvovaginal candidiasis
The crosstalk of bacterial strains between the gut and vagina stimulates both local and systemic immune responses with subsequent effects on the host. The Gram-positive bacilli Lactobacillus that dominates the vaginal microbiota is believed to originate from the gut. The intestinal flora is believed to be the source of infection in recurrent bacterial vaginosis and group B streptococcal infection in pregnancy.85,86
Role of prebiotics, probiotics and microbiome transplant
Bacteriotherapy refers to the therapeutic administration of commensal bacteria. This has been used in the management of many inflammatory and non-inflammatory diseases. It involves the reconstitution of the microbiome ecology with the reintegration of certain beneficial strains of microbiota.87
Probiotic consumption is one of the most commonly practiced methods of bacteriotherapy. Probiotics have been defined as ‘live microorganisms which, when administered in adequate amounts, confer a health benefit on the host’. The role of probiotics has changed from being a natural constituent of our diet to a therapeutic supplement. Consumption of probiotics induces significant colonisation by beneficial bacteria, which can effectively promote the formation of intestinal barrier while reducing allergic and inflammatory responses. Lactobacillus, Bifidobacterium and Streptococcus genera are the most commonly tested probiotics.88
Prebiotics refers to a ‘nondigestible food ingredient that beneficially impacts the host by selectively stimulating growth and/or activity of one or a limited number of bacteria already resident in the colon, and thus helps to improve host health’.32 These compounds are metabolised by microbiome of the colon after bypassing digestion and absorption in the upper intestine. ‘Synbiotics’ is a term introduced as a combination of probiotics and prebiotics.88–90 The term postbiotic has been recently defined as ‘a preparation of inanimate microorganisms and/or their components that confers a health benefit on the host’. It is composed of cell-free supernatants without metabolite specification/individualisation, cell wall components and/or intracellular compounds.91,92
Table 3 shows the details of studies of bacteriotherapy in dermatological conditions.93–105 A systematic review of probiotics and prebiotics in atopic dermatitis showed a mixed response with many studies showing a lower SCORAD score following consumption of oral probiotics, while few studies showed moderate or no response. Multistrain synbiotics and vitamin D3 supplements in addition to regular treatments have also shown to be effective in reducing severity of AD in infants.94,106,107
| Author | Disease | Probiotic | Results |
|---|---|---|---|
| Gerasimov et al.93 | Atopic dermatitis | Lactobacillus acidophilus Bifidobacterium lactis prebiotics |
a. 33.7% decrease in the SCORAD score b. Decrease in the requirement of topical corticosteroid |
| Navarro-López et al.94 | Atopic dermatitis | Bifidobacterium lactis Bifidobacterium longum Lactobacillus casei | a. Change of ˗83% in the probiotic group and ˗24% in the placebo group (P < .001) was noted |
| Gilli et al.95 | Psoriasis | Lactobacillus rhamnosus | a. A reduction in the index of all clinical scores (Psoriasis Area and Severity Index from 4.53 ± 4.457 to 3.57 ± 3.333, body surface area from 5.44 ± 6.451 to 4.94 ± 5.961 and Dermatology Life Quality Index from 8.83 ± 8.631 to 7 ± 7.814 in the probiotic group) |
| Buhaș MC et al.96 | Psoriasis | Probiotics (Bacillus indicus, Bacillus subtilis, Bacillus coagulans, Bacillus licheniformis and Bacillus clausii) and precision prebiotics (fructooligosaccharides, xylooligosaccharides and galactooligosaccharides) |
a. Probiotic and prebiotic supplementation significantly improved quality of life b. Reduced levels of tumour necrosis factor alpha, interleukin 6 and interferon gamma and enhanced levels of interleukin 10 c. Lower levels of total cholesterol, low-density lipoprotein cholesterol, triglyceride and uric acid d. Enhancement in the diversity of gut microbiota was noted |
| Di Marzio97 | Acne | Streptococcus thermophiles (topical) | a. Increase the production of ceramides when applied topically to the skin for 7 days |
| Jung GW et al.98 | Acne | Lactobacillus acidophilus, Lactobacillus delbrueckii bulgaricus Bifidobacterium bifidum | a. Significant improvement in total lesion count four weeks |
| Golkar et al.99 | Wound healing | Postbiotics obtained from Lactobacillus fermentum, Lactobacillus reuteri, Bacillus subtilis sp. natto |
a. All three formulations containing postbiotics significantly accelerated the wound healing process b. Bacillus subtilis sp. natto cold cream manifested a better wound healing property |
| Rong et al.100 | Pigmentation | Postbiotic supernatant of Lactobacillus helveticus (NS8-FS) NS8‐fermented milk |
a. NS8‐FS exhibited significant radical scavenging activity b. Inhibit melanin production c. Exhibit inhibitory effects both to the enzymatic activity of tyrosinase in hairless mice and tanning guinea pig models |
| Catic et al.101 | Wrinkles and ageing | CLS02021 is a postbiotic blend of metabolites, including organic acids, enzymes and peptides, which are a result of the co-fermentation of three proprietary probiotic strains; Lactobacillus plantarum, Lactobacillus casei and Streptococcus thermophilus was used | a. A significant difference of CLS02021 over the placebo group was observed for moisture and elasticity increase (both p < 0.001), pore size (p < 0.01) and a wrinkle depth decrease (p < 0.05) |
| Qi et al.102 | Rosacea | intervention effects of probiotics and postbiotics of L. salivarius 23-006 and L. paracasei 23-008 on rosacea by constructing an LL37-induced rosacea-like mouse model |
a. L. salivarius 23-006 and L. paracasei 23-008 alleviated skin lesions, reduced skin inflammatory infiltrates and decreased the expression of inflammatory factors b. Reduced the expression of cathelicidin LL37 and rosacea-associated factors by inhibiting the toll-like receptor 2/MyD88/NF-κB pathway c. Postbiotics of L. salivarius 23-006 and L. paracasei 23-008 could also ameliorate the rosacea-like phenotype in mice via the toll-like receptor 2/MyD88/NF-κB pathway, but less than probiotic therapy |
| Navarro-Belmonte et al.103 | Alopecia areata | Lactobacillus rhamnosus Bifidobacterium longum |
a. A higher proportion of Alopecia Areata patients treated with the probiotic formula showed improvement with respect to the reduction in the number of Alopecia Areata plaques (56% vs. 30%) and the affected scalp surface area (45% vs. 20%) b. Probiotic treatment led to a modification in the skin microbiota |
| Esmaeili et al.104 | Systemic lupus erythematosus | Lactobacillus delbrueckii, Lactobacillus rhamnosus |
a. ↓Inflammatory responses b. ↑Production of regulatory cells (dendritic cells) c. Live probiotics could modify properties of dendritic cells to modulatory cells, which might contribute to the induction of tolerance and renovation of immune haemostasis |
| Liu et al.105 | Atopic dermatitis | Faecal microbiota transplant once a week for three weeks |
a. Improvements in Eczema Area and Severity Index scores b. Decreased the helper T (Th2 and Th17) cell proportions and the levels of tumour necrosis factor alpha and total immunoglobulin E in serum |
SCORAD: SCORing Atopic Dermatitis
A Delphi Consensus Statement on the Role of Probiotics in the treatment of AD noted that probiotic supplementation for 8–12 weeks should be a part of the complementary therapy in the management of AD and associated flare-ups. Probiotic therapy may also serve as a strategy to reduce steroid usage or maintenance therapy in high-risk cases with frequent flares.108
There are few clinical trials of probiotic use in psoriasis. Probiotics and prebiotic supplementation in patients with psoriasis on therapy resulted in better psoriasis score, decreased inflammatory markers and skin thickness compared to those not receiving supplementation.96,109
Probiotic supplementation in acne is known to suppress inflammation through their antibacterial and anti-inflammatory effects. Probiotics may also lower the glycaemic load, reducing mammalian target of rapamycin and insulin-like growth factor-1 signaling, and therefore decreasing sebaceous gland hyperplasia and keratinocyte proliferation in acne.110,111
A Cochrane review on the use of probiotics in non-pregnant women with vulvovaginal candidiasis as an adjuvant therapy found an increased rate of short-term clinical and mycological cure and decreased relapse rate at one month, but this did not translate into a higher frequency of long-term clinical or mycological cure.112
Microbiome transplantation has been used for a variety of diseases like ulcerative colitis, Crohn’s disease, recalcitrant Clostridioides difficile infection, bacterial vaginosis, recurrent hepatic encephalopathy, milk allergy, autism spectrum disorder and intractable melanoma. While the concept of microbiome transplantation was earlier synonymous with faecal microbiota transplantation, it has now expanded to encompass the skin and vaginal microbiome for the treatment of disorders related to these body sites.113–119 Faecal microbiota transplant introduces a healthy donor’s gut microbiome to a patient with underlying disease.87 It can be delivered to the recipient by rectal enemas, endoscopy, tube insertion or oral capsules via colonoscopy or endoscopy. It is important in faecal microbiota transplant that the stool donor should have good general and bowel health without any detectable infectious agents.117 It has been tried in patients with atopic dermatitis, alopecia universalis, psoriasis and psoriatic arthritis with varied responses.120–122
Currently, the overall clinical efficacy due to bacteriotherapy appears to be variable and contradictory, with many studies showing limited evidence, while some systematic reviews and meta-analyses have shown strain and region-specific efficacy. These disparities may be due to the lower power of studies and strain variability between studies. In the future, a clearer curated database of microbial colonies in various diseases will enable appropriate donor-recipient combinations. Also, a deeper understanding of the mechanisms involved in the microbiome composition and activity post-therapy will enable the application of bacteriotherapy in an individualised manner.117
Conclusion
The gut microbiome and the human body are intricately woven and synergistically related as a part of the evolutionary process. The gut microbiome is very unique to an individual with many beneficial functions in a homoeostatic state. Dysbiosis of the microbiome appears to play a role in many diseases, including inflammatory diseases and autoimmune disorders. Recent studies have recognised the role of the gut microbiome in various dermatoses, revealing pathogenic patterns analogous to other inflammatory conditions and identifying novel mechanisms linking the gut to skin health. Understanding the interplay between the gut microbiome and diverse dermatological factors responsible for diseases, along with targeted modulation of the microbiome, presents a promising avenue for alleviating disease processes.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of AI-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
References
- Healthy microbiome – A mere idea or a sound concept? Physiol Res. 2022;71:719-38.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The role of gut microbiome in inflammatory skin disorders: A systematic review. Dermatol Reports. 2021;14:9188.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The gut microbiome in health and in disease. Curr Opin Gastroenterol. 2015;31:69-75.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Gut-skin axis: Current knowledge of the interrelationship between microbial dysbiosis and skin conditions. Microorganisms. 2021;9:353.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- An estimation of the number of cells in the human body. Ann Hum Biol. 2013;40:463-71.
- [CrossRef] [PubMed] [Google Scholar]
- The microbiome in health and disease from the perspective of modern medicine and ayurveda. Medicina (Kaunas). 2020;56:462.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Impact of gut microbiome on skin health: Gut-skin axis observed through the lenses of therapeutics and skin diseases. Gut Microbes. 2022;14:2096995.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Acne vulgaris, probiotics and the gut-brain-skin axis – Back to the future? Gut Pathog. 2011;3:1.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The human microbiome: Our second genome. Annu Rev Genomics Hum Genet. 2012;13:151-70.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Structure, function and diversity of the healthy human microbiome. Nature. 2012;486:207-14.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The normal bacterial flora of the human intestine and its regulation. J Clin Gastroenterol. 2007;41:S2-6.
- [CrossRef] [Google Scholar]
- Molecular-phylogenetic characterization of microbial community imbalances in human inflammatory bowel diseases. Proc Natl Acad Sci U S A. 2007;104:13780-5.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Role of the normal gut microbiota. World J Gastroenterol. 2015;21:8787-803.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- What is the healthy gut microbiota composition? A changing ecosystem across age, environment, diet, and diseases. Microorganisms. 2019;7:14.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The second brain: Is the gut microbiota a link between obesity and central nervous system disorders? Curr Obes Rep. 2016;5:51-64.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Antibiotic-associated diarrhoea. Best Pract Res Clin Gastroenterol. 2004;18:337-52.
- [CrossRef] [PubMed] [Google Scholar]
- Enterotypes of the human gut microbiome. Nature. 2011;473:174-80.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Human gut microbiota in health and disease: Unveiling the relationship. Front Microbiol. 2022;13:999001.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The skin microbiome: Current perspectives and future challenges. J Am Acad Dermatol. 2013;69:143-55.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Spatial organization of bacterial flora in normal and inflamed intestine: A fluorescence in situ hybridization study in mice. World J Gastroenterol. 2005;11:1131-40.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- factors influencing the gut microbiota, inflammation, and type 2 diabetes. J Nutr. 2017;147:1468S-75S.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Factors affecting the composition of the gut microbiota, and its modulation. PeerJ. 2019;7:e7502.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Part 1: The human gut microbiome in health and disease. Integr Med (Encinitas). 2014;13:17-22.
- [PubMed] [PubMed Central] [Google Scholar]
- Probiotics, prebiotics, and synbiotics: Gut and beyond. Gastroenterol Res Pract. 2012;2012:872716.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Role of the gut microbiota in nutrition and health. BMJ. 2018;361:k2179.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- In vitro colonic fermentation of dietary fibers: Fermentation rate, short-chain fatty acid production and changes in microbiota. Trends Food Sci Technol. 2019;88:1-9.
- [CrossRef] [Google Scholar]
- Gut microbiome and health: Mechanistic insights. Gut. 2022;71:1020-32.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Conversion of cholic acid and chenodeoxycholic acid into their 7-oxo derivatives by Bacteroides intestinalis AM-1 isolated from human feces. FEMS Microbiol Lett. 2009;293:263-70.
- [CrossRef] [PubMed] [Google Scholar]
- Influence of foods and nutrition on the gut microbiome and implications for intestinal health. Int J Mol Sci. 2022;23:9588.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Modulation of the gastrointestinal microbiome with nondigestible fermentable carbohydrates to improve human health. Microbiol Spectr. 2017;5
- [Google Scholar]
- Intestinal bacteria trigger T cell-independent immunoglobulin A(2) class switching by inducing epithelial-cell secretion of the cytokine APRIL. Immunity. 2007;26:812-26.
- [CrossRef] [PubMed] [Google Scholar]
- The gut microbiota of rural papua new guineans: Composition, diversity patterns, and ecological processes. Cell Rep. 2015;11:527-38.
- [CrossRef] [PubMed] [Google Scholar]
- Prakriti phenotypes as a stratifier of gut microbiome: A new frontier in personalized medicine? J Ayurveda Integr Med. 2020;11:360-5.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The gut-skin axis in health and disease: A paradigm with therapeutic implications. Bioessays. 2016;38:1167-76.
- [CrossRef] [PubMed] [Google Scholar]
- The skin microbiome and the gut-skin axis. Clin Dermatol. 2021;39:829-39.
- [CrossRef] [PubMed] [Google Scholar]
- Gut-skin axis: Unravelling the connection between the gut microbiome and psoriasis. Biomedicines. 2022;10:1037.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Interaction between microbiota and immunity in health and disease. Cell Res. 2020;30:492-506.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Rosacea, microbiome and probiotics: The gut-skin axis. Front Microbiol. 2024;14:1323644.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Microbiome of the skin and gut in Atopic Dermatitis (AD): Understanding the pathophysiology and finding novel management strategies. J Clin Med. 2019;8:444.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Development of the gut microbiota in infancy and its impact on health in later life. Allergol Int. 2017;66:515-22.
- [CrossRef] [PubMed] [Google Scholar]
- Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature. 2013;504:446-50.
- [CrossRef] [PubMed] [Google Scholar]
- Microbial perspective on the skin-gut axis and atopic dermatitis. Open Life Sci. 2024;19:20220782.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Gut microbiome and autoimmune disorders. Clin Exp Immunol. 2022;209:161-74.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Identifying species of symbiont bacteria from the human gut that, alone, can induce intestinal Th17 cells in mice. Proc Natl Acad Sci U S A. 2016;113:E8141-50.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- A Diet-sensitive commensal lactobacillus strain mediates TLR7-dependent systemic autoimmunity. Cell Host Microbe. 2019;25:113-127.e6.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Th17 responses and natural IgM antibodies are related to gut microbiota composition in systemic lupus erythematosus patients. Sci Rep. 2016;6:24072.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Gut microbiota, leaky gut, and autoimmune diseases. Front Immunol. 2022;13:946248.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The role of gut microbiota in cancer treatment: Friend or foe? Gut. 2020;69:1867-76.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Host and gut microbial tryptophan metabolism and type 2 diabetes: An integrative analysis of host genetics, diet, gut microbiome and circulating metabolites in cohort studies. Gut. 2022;71:1095-105.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Gut microbiota regulate motor deficits and neuroinflammation in a model of Parkinson’s disease. Cell. 2016;167:1469-80.e12.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Gut microbiome and serum metabolome alterations in obesity and after weight-loss intervention. Nat Med. 2017;23:859-68.
- [CrossRef] [PubMed] [Google Scholar]
- The impact of the gut microbiome on extra-intestinal autoimmune diseases. Nat Rev Immunol. 2023;23:9-23.
- [CrossRef] [PubMed] [Google Scholar]
- Gut microbiota, probiotics, and their interactions in prevention and treatment of atopic dermatitis: A review. Front Immunol. 2021;12:720393.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Aberrant composition of gut microbiota of allergic infants: A target of bifidobacterial therapy at weaning? Gut. 2002;51:51-5.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Gut microbiota composition and development of atopic manifestations in infancy: The KOALA birth cohort study. Gut. 2007;56:661-7.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Faecalibacterium prausnitzii subspecies-level dysbiosis in the human gut microbiome underlying atopic dermatitis. J Allergy Clin Immunol. 2016;137:852-60.
- [CrossRef] [PubMed] [Google Scholar]
- Microbiome in the gut-skin axis in atopic dermatitis. Allergy Asthma Immunol Res. 2018;10:354-62.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Gut microbiota of children with atopic dermatitis: Controlled study in the metropolitan region of São Paulo, Brazil. Allergol Immunopathol (Madr). 2020;48:107-15.
- [CrossRef] [PubMed] [Google Scholar]
- Clostridia in the gut and onset of atopic dermatitis via eosinophilic inflammation. Ann Allergy Asthma Immunol. 2016;117:91-2.e1.
- [CrossRef] [PubMed] [Google Scholar]
- Microbiome in the gut-skin axis in atopic dermatitis. Allergy Asthma Immunol Res. 2018;10:354-62.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- A population-based study on associations of stool microbiota with atopic diseases in school-age children. J Allergy Clin Immunol. 2021;148:612-20.
- [CrossRef] [PubMed] [Google Scholar]
- Skin and gut microbiome in psoriasis: Gaining insight into the pathophysiology of it and finding novel therapeutic strategies. Front Microbiol. 2020;11:589726.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Decreased bacterial diversity characterizes the altered gut microbiota in patients with psoriatic arthritis, resembling dysbiosis in inflammatory bowel disease. Arthritis Rheumatol. 2015;67:128-39.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Metabolic syndrome and psoriasis: Mechanisms and future directions. Front Immunol. 2021;12:711060.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Intestinal fatty acid binding protein, a biomarker of intestinal barrier, is associated with severity of psoriasis. J Clin Med. 2019;8:1021.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Assessing causal relationships between gut microbiota and psoriasis: Evidence from two sample Mendelian randomization analysis. Sci Rep. 2024;14:8831.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Skin expression of mammalian target of rapamycin and forkhead box transcription factor O1, and serum insulin-like growth factor-1 in patients with acne vulgaris and their relationship with diet. Br J Dermatol. 2016;174:1299-307.
- [CrossRef] [PubMed] [Google Scholar]
- Acne, microbiome, and probiotics: The gut-skin axis. Microorganisms. 2022;10:1303.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Mechanistic target of rapamycin (mTOR) expression is increased in acne patients’ skin. Exp Dermatol. 2016;25:153-5.
- [CrossRef] [PubMed] [Google Scholar]
- Potential role of the microbiome in acne: A comprehensive review. J Clin Med. 2019;8:987.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Patients with acne vulgaris have a distinct gut microbiota in comparison with healthy controls. Acta Derm Venereol. 2018;98:783-90.
- [CrossRef] [PubMed] [Google Scholar]
- The increasing importance of the gut microbiome in acne vulgaris. Folia Microbiol (Praha). 2022;67:825-35.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The role of small intestinal bacterial overgrowth in rosacea: A 3-year follow-up. J Am Acad Dermatol. 2016;75:e113-5.
- [CrossRef] [PubMed] [Google Scholar]
- Microbiome in hidradenitis suppurativa – What we know and where we are heading. Int J Mol Sci. 2022;23:11280.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Causal relationship between gut microbiota and hidradenitis suppurativa: A two-sample Mendelian randomization study. Front Microbiol. 2024;15:1302822.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Comparative diet-gut microbiome analysis in Crohn’s disease and Hidradenitis suppurativa. Front Microbiol. 2023;14:1289374.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The role of the microbiota in connective tissue diseases. Dermatology Review/Przegląd Dermatologiczny. 2022;109:417-26.
- [PubMed] [Google Scholar]
- Microbiome changes in connective tissue diseases and vasculitis: Focus on metabolism and inflammation. Int J Mol Sci. 2022;23:6532.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The Microbiome in connective tissue diseases and vasculitides: An updated narrative review. J Immunol Res. 2017;2017:6836498.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Analysis of matched skin and gut microbiome of patients with vitiligo reveals deep skin dysbiosis: Link with mitochondrial and immune changes. J Invest Dermatol. 2021;141:2280-90.
- [CrossRef] [PubMed] [Google Scholar]
- Metagenomic sequencing reveals altered gut microbial compositions and gene functions in patients with non-segmental vitiligo. BMC Microbiol. 2023;23:265.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Fecal microbiota transplantation: A potential tool for treatment of human female reproductive tract diseases. Front Immunol. 2019;10:2653.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Female gut and genital tract microbiota-induced crosstalk and differential effects of short-chain fatty acids on immune sequelae. Front Immunol. 2020;11:2184.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Gut-skin-axis modulation via fecal microbiome transplant. An ecological approach for atopic dermatitis treatment. J Pak Assoc Dermatol.. 202;33:1660-8.
- [Google Scholar]
- Effect of diet on the gut microbiota: Rethinking intervention duration. Nutrients. 2019;11:2862.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Dietary fiber and prebiotics and the gastrointestinal microbiota. Gut Microbes. 2017;8:172-84.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Probiotics and prebiotics in atopic dermatitis: Pros and cons (Review) Exp Ther Med. 2021;22:1376.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The International Scientific Association of Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of postbiotics. Nat Rev Gastroenterol Hepatol. 2021;18:649-67.
- [CrossRef] [PubMed] [Google Scholar]
- Current postbiotics in the cosmetic market-an update and development opportunities. Appl Microbiol Biotechnol. 2022;106:5879-91.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Probiotic supplement reduces atopic dermatitis in preschool children: A randomized, double-blind, placebo-controlled, clinical trial. Am J Clin Dermatol. 2010;11:351-61.
- [PubMed] [Google Scholar]
- Effect of oral administration of a mixture of probiotic strains on SCORAD index and use of topical steroids in young patients with moderate atopic dermatitis: A randomized clinical trial. JAMA Dermatol. 2018;154:37-43.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The role of probiotics as an adjunctive therapy in psoriasis. J Psoriasis Psoriatic Arthritis. 2023;8:49-55.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Transforming psoriasis care: Probiotics and prebiotics as novel therapeutic approaches. Int J Mol Sci. 2023;24:11225.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Effect of the lactic acid bacterium Streptococcus thermophilus on ceramide levels in human keratinocytes in vitro and stratum corneum in vivo. J Invest Dermatol. 1999;113:98-106.
- [CrossRef] [PubMed] [Google Scholar]
- Prospective, randomized, open-label trial comparing the safety, efficacy, and tolerability of an acne treatment regimen with and without a probiotic supplement and minocycline in subjects with mild to moderate acne. J Cutan Med Surg. 2013;17:114-22.
- [CrossRef] [PubMed] [Google Scholar]
- A novel effective formulation of bioactive compounds for wound healing: preparation, in vivo characterization, and comparison of various postbiotics cold creams in a rat model. Evid Based Complement Alternat Med. 2021;2021:8577116.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Skin resistance to UVB-induced oxidative stress and hyperpigmentation by the topical use of Lactobacillus helveticus NS8-fermented milk supernatant. J Appl Microbiol. 2017;123:511-23.
- [CrossRef] [PubMed] [Google Scholar]
- The moisturizing efficacy of a proprietary dermo-cosmetic product(CLS02021) versus placebo in a 4-week application period. Med Arch. 2022;76:108-114.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Probiotics suppress LL37 generated rosacea-like skin inflammation by modulating the TLR2/MyD88/NF-κB signaling pathway. Food Funct. 2024;15:8916-34.
- [CrossRef] [PubMed] [Google Scholar]
- The effect of an oral probiotic mixture on clinical evolution and the gut and skin microbiome in patients with alopecia areata: A randomized clinical trial. Cosmetics. 2024;11:119.
- [Google Scholar]
- Inhibitory effects of tolerogenic probiotics on migratory potential of lupus patient-derived DCs. Iran J Basic Med Sci. 2021;24:1509-14.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Fecal microbiota transplantation against moderate-to-severe atopic dermatitis: A randomized, double-blind controlled explorer trial. Allergy. 2024;2024
- [Google Scholar]
- Probiotics in the therapeutic arsenal of dermatologists. Microorganisms. 2021;9:1513.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The effect of multistrain synbiotic and vitamin D3 supplements on the severity of atopic dermatitis among infants under 1 year of age: A double-blind, randomized clinical trial study. J Dermatolog Treat. 2022;33:812-7.
- [CrossRef] [PubMed] [Google Scholar]
- Delphi consensus statement on the role of probiotics in the treatment of atopic dermatitis. Cureus. 2024;16:e64583.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The effectiveness and safety of probiotic supplements for psoriasis: A systematic review and meta-analysis of randomized controlled trials and preclinical trials. J Immunol Res. 2021;2021:7552546.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The effect of probiotics on immune regulation, acne, and photoaging. Int J Womens Dermatol. 2015;1:85-9.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Supplementation with Lactobacillus rhamnosus SP1 normalises skin expression of genes implicated in insulin signalling and improves adult acne. Benef Microbes. 2016;7:625-30.
- [CrossRef] [PubMed] [Google Scholar]
- Probiotics for vulvovaginal candidiasis in non-pregnant women. Cochrane Database Syst Rev. 2017;11:CD010496.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Randomised clinical trial: Faecal microbiota transplantation by colonoscopy vs. vancomycin for the treatment of recurrent Clostridium difficile infection. Aliment Pharmacol Ther. 2015;41:835-43.
- [CrossRef] [PubMed] [Google Scholar]
- The role of faecal microbiota transplantation: Looking beyond Clostridioides difficile infection. Ther Adv Infect Dis. 2021;8:2049936120981526.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Fecal microbiota transplantation to maintain remission in Crohn’s disease: A pilot randomized controlled study. Microbiome. 2020;8:12.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Vaginal microbiome transplantation in women with intractable bacterial vaginosis. Nat Med. 2019;25:1500-4.
- [CrossRef] [PubMed] [Google Scholar]
- The emerging potential of microbiome transplantation on human health interventions. Comput Struct Biotechnol J. 2022;20:615-27.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Fecal microbiota transplant promotes response in immunotherapy-refractory melanoma patients. Science. 2021;371:602-9.
- [CrossRef] [PubMed] [Google Scholar]
- Fecal microbiota transplantation in hepatic encephalopathy: A systematic review. Scand J Gastroenterol. 2021;56:560-9.
- [CrossRef] [PubMed] [Google Scholar]
- Fecal microbiota transplant and dermatologic disorders: A retrospective cohort study assessing the gut microbiome’s role in skin disease. World J Dermatol. 2021;9:1-10.
- [Google Scholar]
- Safety and efficacy of faecal microbiota transplantation for active peripheral psoriatic arthritis: An exploratory randomised placebo-controlled trial. Ann Rheum Dis. 2021;80:1158-67.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical efficacy of fecal microbial transplantation treatment in adults with moderate-to-severe atopic dermatitis. Immun Inflamm Dis. 2022;10:e570.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]







