Translate this page into:
HIV and tuberculosis: Partners in crime
2 Foundation for Medical Research, Mumbai, India
3 Department of Medicine, Imperial College London, Chelsea and Westminster Hospital London, United Kingdom
4 Department of HIV Medicine, Apollo Hospital, Ahmedabad, India
5 Dr. Maniar's ID clinic, Mumbai, India
Correspondence Address:
Janak K Maniar
69/2, Walkeshwar Road, Mumbai - 400 006
India
| How to cite this article: Maniar JK, Kamath RR, Mandalia S, Shah K, Maniar A. HIV and tuberculosis: Partners in crime. Indian J Dermatol Venereol Leprol 2006;72:276-282 |
Abstract
Background: Tuberculosis is the commonest infection detected in HIV-infected individuals worldwide. Aim: The aim of this study is to describe the clinical, bacteriologic and radiological spectrum of tuberculosis (TB) in the setting of human immunodeficiency virus (HIV) infection in a tertiary care centre in Mumbai. Methods: A total of 8640 HIV-infected individuals were screened for tuberculosis routinely from January 1998 to December 2003, using clinical examination, chest X-ray and abdominal ultrasonography, sputum smears for acid-fast bacilli (AFB) and culture on Lowenstein-Jensen medium. Results: TB was detected in 8078 (93.5%) patients of whom 3393 (42%) had pulmonary, 3514 (43.5%) had extrapulmonary TB and 1171 (14.5%) had disseminated disease. One thousand two hundred thirty eight patients (36.5%) showed AFB in sputum, while 1154 (34%) showed growth on culture medium and 4174 had radiographic involvement. In 781 (67%) individuals disseminated disease coexisted with pulmonary involvement. All 8078 coinfected patients were treated with anti-TB therapy (ATT), of whom 6422 patients (79.5%) showed one or more adverse events. Gastritis was the commonest complaint followed by hepatitis and skin rashes. ATT resistance was detected in 482 individuals. Conclusion: Tuberculosis is the commonest opportunistic infection (OI) in HIV positive patients in India, showing a higher prevalence of extrapulmonary and disseminated TB and adverse events due to ATT. Early recognition of concurrent OIs and their adequate treatment and prophylaxis is essential.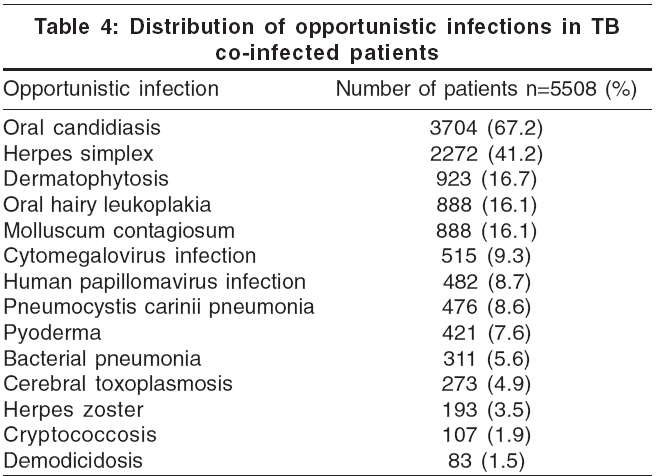





 |
 |
 |
 |
 |
 |
Introduction
Around one out of three people worldwide (i.e., approximately 1.9 billion) are infected with Mycobacterium tuberculosis.[1] Approximately 8 million new cases occur annually and two-thirds of all tuberculosis (TB) cases reported globally are from the Asia-Pacific region.[1] Fourteen million TB cases in India are estimated, of which 3.5 million are sputum positive. TB kills more than 2 million people annually and is the leading cause of death among adults (15-59 years) in Africa and Asia.[1] WHO estimates are that there are 42 million HIV-infected people worldwide, with an estimated 5 million in India.[1],[2] Mycobacterium tuberculosis is the commonest infecting organism in HIV-infected patients worldwide and one in three people with AIDS will die of TB.[2] The aim of this retrospective study was to examine the clinical, bacteriologic and radiological spectrum of tuberculosis in the setting of HIV infection in India.
Methods
Data were recorded routinely on a study-specific database in a computerized format since 2003 from the case-files maintained by the department. All adults who tested HIV-positive between 1st January 1998 and 31st December 2003 were routinely investigated for TB. The study protocol envisaged a tuberculin test, examination of the sputum for acid-fast bacilli (AFB) by the Ziehl-Neelsen method (especially in those with productive cough), chest X-ray and abdominal ultrasound (USG) in all patients. Lymph node biopsy and study of pleural, peritoneal and cerebrospinal fluid were performed when indicated. The same samples were used for inoculation on the culture medium. Tests for hepatitis B and hepatitis C infections were performed at the beginning of the study in all patients.
Patients were classified as confirmed TB or presumptive TB. Confirmed TB was defined as a clinical illness consistent with TB accompanied by either a sputum smear positive for AFB, growth on culture or typical histopathological findings in a surgical specimen. Presumptive TB was defined as a clinical illness with USG of the abdomen showing enlarged lymph nodes, splenic infiltration, chest X-ray showing features suggestive of pulmonary involvement or as matted peripheral lymphadenopathy.
After the diagnosis was confirmed, patients were given first-line anti-tuberculosis treatment (ATT) utilizing standard dosages, viz. isoniazid (H), rifampicin (R), ethambutol (E) and pyrazinamide (Z), for a minimum of 9 months for pulmonary TB. This was given as 4 drugs (HREZ) for 3 months, 3 drugs (HRE) for the following 3 months and 2 drugs (HR) for the final 3 months. ATT was continued for 2 years in cases where spinal, bone or CNS TB was detected and response to treatment was recorded at follow-up.
Patients were regularly followed up throughout the duration of their individual treatment periods. Routine blood tests were performed to monitor hemoglobin, total counts, erythrocyte sedimentation rate, liver function and renal function. Fundus examination was performed to rule out ethambutol induced optic neuritis. A chest X-ray was performed every three months to judge the response to ATT and to detect new co-infections. Drug resistance studies were performed in patients who showed suboptimal clinical and investigational response such as sputum smear positivity or non-resolution of radiological opacities at 3 months. These patients were prescribed second line drugs, which were selected from kanamycin/amikacin, ciprofloxacin/other fluoroquinolones, cycloserine, para-aminosalicylic acid (PAS) and ethionamide, in addition to ethambutol and pyrazinamide.
All coinfected patients were identified retrospectively. After informed consent for inclusion into the study; demographic characteristics and clinical features of these patients such as type of infection, whether pulmonary or extrapulmonary; organ-systems involved; radiographic findings; response to anti-tuberculous drugs, were recorded at onset and on subsequent follow-up visits.
All information collected on these patients was extracted as ASCII text files and transferred to SAS statistical package for data analysis. All results are presented as numbers with percentages and denominators are quoted where deemed necessary.
Results
Eight thousand six hundred and forty HIV-positive individuals were followed up for 6 years. Of these, co-infection with TB was detected in 8078 (93.5%). At presentation the mean age was 35 years and the mean weight was 55 kg. The male: female ratio was 3:1 and 98% had a sexual transmission of HIV. Pulmonary disease was detected in 3393 (42%) patients, while 3514 (43.5%) had extrapulmonary disease. Disseminated disease was diagnosed in 1171 (14.5%) patients, with 781 (66.7%) of them showing some pulmonary involvement, either microbiologically or radiologically [Figure - 1].
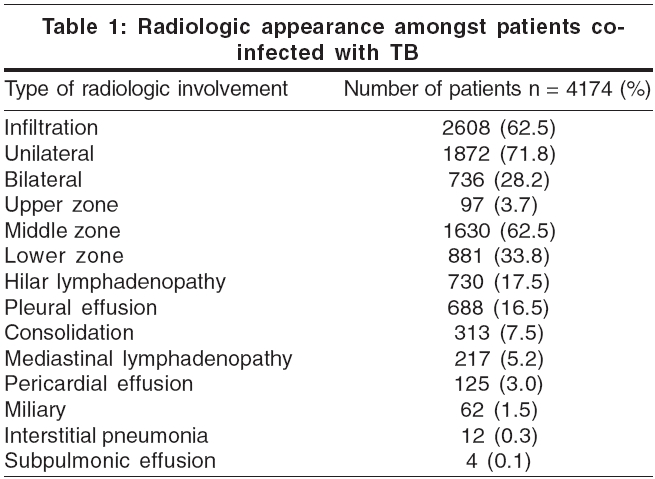
Radiologic features suggestive of tuberculosis were seen in 4174 patients. More than one type of radiological involvement was noted on a chest X-ray in 1878 (44.99%) of these patients. The commonest radiologic feature was pulmonary infiltration, seen in 2608 patients (62.5%) and visualized as opacities in the lung fields. Unilateral involvement was more common than bilateral involvement (71.8% vs 28.2%) and the majority of patients had middle zone infiltration followed by involvement of the lower zone [Table - 1].
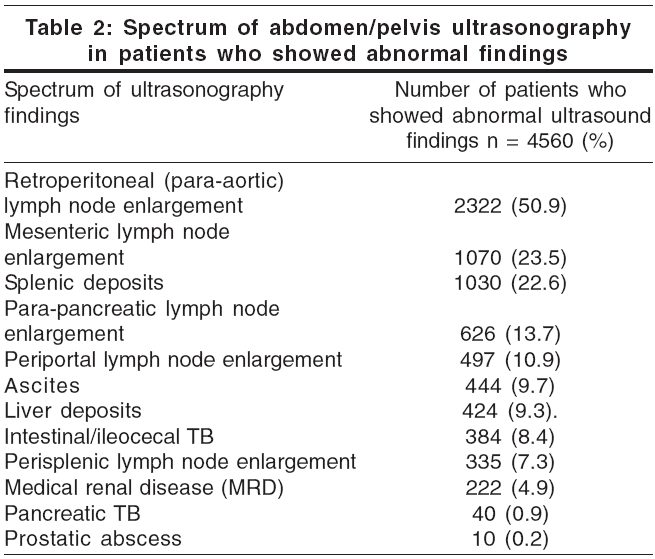
An ultrasound examination of the abdomen and pelvis was done in all 8078 patients; 4560 (56.4%) showed abnormal findings. Lymph node enlargement was the commonest finding, followed by tuberculous deposits in various organs [Table - 2].
Involvement of almost every organ system was noted in patients with extrapulmonary (n=3514) and disseminated TB (n=1171) . Neurological involvement was seen as meningitis and meningo-encephalitis in 497 (10.6%) patients; other neurological infections detected were tuberculoma (8.9%), hydrocephalus (8.5%), spinal myelitis (1.3%), Pott′s disease of the spine (0.6%) , psoas abscess (0.3%) and Froin′s syndrome (0.2%) [Figure - 1]. Pott′s disease of the spine and psoas abscess are included in neurological involvement as the patients presented with neuromuscular symptoms such as radicular pain and weakness. Other presentations seen were skin tuberculosis (3.0%), tuberculous osteomyelitis (1.3%) and tubercles in the eyes (0.2%).
Due to practical difficulties, the tuberculin test was performed only in 7905 (97.9%) patients; and was found to be reactive in 3090 (39.09%) of them [Table - 3].
Of 3393 patients with pulmonary and 781 patients with disseminated TB, sputum examination showed the presence of acid-fast bacilli in 1523 (36.5%). Of these, 110 (7.2%) patients had no radiologic abnormalities but only constitutional symptoms and signs. In the 781 patients with disseminated disease, 285 (36.5%) had a positive sputum smear, indicating pulmonary involvement [Figure - 1].
After 6 weeks on Lowenstein-Jensen medium, 1154/3393 (34%) sputum samples from patients with pulmonary disease grew Mycobacterium spp . on culture. Thus, in 4174 patients with chest radiograph abnormalities, 1419 sputum samples grew AFB on culture (34%). 1191 (84%) samples of positive sputum cultures were identified as Mycobacterium tuberculosis complex while 228 (16%) grew mycobacteria other than Mycobacterium tuberculosis (MOTT).
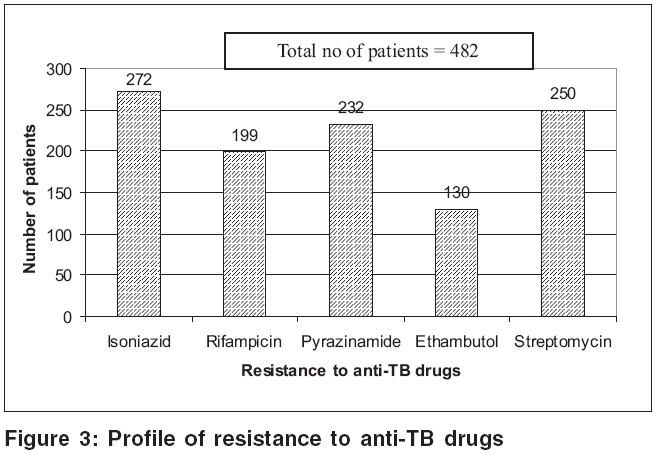
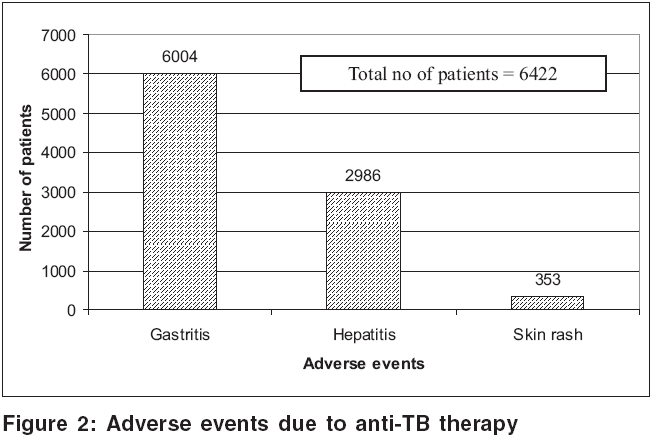
Adverse drug reactions to ATT were reported in 6422 (79.5%) patients. Gastritis was the commonest adverse reaction (93.5%), followed by hepatitis (46.5%); and skin rashes (5.5%) [Figure - 2]. Drug resistance to anti-TB drugs was detected in 482 patients: isoniazid (56.4%), rifampicin (41.3%), pyrazinamide (48.1%), ethambutol (27%) and streptomycin (51.9%) [Figure - 3]. Other opportunistic infections (OIs) were noted in 5508 (68.2%) patients. Oral candidiasis, herpetic reactivations and oral hairy leukoplakia predominated [Table - 4]. Hepatitis testing was routinely performed in all HIV positive patients who tested positive to TB. Co-infection with hepatitis B virus (HBV) was detected in 666 (8.2%) patients, while 101 (1.3%) showed evidence of hepatitis C virus (HCV) infection. Four (0.05%) patients suffered from dual (HBV + HCV) infection.
The majority of patients completed the prescribed course of anti-tuberculous therapy. Of the 8078 patients in whom TB was detected and ATT prescribed, 273 stopped treatment on their own and were lost to follow-up. The compliance to treatment was 96.6%.
Discussion
Patients with HIV infection exhibit a unique susceptibility to Mycobacterium tuberculosis . The rate of progression of TB infection to active disease following initial exposure is greater than 40% in people with HIV, compared to approximately 5% in HIV-negative individuals.[1] They are more likely to develop active rather than latent disease and are also susceptible to reactivation of latent tuberculosis infection with an annual risk of 5-10%.[1] When infection becomes established, either endogenously reactivated or exogenously, there is usually rapid progression to active disease.[3] Tuberculin-positive coinfected persons have a risk of TB development of 8 to 10% per-year compared to < 0.1% annually in those without HIV infection.[4] Thus, once infection is established, progression to clinical disease is faster and more severe in HIV-positive patients.
A prospective cohort study undertaken in the United States followed 1130 HIV positive patients for a median of 53 months.[5] Active TB was diagnosed in 31 patients from this group and occurred more frequently in persons with CD4 cell counts of less than 200/µL. Our study showed that 93.5% of HIV-positive patients followed up for 6 years developed coinfection with tuberculosis. Unfortunately, immunological and virologic markers were not assessed due to financial constraints.
Clinical features Almost all patients in our study sample had systemic manifestations of tuberculosis (fever, anorexia, weight loss, night sweats and cough), regardless of whether they had pulmonary or extrapulmonary disease. While 3393 patients presented with symptoms suggestive of pulmonary disease, 43% had extrapulmonary and 14.5% had disseminated disease. Abdominal lymphadenopathy was the commonest extrapulmonary manifestation, followed by neurological involvement. Other sites involved were the skin, bone and the eye. An earlier study showed 30% of TB in HIV-infected patients was extrapulmonary and another 32% of patients had both pulmonary and extrapulmonary TB.[6]
In extrapulmonary disease, the commonly involved sites are the lymph nodes, pleura, pericardium, spleen, liver, kidneys, prostate, intestine, meninges, brain parenchyma, joints and bone marrow. Visceral lesions and intra-abdominal lymphadenopathy with necrosis characterize abdominal tuberculosis in HIV-infected patients.[7] In our study, the most commonly involved lymph nodes were the paraaortic and mesenteric groups.
Central nervous system involvement is also more common in HIV-positive patients.[8] The clinical presentation of meningitis is similar in both groups, although mass lesions are more common in HIV-infected patients.[9] We diagnosed meningitis and encephalitis in 497 (10.6%) patients and tuberculoma in 416 (8.9%).
Diagnosis The fundamental laboratory techniques for detecting TB infection are the acid-fast smear and cultivation of the organism.[10] Pulmonary infiltrates, cavities, etc. are visualized well on chest X-ray, while hilar adenopathy is better visualized on computed tomography (CT).[11],[12] Fine needle aspiration cytology and excisional biopsy that showed the presence of tuberculoid granulomas with epithelioid cells and giant cells facilitated our diagnosis of lymph node TB. We found lumbar puncture and pleuro-, peritoneo- and pericardiocentesis crucial for the diagnosis of meningitis and effusions. The aspirated fluid was analyzed for the cell type and biochemistry. Adenosine deaminase (ADA) levels were done in all cases.
About 5% HIV-infected patients with pulmonary tuberculosis have positive results on acid-fast staining of sputum; despite normal chest radiographs.[11] In our study, 3.2% of patients with pulmonary TB had positive sputum smears and no radiological features consistent with TB. The cultivation of mycobacteria takes 4-6 weeks on Lowenstein-Jensen medium, with the sensitivity varying according to the nature of the specimen and ranging from 26 to 42%.[12] In our sample the sensitivity of this test was 34%.
On radiology, the classical features of apical distribution, cavitation, pulmonary fibrosis, shrinkage and calcification are seen in those with well-preserved immune function. In advanced disease, atypical pictures such as lower lobe infiltration, mediastinal lymphadenopathy, miliary mottling and even a normal radiograph may be seen.[13] Infiltration was the commonest and was seen in 62.5% patients; lower lobe involvement was noted in 38%. Other diagnostic modalities include fluorescent microscopy of a 24-hour sputum sample using auramine-rhodamine, use of rapid culture methods such as BACTEC and polymerase chain reaction (PCR) for M. tuberculosis .
Regardless of age, induration of 5 mm or more on tuberculin test is considered as positive in HIV-infected patients.[14] In HIV and TB co-infection, there is a reduction in the proportion of those reacting to tuberculin as the CD4 count falls.[14] Anergy (tuberculin test 0 mm) is a fair indication of significant immunosuppression, i.e., absolute CD4 count < 200/cmm. In our study, this test was performed in 7905 patients and was found to be reactive in 3090 patients, of whom 73% showed a reaction between 10 and 20 mm. Any reaction above 10 mm should be considered a marker of latent TB infection and ATT should be considered for these patients.
Treatment of tuberculosis in HIV-infected patients Treatment guidelines for HIV-infected patients are the same as those for non-infected individuals.[12] If directly observed treatment (DOT) is not possible, self-administered treatment with a fixed dose drug combination is preferred to improve the compliance.
Treatment was given for a minimum of 9 months to our patients. ATT was continued for 2 years in cases of spinal, bone or CNS TB due to occurrence of higher relapse rates (>50%) in TB co-infected patients who were given the standard 6 month regimen (personal observation of the first author). The extended duration of treatment showed no unexpected increase in the number of adverse events.
The response to anti-tuberculosis therapy was found to be the same in HIV-infected patients as in HIV negatives. The median survival in HIV-infected population is about 18 months after TB diagnosis, in the absence of ATT or ART.[15],[16] In Abidjan, C τte d′Ivoire, where the rate of M. tuberculosis HIV co-infection is 20 to 70%, the mortality rate was 20% when there was co-infection versus 2.2% for patients with TB alone.[16] The increased mortality among HIV-infected persons in these studies has been attributed largely to other concurrent opportunistic infections rather than to a recurrence of TB. It is thought that the tuberculous coinfection also impairs the immune response to the opportunistic pathogen leading to severe disease. Hence, it is imperative that other opportunistic infections are treated as early as possible. In our study, 5508 patients were diagnosed with OI, with candidiasis, herpes and OHL being the most frequent [Table - 4].
A study by Wiktor and colleagues showed that prescription of trimethoprim-sulfamethoxazole (TMP-SMX) to HIV/TB patients upon diagnosis of active TB protected them from a variety of infectious causes of death.[16] We prescribed co-trimoxazole prophylactically to those patients whose tuberculin test was negative or showed anergy and who had no known sensitivity to the drug.
Adverse reactions Adverse drug reactions are more common in HIV-infected individuals than in their normal counterparts. A high frequency of drug reactions (>20%) has been noted in HIV-infected patients on ATT.[14] In our study, 79.5% patients showed adverse events, most commonly gastritis (93.5%), followed by hepatitis (46.5%) and skin rashes (5.5%). Co-infection with HBV and HCV may play a role in the genesis of drug-induced hepatitis.
Role of TB prophylaxis In India, the issue of TB chemoprophylaxis is complex due to difficulty in excluding active disease in those with HIV/TB co-infection; chemoprophylaxis may not prevent reinfection in endemic areas; INH use may inadvertently foster resistance, while the tuberculin test is unreliable. We offered no chemoprophylaxis as our observations have shown a high failure rate.
In conclusion, tuberculosis is the commonest opportunistic infection in HIV positive patients in India. HIV-related TB shows a higher prevalence of extrapulmonary and disseminated TB and adverse events due to ATT. Hepatitis B or hepatitis C co-infection may contribute to the higher incidence of ATT-induced hepatitis. Early recognition of concurrent OIs, their adequate treatment and chemoprophylaxis is essential. The increased risk of TB in HIV-positive patients and the complex clinical presentation means that HIV positive patients should be regularly screened for tuberculosis.
| 1. |
Narain JP, Lo YR. Epidemiology of HIV-TB in Asia. Indian J Med Res 2004;120:277-89.
[Google Scholar]
|
| 2. |
UNAIDS. AIDS Epidemic Update. Geneva: WHO; 1998.
[Google Scholar]
|
| 3. |
Goletti D, Weissman D, Jackson RW, Graham NM, Vlahov D, Klein RS, et al . Effect of Mycobacterium tuberculosis on HIV replication: Role of immune activation. J Immunol 1996;157:1271-8.
[Google Scholar]
|
| 4. |
World Health Organization. Guidelines for implementing collaborative TB and HIV programmes, WHO report. Geneva: WHO; 2003.
[Google Scholar]
|
| 5. |
Markowitz N, Hansen NI, Hopewell PC, Glassroth J, Kvale PA, Mangura BT, et al . Incidence of tuberculosis in the United States among HIV-infected persons. Ann Intern Med 1997;126:123-32.
[Google Scholar]
|
| 6. |
Small PM, Schecter GF, Goodman PC, Sande MA, Chaisson RE, Hopewell PC. Treatment of tuberculosis patients with advanced human immunodeficiency virus. N Engl J Med 1991;324:289-94.
[Google Scholar]
|
| 7. |
Fee MJ, Oo MM, Gabayan AE, Radin DR, Barnes PF. Abdominal tuberculosis in patients with the human immunodeficiency virus. Clin Infect Dis 1995;20:938-44.
[Google Scholar]
|
| 8. |
Berenguer J, Moreno S, Laguna F, Vicente T, Adrados M, Ortega A, et al . Tuberculous meningitis in patients infected with the human immunodeficiency virus. N Engl J Med 1992;326:668-72.
[Google Scholar]
|
| 9. |
Dube M-P, Holtom PD, Larsen RA. Tuberculous meningitis in patients with and without human immunodeficiency virus infection. Am J Med 1992;93:520-4.
[Google Scholar]
|
| 10. |
Chaisson RE, Bishai WR. MAC and Tuberculosis Infection: Management in HIV Disease. CME at http://www.medscape.com/viewprogram/664 - last updated July 14, 1999; last accessed on March 25, 2006.
[Google Scholar]
|
| 11. |
Perlman DC, el-Sadr WM, Nelson ET, Matts JP, Telzak EE, Salomon N, et al . Variation of chest radiographic patterns in pulmonary tuberculosis by degree of human immunodeficiency virus-related immunosuppression: The Terry Beirn Community Programs for Clinical Research on AIDS (CPCRA). Clin Infect Dis 1997;25:242-6.
[Google Scholar]
|
| 12. |
Mc Shane H. Co-infection with HIV and TB: Double trouble. Int J STD AIDS 2005;16:91-7.
[Google Scholar]
|
| 13. |
Jones BE, Ryu R, Yang Z, Cave MD, Pogoda JM, Otaya M, et al . Chest radiographic findings in patients with recent or remote infection. Am J Respir Crit Care Med 1997;156:1270-3.
[Google Scholar]
|
| 14. |
Gordin FM. Mycobacterium tuberculosis infection. Dolin R, Masur H, Saag MS, editors. AIDS Therapy. 2nd ed. Philadelphia: Elsevier Science Health Division; 2003. p. 459-74.
[Google Scholar]
|
| 15. |
Greenberg AE, Lucas S, Tossou O, Coulibaly IM, Coulibaly D, Kassim S, et al . Autopsy-proven causes of death in HIV-infected patients treated for tuberculosis in Abidjan, C τte d'Ivoire. AIDS 1995;9:1251-4.
[Google Scholar]
|
| 16. |
Wiktor SZ, Sassan-Morroko M, Grant AD, Abouya L, Karon JM, Maurice C, et al . Efficacy of trimethoprim-sulphamethoxazole prophylaxis to decrease morbidity and mortality in HIV-1-infected patients with tuberculosis in Abidjan, Cote d'Ivoire: A randomized controlled trial. Lancet 1999;353:1469-75.
[Google Scholar]
|
Fulltext Views
1,899
PDF downloads
1,082





