Translate this page into:
IgG/IgA pemphigus reactive with desmoglein 1 with additional undetermined reactivity with epidermal basement membrane zone
2 Department of Histopathology, Postgraduate Institute of Medical Education and Research, Chandigarh, India
3 Kurume University School of Medicine, and Kurume University Institute of Cutaneous Cell Biology, Fukuoka, Japan
4 Kurume University School of Medicine, and Kurume University Institute of Cutaneous Cell Biology, Fukuoka; Osaka City University Graduate School of Medicine, Osaka, Japan
Correspondence Address:
Amrinder J Kanwar
Department of Dermatology, Venereology and Leprology, Postgraduate Institute of Medical Education and Research, Sector 12, Chandigarh - 160 012
India
| How to cite this article: Kanwar AJ, Vinay K, Saikia UN, Koga H, Teye K, Tsuruta D, Hashimoto T. IgG/IgA pemphigus reactive with desmoglein 1 with additional undetermined reactivity with epidermal basement membrane zone. Indian J Dermatol Venereol Leprol 2014;80:46-50 |
Abstract
IgG/IgA pemphigus is an extremely rare subset of pemphigus, showing anti-keratinocyte cell surface antibodies of both IgG and IgA classes. Herein, we describe a unique case of IgG/IgA pemphigus with clinical features of edematous erythema and peripheral vesiculopustules. Histopathology showed the presence of subcorneal pustules and acantholytic blisters in the mid-epidermis with neutrophilic infiltration and eosinophilic spongiosis. Direct immunofluorescence of perilesional skin showed both IgG and IgA deposits to keratinocyte cell surfaces and unusual granular deposits of IgG, IgM, and C3 along basement membrane zone. On enzyme linked immunosorbent assay , the auto-antibodies were found to be reactive to desmoglein 1 antigen. Various clinical, histopathological, and immunological findings in our case overlapped with the features of IgA pemphigus, pemphigus herpetiformis, and pemphigus foliaceus. These findings indicate that IgG/IgA pemphigus may be a transitional form between IgA pemphigus and pemphigus herpetiformis, and thus provides insight into the pathogenicity of this rare disorder.Introduction
Pemphigus is an autoimmune mucocutaneous blistering disorder, characterized by antibodies to keratinocyte cell surface proteins, mainly desmogleins (Dsg). There are two major classical types: pemphigus vulgaris and pemphigus foliaceus. There are also several atypical variants, including pemphigus herpetiformis, paraneoplastic pemphigus, and IgA pemphigus. IgA pemphigus is divided into subcorneal pustular dermatosis type and intra-epidermal neutrophilic IgA dermatosis type. Desmocollin 1 (Dsc1) is the target antigen in sub-corneal pustular dermatosis type of IgA pemphigus, [1] whereas no specific antigen has been identified in most cases of intra-epidermal neutrophilic type.
IgG/IgA pemphigus is an extremely rare subtype of pemphigus, with both IgG and IgA anti-cell surface antibodies. Although a relationship with IgA pemphigus or pemphigus herpetiformis has been suggested, the disease entity has not been established. We report the first Indian case of IgG/IgA pemphigus with additional curious reaction to epidermal basement membrane zone, and describe the results of histopathology, immunofluorescence, immunoblotting, and enzyme-linked immunosorbent assay (ELISA) studies.
Case Report
A 55-year-old Indian man presented with a 5-year history of recurrent, fluid-filled skin eruptions over the trunk and extremities. The lesions were intensely itchy and impeding his daily activities. There was no mucosal involvement during the disease course and no history suggestive of any other autoimmune diseases. The patient also had an episode of erythroderma, 2 years prior to presentation. He was treated previously with dexamethasone cyclophosphamide pulse therapy under diagnosis of pemphigus foliaceus, with poor results.
Physical examination revealed annular erythematous skin lesions with grouped vesicles and pustules on the trunk and extremities [Figure - 1]a and b. Nikolsky′s sign was negative. The results of full blood count, biochemical studies, and anti-nuclear antibodies were normal. Serum and urine electrophoresis did not suggest the presence of myeloma. Chest X-ray and ultrasound scan of abdomen and pelvis did not detect any malignancies.
 |
| Figure 1: Clinical appearance of skin lesions on the left leg showing grouped vesicles and background erythema (a) and lesions over left forearm showing annular erythematous plaques with peripheral vesiculo-pustules (b) Histopathology of three different skin biopsies taken from scaly lesion (c, ×200), superfi cial pustule (d, ×200), and deeper vesiculo-pustule (e, ×100) on the forearm (H and E stain) |
Biopsies taken from five different skin lesions during the disease course were reviewed. One biopsy from a scaly lesion showed a neutrophilic pustule within cornified layers of the epidermis [Figure - 1]c. All three biopsies from superficial pustules showed similar sub-corneal neutrophilic pustules, with occasional eosinophils and a few acantholytic cells [Figure - 1]d. Biopsy from a deeper vesiculo-pustule showed a mid-epidermal cleft with infiltrate of neutrophils and a few eosinophils. A few acantholytic cells were also present in the cleft [Figure - 1]e. None of the biopsies showed subepidermal blisters or any abnormal changes around the basement membrane zone.
Direct immunofluorescence of perilesional skin showed IgG deposits to keratinocyte cell surfaces [Figure - 2]a, weak IgA deposits to cell surfaces and basement membrane zone [Figure - 2]b, and C3 deposits to cell surfaces [Figure - 2]d. In addition, prominent granular deposits of IgG [Figure - 2]a, IgM [Figure - 2]c, and C3 [Figure - 2]d were seen along the basement membrane zone and in the upper papillary dermis.
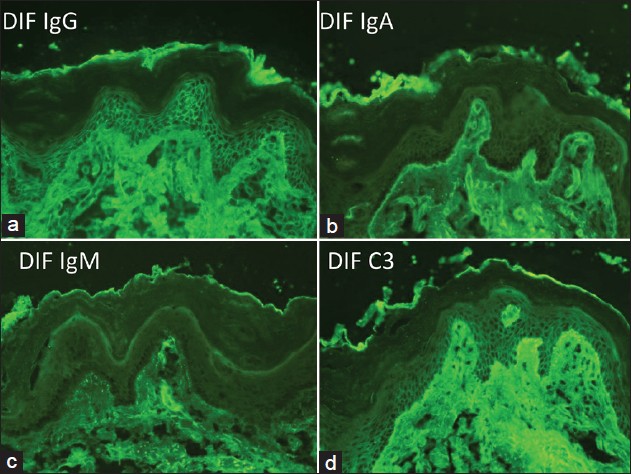 |
| Figure 2: The results of direct immunofluorescence of the skin biopsy for IgG (a), IgA (b), IgM (c), and C3 (d) |
Indirect immunofluorescence of normal human skin showed IgG [Figure - 3]a, but not IgA [Figure - 3]b, anti-keratinocyte cell surface antibodies over a titer of 1:160. In indirect immunofluorescence of 1M NaCl-split human skin, IgG [Figure - 3]c, but not IgA [Figure - 3]d, antibodies reacted with the epidermal side of the split. Indirect immunofluorescence of monkey esophagus showed IgG [Figure - 3]e and IgA [Figure - 3]f antibodies to cell surfaces of the epithelia.
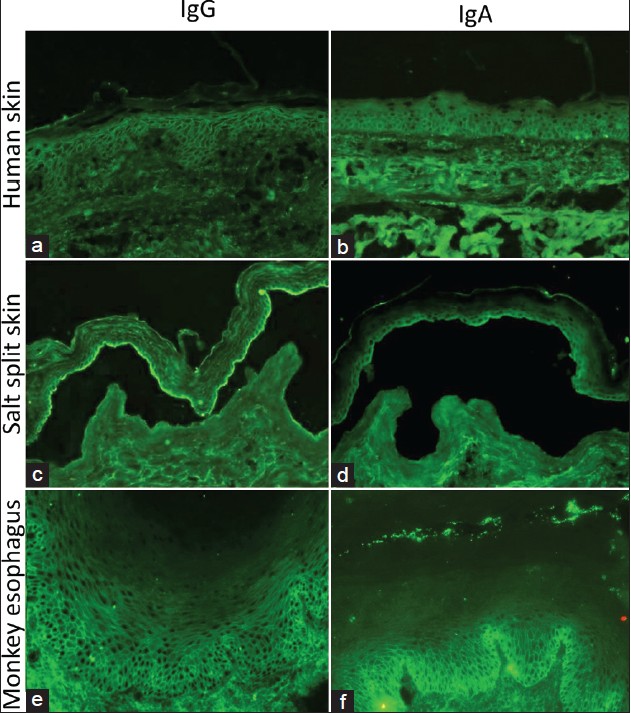 |
| Figure 3: Indirect immunofluorescence of normal human skin for IgG (a) and IgA (b) antibodies, of 1M NaCl-split human skin for IgG (c) and IgA (d) antibodies, and of monkey esophagus for IgG (e) and IgA (f) antibodies |
In immunoblot analysis of normal human epidermal extracts, IgG antibodies in patient serum reacted strongly with the 160 kDa Dsg1 but IgA antibodies showed no reactivity [Figure - 4]. Immunoblot analyses of recombinant proteins of BP180 NC16a and C-terminal domains, concentrated HaCaT cell culture medium, normal human dermal extracts, and purified human laminin332 showed negative results.
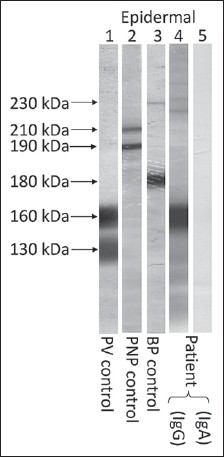 |
| Figure 4: The results of immunoblot assay of normal human epidermal extracts (Epidermal). The control pemphigus vulgaris (PV) serum reacted with 160 kDa Dsg1 and 130 kDa Dsg3 (lane 1), the control paraneoplastic pemphigus (PNP) serum reacted with 210 kDa envoplakin and 190 kDa periplakin (lane 2), and the control bullous pemphigoid (BP) serum reacted with 230 kDa BP230 and 180 kDa BP180 (lane 3). IgG (lane 4), but not IgA (lane 5), antibodies of this case (Patient) reacted strongly and exclusively with Dsg1 (lane 4) |
Commercially available IgG ELISA kits of recombinant baculoproteins of Dsg1 and Dsg3 (MBL, Nagoya, Japan) showed Dsg1 index of 218.99 and Dsg3 index of 0.20 (cut-off value: 20). IgA ELISA of Dsg1 and Dsg3 was performed as described previously, [2] showing Dsg1 OD 0.385 and Dsg3 OD 0.028 (cut-off value: 0.15). IgG or IgA antibodies to Dsc1-3 were not disclosed in Dsc cDNA-transfected COS-7 cell method. [1] The results of novel ELISA using eukaryotic recombinant proteins of human Dsc1-3 (Teye et al., in preparation) were negative for both IgG and IgA antibodies.
The patient was started on dapsone 100 mg/day, which induced dramatic improvement. All the eruptions healed within 2 weeks and no recurrence was observed.
Dicussion
The clinical and histopathological presentations and the results of various studies of auto-antibodies and auto-antigens in this case provided several important insights into understanding the pathogenicity of IgG/IgA pemphigus.
We diagnosed this case as IgG/IgA pemphigus based on the findings of direct and indirect immunofluorescence which showed the presence of both IgG and IgA immune reactants. On ELISA, IgG and IgA reactivity exclusively with Dsg1 was also detected. However, various histopathological, immunological, and clinical findings in our case encompassed features of IgA pemphigus, pemphigus herpetiformis, and pemphigus foliaceus.
Histopathologically, subcorneal pustules and spongiform pustules suggested subcorneal pustular dermatosis type IgA pemphigus while a mid-epidermal cleft containing acantholytic cells with neutrophilic infiltrate suggested intra-epidermal neutrophilic type IgA pemphigus. However, co-existence of eosinophils in the pustules in all biopsies suggested eosinophilic spongiosis, a feature more consistent with pemphigus herpetiformis. These findings may indicate that IgG/IgA pemphigus is a transitional form between IgA pemphigus and pemphigus herpetiformis, as suggested previously. [3],[4]
IgG/IgA pemphigus has been reported to show IgG and IgA reactivity with Dsg1 and/or Dsg3, [4],[5],[6] or with Dsc1. [7],[8] Kozlowska et al. reported a case of IgG/IgA pemphigus with clinical features of pemphigus herpetiformis and IgG and IgA reactivity with Dsg1 with additional IgG reactivity to Dsc1. [9] Pemphigus herpetiformis show IgG reactivity frequently with Dsg1 and occasionally with Dsg3. [10] Interestingly, all reported cases of IgG/IgA pemphigus (including the present case) with IgG and IgA reactivity exclusively with Dsg1 clinically behaved like pemphigus herpetiformis. [3],[4],[6],[11] The target antigen in most cases of subcorneal pustular dermatosis type of IgA pemphigus is Dsc1, whereas in intra-epidermal neutrophilic-type, occasional reactivity with Dsg1 or Dsg3 has been reported. [12],[13] These results of antigen analyses also suggest the relationship of IgG/IgA pemphigus with pemphigus herpetiformis and IgA pemphigus. In addition, this case showed pemphigus foliaceus-like findings, including superficial vesiculopustules, mid-epidermal cleft containing acantholytic cells, and the presence of IgG anti-Dsg1 antibodies. Therefore, the present case can also be diagnosed as a variant of pemphigus foliaceous and in a spectrum with IgA pemphigus and pemphigus herpetiformis.
Besides the pathological features, IgA pemphigus, IgG/IgA pemphigus, and pemphigus herpetiformis share many clinical features. All these disorders clinically present with extremely itchy annular erythemas with peripheral vesiculopustules. They show an excellent response to dapsone and are generally considered to be benign disorders with a better prognosis than pemphigus vulgaris. [14] However, unlike pemphigus herpetiformis, both IgA pemphigus and IgG/IgA pemphigus can rarely be associated with other medical disorders, including monoclonal gammopathy, Crohn′s disease, and gluten-sensitive enteropathy. [14]
Our case showed prominent granular deposits of IgG, IgM, and C3, as well as linear IgG and IgA reactivity with basement membrane zone. Granular deposits in basement membrane zone may be seen in lupus erythematosus, systemic sclerosis, pemphigus erythematosus as lupus band, or in lichen planus. Linear IgG and/or IgA reactivity may be seen in linear IgA bullous dermatosis or bullous lupus erythematosus. [15] However, our case cannot be diagnosed as these diseases because histopathology showed no subepidermal blisters or abnormal changes around basement membrane zone, and various antigen analyses could not detect any basement membrane zone antigens. The patient had no history of photosensitivity or malar erythema and did not show anti-nuclear antibodies to support the diagnosis of pemphigus erythematosus.
Granular deposits of IgM and C3 in papillary dermis can also be seen in vasculitis, although no forms of pemphigus have been reported to be associated with vasculitis. Our patient did not show any evidence of vasculitis either. Another possibility is that epitope spreading mechanism may produce auto-antibodies of IgG, IgA, and IgM classes reactive with novel antigens, which localize granularly in the papillary dermis or basement membrane zone.
Tsuruta et al. have proposed a possible new disease entity, tentatively called as C3 dermatitis herpetiformis or granular C3 dermatosis, which shows granular C3 deposits at papillary dermis or basement membrane zone without deposition of any immunoglobulins in direct immunofluorescence. [16] Such patients clinically show bullous or eczematous lesions and histopathologically show subepidermal edema with infiltrates of eosinophils and neutrophils, although no circulating auto-antibodies to basement membrane zone are detected. At present, the origin of C3 deposits is not known, although we suspect non-immunological pathways. Our case might be an atypical presentation of C3 dermatitis herpetiformis/granular C3 dermatosis coexisting with IgG/IgA pemphigus.
Santiago-et-Sanchez-Mateos et al. suggested that IgG/IgA pemphigus with anti-Dsg1 antibodies may show underlying malignancy. [4] However, no malignant tumor was found in our case. Dapsone was effective in our patient, similar to previously reported cases. [3],[5] Dapsone should be considered as a first-line therapy in the management of IgG/IgA pemphigus.
Acknowledgment
We gratefully appreciate the secretarial work of Ms. Hanako Nakagawa, Ms. Mihoko Ikeda and Ms. Sachika Notomi. We thank the patient for his participation.
| 1. |
Hashimoto T, Kiyokawa C, Mori O, Miyasato M, Chidgey MA, Garrod DR, et al. Human desmocollin 1 (Dsc1) is an autoantigen for the subcorneal pustular dermatosis type of IgA pemphigus. J Invest Dermatol 1997;109:127-31.
[Google Scholar]
|
| 2. |
Hashimoto T, Komai A, Futei Y, Nishikawa T, Amagai M. Detection of IgA autoantibodies to desmogleins by an enzyme-linked immunosorbent assay: The presence of new minor subtypes of IgA pemphigus. Arch Dermatol 2001;137:735-8.
[Google Scholar]
|
| 3. |
Kowalewski C, Hashimoto T, Amagai M, Jablonska S, Mackiewicz W, Wozniak K. IgA/IgG pemphigus: A new atypical subset of pemphigus? Acta Derm Venereol 2006;86:357-8.
[Google Scholar]
|
| 4. |
Santiago-et-Sanchez-Mateos D, Juarez Martin A, Gonzalez De Arriba A, Delgado Jimenez Y, Fraga J, Hashimoto T, et al. IgG/IgA pemphigus with IgA and IgG antidesmoglein 1 antibodies detected by enzyme-linked immunosorbent assay: Presentation of two cases. J Eur Acad Dermatol Venereol 2011;25:110-2.
[Google Scholar]
|
| 5. |
Nakajima K, Hashimoto T, Nakajima H, Yokogawa M, Ikeda M, Kodama H. IgG/IgA pemphigus with dyskeratotic acantholysis and intraepidermal neutrophilic microabscesses. J Dermatol 2007;34:757-60.
[Google Scholar]
|
| 6. |
Oiso N, Yamashita C, Yoshioka K, Amagai M, Komai A, Nagata Y, et al. IgG/IgA pemphigus with IgG and IgA antidesmoglein 1 antibodies detected by enzyme-linked immunosorbent assay. Br J Dermatol 2002;147:1012-7.
[Google Scholar]
|
| 7. |
Chorzelski TP, Hashimoto T, Nishikawa T, Ebihara T, Dmochowski M, Ismail M, et al. Unusual acantholytic bullous dermatosis associated with neoplasia and IgG and IgA antibodies against bovine desmocollins I and II. J Am Acad Dermatol 1994;31:351-5.
[Google Scholar]
|
| 8. |
Heng A, Nwaneshiudu A, Hashimoto T, Amagai M, Stanley JR. Intraepidermal neutrophilic IgA/IgG antidesmocollin 1 pemphigus. Br J Dermatol 2006;154:1018-20.
[Google Scholar]
|
| 9. |
Kozlowska A, Hashimoto T, Jarzabek-Chorzelska M, Amagai A, Nagata Y, Strasz Z, et al. Pemphigus herpetiformis with IgA and IgG antibodies to desmoglein 1 and IgG antibodies to desmocollin 3. J Am Acad Dermatol 2003;48:117-22.
[Google Scholar]
|
| 10. |
Ishii K, Amagai M, Komai A, Ebihara T, Chorzelski TP, Jablonska S, et al. Desmoglein 1 and desmoglein 3 are the target autoantigens in herpetiform pemphigus. Arch Dermatol 1999;135:943-7.
[Google Scholar]
|
| 11. |
Maruyama H, Kawachi Y, Fujisawa Y, Itoh S, Furuta J, Ishii Y, et al. IgA/IgG pemphigus positive for anti-desmoglein 1 autoantibody. Eur J Dermatol 2007;17:94-5.
[Google Scholar]
|
| 12. |
Karpati S, Amagai M, Liu WL, Dmochowski M, Hashimoto T, Horvath A. Identification of desmoglein 1 as autoantigen in a patient with intraepidermal neutrophilic IgA dermatosis type of IgA pemphigus. Exp Dermatol 2000;9:224-8.
[Google Scholar]
|
| 13. |
Tajima M, Mitsuhashi Y, Irisawa R, Amagai M, Hashimoto T, Tsuboi R. IgA pemphigus reacting exclusively to desmoglein 3. Eur J Dermatol 2010;20:626-9.
[Google Scholar]
|
| 14. |
Robinson ND, Hashimoto T, Amagai M, Chan LS. The new pemphigus variants. J Am Acad Dermatol 1999;40:649-71.
[Google Scholar]
|
| 15. |
Huilgol SC, Bhogal BS, Black MM. Immunofluorescence of the immunobullous disorders Part two: The clinical disorders. Indian J Dermatol Venereol Leprol 1995;61:255-64.
[Google Scholar]
|
| 16. |
Tsuruta D, Ishii N, Koga H, Hamada T, Dainichi T, Ohata C, et al. Granular C3 dermatosis, a possibly new disease entity. Abstracts of the Annual Meeting of the Society for Investigative Dermatology. May 9-12, 2012. Raleigh, North Carolina, USA. J Invest Dermatol 2012;132:S1-180.
[Google Scholar]
|
Fulltext Views
3,094
PDF downloads
1,780





