Translate this page into:
Interstitial granulomatous drug reaction due to febuxostat
2 Department of Cyto Morphology, Division of Pathology, University of Cagliari, Italy
Correspondence Address:
Laura Atzori
Clinica Dermatologica, Via Ospedale 54, 09124 Cagliari
Italy
| How to cite this article: Atzori L, Pilloni L, Pinna AL. Interstitial granulomatous drug reaction due to febuxostat. Indian J Dermatol Venereol Leprol 2014;80:182-184 |
Sir,
Febuxostat is a selective xanthine oxidase inhibitor, approved as second-line therapy in adult symptomatic hyperuricemia. Adverse effects of the drug are poorly documented. A regulatory trial reported the occurrence of a skin rash in 0.5-1.6% of patients [1] while a retrospective study on gout patients with prior documented severe allopurinol reactions noted the development of a hypersensitivity type cutaneous vasculitis in one of 13 patients. [2] We report a case in which an interstitial granulomatous drug reaction developed during febuxostat administration.
A 78-year-old Caucasian man presented with a 1-month history of asymptomatic, sharply demarcated annular erythematous lesions, symmetrically distributed on the legs [Figure - 1]. Older lesions merged into polycyclic patches, with rather pale and atrophic centers, covered with adherent follicular scales. There was no regional lymphadenopathy. Application of topical steroids for several weeks induced no improvement.
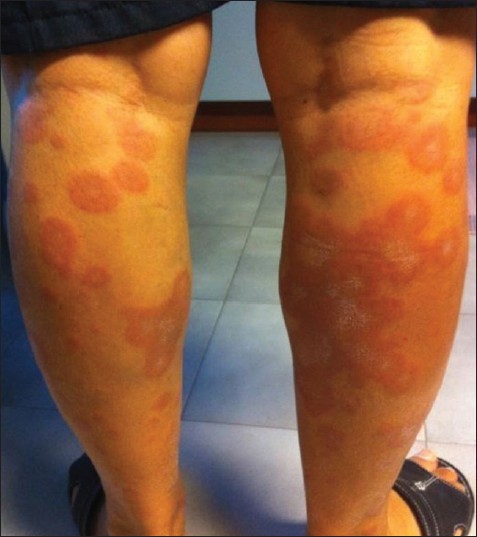 |
| Figure 1: Annular erythematous-edematous plaques on the posterior side of both legs tending to coalescence in polycyclic figures and showing central follicular scaling in older lesions |
Skin biopsy showed a normal epidermis, mild diffuse interstitial and perivascular inflammation, with a prominent histiocytic component, spilled interstitially between the collagen bundles [Figure - 2], upper left inset]. On closer view [Figure - 2], upper right inset] piecemeal fragmentation of collagen and elastic fibers, small and medium sized lymphocytes, sometimes with hyperchromic nuclei, but showing no atypia and some eosinophils were also present.
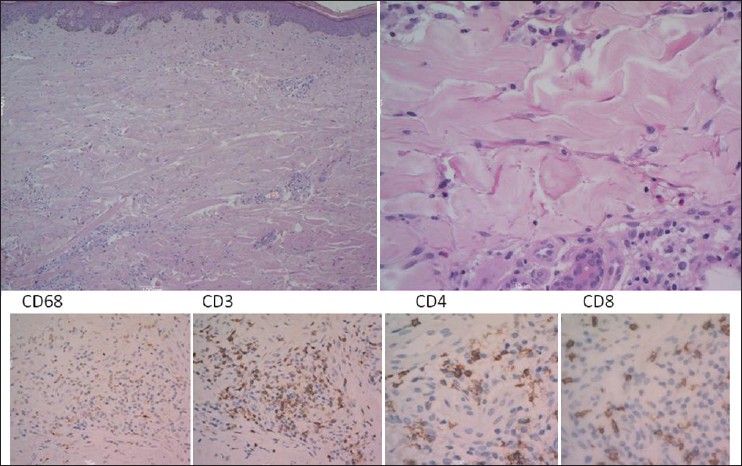 |
| Figure 2: Histology of the skin biopsy showed normal thickness epidermis, mild pervascular and interstitial lymphocytic inflammation in the underlying dermis, with a prominent histiocytes component (H and E, ×100). On higher magnification (H and E, ×400), histiocytes appeared spilled interstitially between fragmented collagen bundles, some small lymphocytes showed hyper chromic nuclei, eosinophils were also noted (right side inset); immunohistochemical stain for histiocytes (CD68) confi rmed their abundance; CD3, CD4 and CD8 were normally expressed |
Medical history revealed a previous myocardial infarction and persistent atrial fibrillation treated daily for more than 5 years with hydrochlorothiazide-fosinopril 20 + 12.5 mg, torasemide 10 mg, barnidipine 10 mg, simvastatin 40 mg, metoprolol 100 mg, isosorbide mononitrate 80 mg, and acenocoumarol 4 mg. In the last 6 months, his general practitioner had prescribed febuxostat 80 mg, twice daily for asymptomatic hyperuricemia. The choice of this novel drug was made because it is not chemically related to allopurinol, which was suspected to have caused a similar skin eruption that spontaneously recovered after drug withdrawal 11 months prior to presentation in our department. On that occasion, the patient declined any investigations. No personal or family history of arthritis or autoimmune diseases was reported.
Investigations were performed to exclude other etiologies or associated conditions, including autoimmune diseases, chronic infections and malignancies. The only alterations documented were a slight leukocytosis with a white blood cell count of 10600/mm 3 (normal range 4000-10000/mm 3) eosinophilia of 160/μl (normal range 50-900/μl), hyperglycemia of 113 mg/dl (normal range 74-106 mg/dl), and hyperuricemia of 9.51 mg/dl (normal range 3.5-7.2 mg/dl).
Febuxostat was withdrawn and after four weeks the lesions disappeared [Figure - 3]a, while reintroduction caused a sudden flare [Figure - 3]b. No relapses occurred in the following 6 months. Final causality assessment for adverse drug reaction following Jones algorithm was definite. [3]
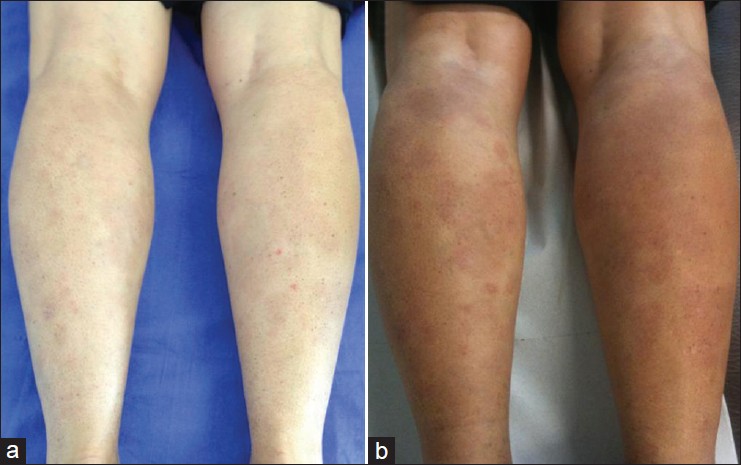 |
| Figure 3: Skin recovery after febuxostat withdrawal (a); flare-up after drug rechallenge (b) |
Among the wide spectrum of cutaneous drug reactions, Magro et al. in 1998 described a rare entity with a peculiar histopathology characterized by the dermal presence of histiocytes between fragmented collagen bundles, coining the name interstitial granulomatous drug reaction. [4] The clinical presentation is variable and includes asymptomatic annular plaques with a predilection for the flexures, generalized erythematous macules and/or papules, erythroderma and DRESS. [5] Our patient had an unusual site of involvement and polycyclic plaques with central follicular scaling suggesting several differential diagnoses including mycosis fungoides. In their report, Magro et al. had pointed out the resemblance of interstitial granulomatous drug reaction to mycosis fungoides, with some cases showing atypical lymphocytes on light microscopy. [4] However, a careful examination performed by our pathologist excluded atypical lymphocytic infiltrates and epidermotropism. In addition, there were no palisading granulomas typical of granuloma annulare, no vasculitis nor septal panniculitis suggestive of erythema nodosum. Features of erythema annulare centrifugum such as papillary edema, spongiosis and parakeratosis were absent.
Interstitial granulomatous drug reaction completely regresses with drug cessation and awareness of this will avoid prolonged disease and unnecessary treatments. The list of culprit drugs has increased with time and includes calcium channel blockers, angiotensin-converting enzyme inhibitors, lipid-lowering agents, antihistamines, diuretics, anticonvulsants, antidepressants, allopurinol, thalidomide and anti-tumor necrosis factor (TNF) agents. [3],[4],[5],[6] The role of TNF-alpha inhibitors in causing interstitial granulomatous drug reaction is controversial, as the drugs have been reported to cure the condition during the course of rheumatoid arthritis but have also been implicated in the onset of interstitial granulomatous reactions in other cases of psoriatic and rheumatoid arthritis. [6] The mechanisms underlying interstitial granulomatous dermatitis, associated with arthritis or drug intake, are unknown. The common histopathological hallmark is activation of histiocytes and recruitment of lymphocytes, which might be further amplified or inhibited by anti-TNF agents. Recovery without immunosuppressant treatment and the long interval between drug initiation and onset of the eruption suggests a temporary immunologic reactivity and/or restoration by spontaneous removal of the causative agent. [3],[4],[5] Previously reported allopurinol-induced interstitial granulomatous reactions were characterized by constitutional symptoms and internal organ involvement characteristic of the DRESS syndrome ],[5] in which a type IV hypersensitivity has been postulated to play a role. Our patient was taking several drugs for years without complaints until the intake of allopurinol caused a cutaneous eruption, not further investigated due to the patient′s refusal, but not suspected to be DRESS.
To the best of our knowledge, febuxostat has not been associated with interstitial granulomatous drug reaction or DRESS. It is possible that xanthine oxidase inhibitors unveil a granulomatous diathesis in certain patients and hyperuricemia is the common feature connecting our case with the previous allopurinol-induced interstitial granulomatous reactions. [5] Both allopurinol and febuxostat prevent uric acid formation by blocking the conversion of xanthine precursors. However, both drugs are ineffective during an acute attack of gout and might even induce it, because the lower circulating uric acid levels favor the release of micro-crystals from the cartilage into the joint cavity triggering the synovial inflammatory cascade. [1] Dermal and subcutaneous soft tissue might react similarly to the dissolution of uric acid crystal deposits with exaggerated histiocyte activation. Drug-induced unknown dermal products and/or a disturbance in the degradation of collagen fibers might be responsible for the final interstitial granulomatous inflammation. [3],[4],[5] Further studies are necessary to confirm the role of febuxostat and predisposing individual conditions to the development of interstitial granulomatous drug reaction.
Acknowledgments
We thank Dr. Robin Mark Weathon for the kind revision of the English text.
| 1. |
Ernst ME, Fravel MA. Febuxostat: A selective xanthine-oxidase/xanthine-dehydrogenase inhibitor for the management of hyperuricemia in adults with gout. Clin Ther 2009;31:2503-18.
[Google Scholar]
|
| 2. |
Chohan S. Safety and efficacy of febuxostat treatment in subjects with gout and severe allopurinol adverse reactions. J Rheumatol 2011;38:1957-9.
[Google Scholar]
|
| 3. |
Jones JK. Criteria for journal reports of suspected drug reactions. Clin Pharm 1982;1:554-5.
[Google Scholar]
|
| 4. |
Magro CM, Crowson AN, Schapiro BL. The interstitial granulomatous drug reaction: A distinctive clinical and pathological entity. J Cutan Pathol 1998;25:72-8.
[Google Scholar]
|
| 5. |
Kim MS, Lee JH, Park K, Son SJ. Allopurinol-induced : DRESS syndrome with a histologic pattern consistent with interstitial granulomatous drug reaction. Am J Dermatopathol 2014;36:193-6.
[Google Scholar]
|
| 6. |
Deng A, Harvey V, Sina B, Strobel D, Badros A, Junkins-Hopkins JM, et al. Interstitial granulomatous dermatitis associated with the use of tumor necrosis factor alpha inhibitors. Arch Dermatol 2006;142:198-202.
[Google Scholar]
|
Fulltext Views
2,649
PDF downloads
679





