Translate this page into:
Intralesional immunotherapy for the management of warts
Correspondence Address:
Laxmisha Chandrashekar
Department of Dermatology and STD, JIPMER, Pondicherry - 605 006
India
| How to cite this article: Chandrashekar L. Intralesional immunotherapy for the management of warts. Indian J Dermatol Venereol Leprol 2011;77:261-263 |
Management of verrucae, particularly over the periungual area and over the soles, is often frustrating to the patient and the physician alike. Currently available treatment modalities include cryosurgery, laser surgery, electrosurgery, bleomycin, curettage, and topical keratolytic applications; many of them are likely to be very painful, unsightly, and prone for recurrences. [1]
Immunotherapy for warts has been performed with diphenylcyclopropenone (DCP), squaric acid dibutyl ester (SADBE), imiquimod, tuberculin jelly, and autologous vaccines. The use of DCP and SADBE is limited by allergic contact dermatitis, urticarial reactions and pigmentary disturbances. Autologous vaccine therapy is limited by the oncogenic potential of the virus. Therefore, a safe, inexpensive, effective and simple immunotheurapeutic agent is needed for the management of warts. [1]
Recurrence rates of warts up to 30% have been reported with cryotherapy probably due to a lack of immune response. Various reasons like lack of production of memory T cells to target human papillomavirus (HPV) infection, failure of clonal expansion of lymphocytes to adequate stimulation, inability of T lymphocytes to traffic to sites of infection and weak effector response mechanism have been reported. [2]
Intralesional immunotherapy utilizes the ability of the immune system to mount a delayed type hypersensitivity response to various antigens and also the wart tissue. This therapy has been found to be associated with the production of Th1 cytokines which activate cytotoxic and natural killer cells to eradicate HPV infection. This clears not only the local warts but also distant warts unlike traditional wart therapies. [3] Kim et al., [4] in their phase 1 clinical trial of the intralesional injection of Candida antigen reported that the immune response to HPV-57 L1-peptide (380-412) was most commonly demonstratable. All the participants who showed this response demonstrated clearing of their wart. This suggests that L-1 specific T cells may be involved in wart regression. It has also been found that responders to intralesional immunotherapy were found to have a significant positive peripheral mononuclear cell proliferation assay as compared to non-responders (P = 0.002). [5]
Although this is an elegant and simplified procedure, it has not become popular. Harada in 1979 first documented the effect of Candida antigen in the treatment of warts. [6] Bolton and Ricker at the University of Minnesota performed a double-blind, placebo-controlled trial to demonstrate the efficacy of Candida antigen in the treatment of warts using Candida extract (1:100). This was associated with excessive tissue sloughing. However, in their previous study with Candida antigen extract (1:1000), they reported a 71% cure rate with no significant adverse effects. [1]
Many authors have used different immunotherapeutic agents for intralesional injection. These include autogenous vaccine, [7] Candida antigen, [1],[2] mumps antigen, [5],[8] trichophytin skin test antigen, [5] tuberculin, [9],[10] BCG vaccine, [11] MMR vaccine, [3] Mycobacterium w vaccine [12] and interferon alpha and gamma injection. This procedure utilizes the fact that there is a high prevalence of immunity to these antigens in the general population. It has been found that older individuals (>40 years) are less likely to respond to this modality as compared to younger individuals on account of the less robust immunological responses with increasing age. [5]
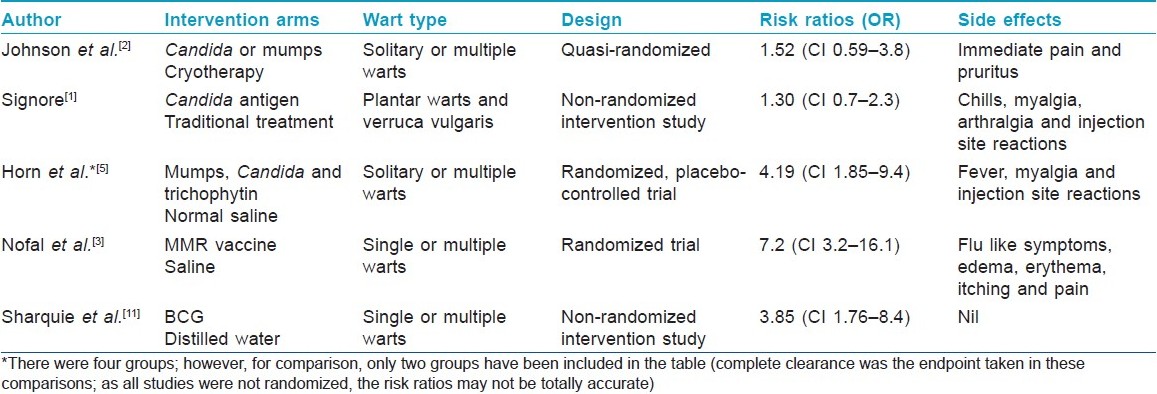
There are two different approaches used by various authors. In the first approach, the individual is injected with an intradermal antigen on the volar aspect of the forearm and the delayed hypersensitivity response is assessed as erythema and induration 48-72 hours later. Responders (with erythema and induration of 5 mm in diameter) are taken up for the study. Johnson et al., [2] injected warts, usually the largest one, with the Candida or mumps antigen in a volume determined by the size of the test reaction [Table - 1]. Some authors have directly injected the antigen into the largest wart without doing a preliminary intradermal testing. Warts are injected intralesionally using an insulin syringe. The syringe is held parallel with the skin surface with the bevel facing upward. This therapy is repeated every 3 weeks till complete clearance of the warts or for a maximum of three treatments without response.
There are a number of studies on immunotherapy. Most of them are open labeled. The studies with a control arm are summarized in [Table - 2]. Analysis is done on an intention to treat basis. However, this procedure is not suitable for those with hypersensitivity to any of these antigens, pregnant females and immunosuppressed individuals.

Immunotherapy in Children
Warts in children are difficult to treat and may be very painful with traditional methods, especially if they are multiple. As distant warts are also cleared with this therapy, they offer a good modality of treatment in this group. Clifton et al., [13] used intralesional mumps and Candida antigen for the treatment of recalcitrant warts in children. They reported a 47% clearance of warts injected directly and a 34% clearance for distant warts. They felt that intralesional immunotherapy could be a first line for large or multiple warts and a second line in warts recalcitrant to standard therapy. Maronn et al., [14] reported their 1 year experience with intralesional Candida antigen therapy for both warts and molluscum contagiosum. In the wart group, 48/55 (87%) had complete resolution. All the wart and molluscum patients experienced some discomfort at the time of injection, but no serious side effects were reported. In the study by Signore, [1] children comprised 45% of the Candida immunotherapy group. The injection of the antigen was well tolerated by most children.
Indian Status
Autoinoculation of warts has been used in India for the management of extensive warts. The mechanism of action postulated is similar to that of intralesional therapy. Gupta et al., [12] have reported on the efficacy of Mycobacterium w in the management of anogenital warts. In eight of nine patients, the genital wart cleared completely. Two of their patients who were seropositive for HIV infection experienced an immune reconstitution inflammatory syndrome. The authors did not recommend the use of this therapy in immunosuppressed individuals.
There are a few side effects reported by most of the studies. The most common include injection site reactions and flu like symptoms. This is probably due to injection of the antigen into the circulation with subsequent immunological response and elaboration of cytokines. [2] Most of these reactions last up to 24 hours and resolve with the use of nonsteroidal anti-inflammatory drugs. Rare adverse events include painful purple digit and post-immunotherapy revealed cicatrix (PIRC). Perman et al., [15] reported a painful purple digit following Candida albicans immunotherapy into a wart. They postulated that this could be a type of compartmental syndrome. PIRC is noted in patients who had been treated with destructive therapies prior to immunotherapy. The scar would be hidden as the wart grows. When the wart clears by immunotherapy, the scar is revealed. This has to be explained to the patients as this could be a medicolegal complication. [1] It should be administered with caution in patients on beta blockers as they may become unresponsive to epinephrine in the event of anaphylaxis. [1]
This form of therapy is inexpensive. Cost of treatment with Candida immunotherapy works out to be less than a dollar per sesion. [1] Treatments with tuberculin and BCG vaccine are likely to be cheaper. This could be a very useful modality of treatment of warts in poor and developing countries like ours.
Although there are many open labeled studies reporting the efficacy of intralesional immunotherapy, there are few randomized control trials (RCT) on this subject. In a systematic review on topical therapy for warts, Gibbs and their colleagues found no advantage in using intralesional immunotherapy as there are few well-designed RCTs addressing this question. [16]
Many of these intervention comparisons are limited by differing concentrations, vehicles, intervals between treatments, number of injections, follow-up period for assessing outcomes and spontaneous wart involution.
I feel that this is an exciting modality of treatment which needs well-designed RCTs to assess its overall efficacy.
| 1. |
Signore RJ. Candida albicans intralesional injection immunotherapy of warts. Cutis 2002;70:185-92.
[Google Scholar]
|
| 2. |
Johnson SM, Roberson PK, Horn TD. Intralesional injection of mumps or Candida skin test antigens: A novel immunotherapy for warts. Arch Dermatol 2001;137:451-5.
[Google Scholar]
|
| 3. |
Nofal A, Nofal E. Intralesional immunotherapy of common warts: Successful treatment with mumps, measles and rubella vaccine. J Eur Acad Dermatol Venereol 2010;24:1166-70.
[Google Scholar]
|
| 4. |
Kim KH, Horn TD, Pharis J, Kincannon J, Jones R, O'Bryan K, et al. Phase 1 clinical trial of intralesional injection of Candida antigen for the treatment of warts. Arch Dermatol 2010;146:1431-3.
et al. Phase 1 clinical trial of intralesional injection of Candida antigen for the treatment of warts. Arch Dermatol 2010;146:1431-3. '>[Google Scholar]
|
| 5. |
Horn TD, Johnson SM, Helm RM, Roberson PK. Intralesional immunotherapy of warts with mumps, Candida, and Trichophyton skin test antigens: A single-blinded, randomized, and controlled trial. Arch Dermatol 2005;141:589-94.
[Google Scholar]
|
| 6. |
Harada S. [Clinical application of fungus extracts and its culture filtrate in the treatment of skin diseases; (3) Candida vaccine in the treatment of warts (author's transl)]. Nippon Hifuka Gakkai Zasshi 1979;89:397-402.
[Google Scholar]
|
| 7. |
Wiltz OH, Torregrosa M, Wiltz O. Autogenous vaccine: the best therapy for perianal condyloma acuminata? Dis Colon Rectum 1995;38:838-41.
[Google Scholar]
|
| 8. |
Johnson SM, Horn TD. Intralesional immunotherapy for warts using a combination of skin test antigens: A safe and effective therapy. J Drugs Dermatol 2004;3:263-5.
[Google Scholar]
|
| 9. |
Eassa BI, Abou-Bakr AA, El-Khalawany MA. Intradermal injection of PPD as a novel approach of immunotherapy in anogenital warts in pregnant women. Dermatol Ther 2011;24:137-43.
[Google Scholar]
|
| 10. |
Kus S, Ergun T, Gun D, Akin O. Intralesional tuberculin for treatment of refractory warts. J Eur Acad Dermatol Venereol 2005;19:515-6.
[Google Scholar]
|
| 11. |
Sharquie KE, Al-Rawi JR, Al-Nuaimy AA, Radhy SH. Bacille Calmette-Guerin immunotherapy of viral warts. Saudi Med J 2008;29:589-93.
[Google Scholar]
|
| 12. |
Gupta S, Malhotra AK, Verma KK, Sharma VK. Intralesional immunotherapy with killed Mycobacterium w vaccine for the treatment of ano-genital warts: An open label pilot study. J Eur Acad Dermatol Venereol 2008;22:1089-93.
[Google Scholar]
|
| 13. |
Clifton MM, Johnson SM, Roberson PK, Kincannon J, Horn TD. Immunotherapy for recalcitrant warts in children using intralesional mumps or Candida antigens. Pediatr Dermatol 2003;20:268-71.
[Google Scholar]
|
| 14. |
Maronn M, Salm C, Lyon V, Galbraith S. One-year experience with Candida antigen immunotherapy for warts and molluscum. Pediatr Dermatol 2008;25:189-92.
[Google Scholar]
|
| 15. |
Perman M, Sterling JB, Gaspari A. The painful purple digit: An alarming complication of Candida albicans antigen treatment of recalcitrant warts. Dermatitis 2005;16:38-40.
[Google Scholar]
|
| 16. |
Gibbs S, Harvey I, Sterling J, Stark R. Local treatments for cutaneous warts: Systematic review. BMJ 2002;325:461.
[Google Scholar]
|
Fulltext Views
10,514
PDF downloads
2,232





