Translate this page into:
Leukemia cutis presenting as scaly plaques in a Christmas tree distribution in a patient with atypical chronic myeloid leukemia
2 Department of Pathology, Jawaharlal Institute of Postgraduate Medical Education and Research, Puducherry, India
Correspondence Address:
Rashmi Kumari
Department of Dermatology, Venereology and Leprology, Jawaharlal Institute of Postgraduate Medical Education and Research, Puducherry - 605 006
India
| How to cite this article: Behera B, Kumari R, Manobalan K, Thappa DM, Basu D, Patel BK. Leukemia cutis presenting as scaly plaques in a Christmas tree distribution in a patient with atypical chronic myeloid leukemia. Indian J Dermatol Venereol Leprol 2017;83:236-238 |
Sir,
A 75-year-old woman presented with multiple scaly lesions on the bilateral upper extremities, trunk and face for 8 months. The patient gave a history of anorexia, fatigue and weight loss of around 10 kg during the last 6 months. On examination, the patient had pallor, generalized lymphadenopathy and hepatosplenomegaly. Cutaneous examination revealed multiple erythematous, infiltrated scaly plaques distributed symmetrically on the back in a “Christmas tree pattern” [Figure - 1]. Some infiltrated plaques were also noticed on the face. The differential diagnoses of Sezary syndrome, leukemia cutis and secondary syphilis was considered. She was hypertensive. Laboratory investigations revealed hemoglobin of 9.4 g/dl, platelet count of 176,000 cells/cumm and total leukocyte count of 75,700 cells/cumm with 48% of neutrophils, 21% lymphocytes, 4% monocytes, 14% myelocytes, 10% metamyelocytes and 3% blasts. Peripheral smear showed a marked leukocytosis with blast cells, which were strongly positive for Sudan Black B. The serum lactic dehydrogenase was 327 IU/L and leukocyte alkaline phosphatase score was 23 as compared to the reference value of 129. Ultrasonography of the abdomen and pelvis revealed hepatosplenomegaly. Other investigations were within normal limits. Skin biopsy showed a dermal interstitial and periadnexal collection of cells of the myeloid series, plasma cells and some lymphocytes [Figure - 2]. On immunohistochemistry, myeloid cells were positive for myeloperoxidase and CD117. Histopathological examination of the lymph node biopsy revealed partial effacement of architecture, paracortical and sinusoidal infiltration by intermediate cells having increased mitotic figures and a prominent nucleus. On immunohistochemistry, malignant cells were positive for CD117, focally positive for myeloperoxidase and negative for TDT, CD10, CD61 and CD56. All these features were suggestive of myeloid-type extramedullary blast crisis without differentiation in the lymph node. Bone marrow study revealed features of a chronic myelodysplastic/myeloproliferative neoplasm with evidence of dyspoiesis in myeloid and megakaryocytic lineage along with focal small clusters of immature cells (that comprised approximately 5% of the marrow nucleated cells). On immunohistochemistry, these cells were positive for myeloperoxidase and CD117. Reticulin fibrosis (World Health Organization Grade 1) was also noted in the marrow biopsy. The breakpoint cluster region-Abelson transcript quantification by real-time reverse transcriptase-polymerase chain reaction was 0%. Based on these, a diagnosis of leukemia cutis secondary to an atypical chronic myeloid leukemia was made. The patient was treated with hydroxyurea in view of her old age and associated comorbidities.
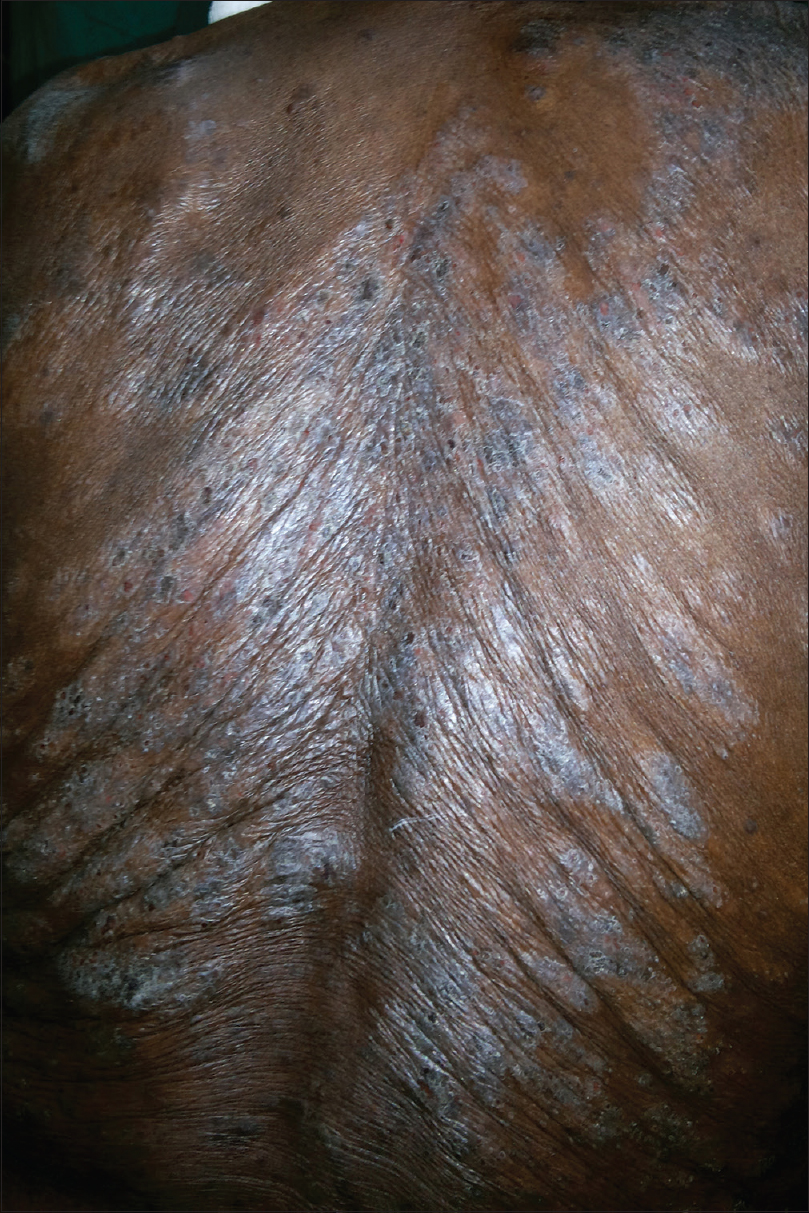 |
| Figure 1: Multiple symmetrical erythematous scaly plaques on the back distributed in a Christmas tree pattern |
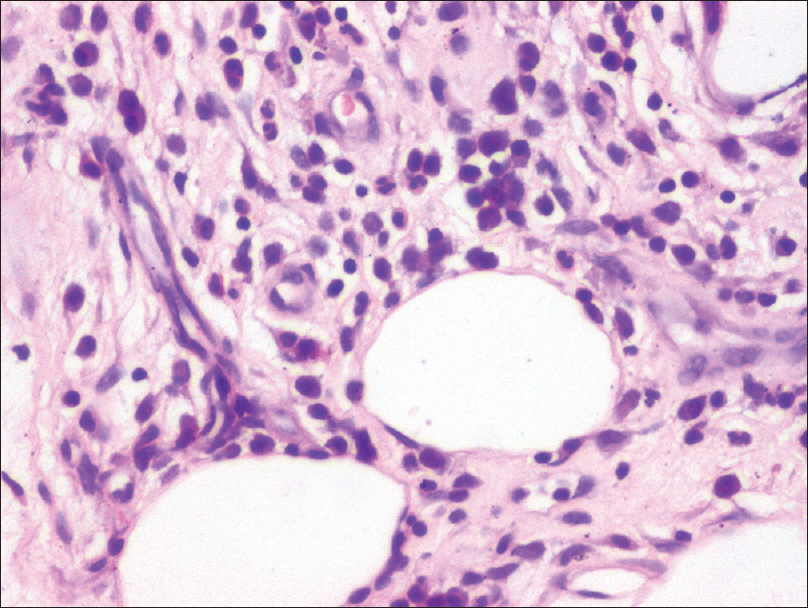 |
| Figure 2: Skin biopsy showing perivascular myeloid cells in the subcutaneous tissue (H and E, ×400) |
Cutaneous manifestations of leukemias/lymphomas can be classified into leukemids or non-specific lesions and leukemia cutis or specific lesions.[1] Leukemia cutis has been described in acute myeloid leukemia (most common, 10–15%), chronic myeloid leukemia, myelodysplastic syndrome and lymphoproliferative diseases.[2] Atypical chronic myeloid leukemia is characterized by a leukemic proliferation of myeloid lineage cells with features of both myeloproliferation and myelodysplasia at the time of initial diagnosis. Atypical chronic myeloid leukemia shares clinical and laboratory features with chronic myeloid leukemia but lacks the breakpoint cluster region-Abelson fusion gene.[3] Leukemia cutis is rare in chronic myeloid leukemia with an incidence of 2–8%, and usually occurs during blast crisis.[4] It appears to be even rarer in atypical chronic myeloid leukemia and we were able to find only one previous report of a violaceous papule on the scalp.[5] Of the various clinical presentations of leukemia cutis, erythematous to violaceous papules or nodules are the most frequent lesions (60%), followed by infiltrated plaques, a generalized cutaneous eruption and erythroderma.[6] In our case, the patient presented with multiple scaly plaques on the back distributed in a Christmas tree pattern. Various unusual morphologies described include figurate erythemas, guttate lesions, stasis dermatitis and ulcers.[6] Leukemia cutis is also found to localize at the sites of trauma, intravenous catheters, surgical sites and herpetic lesions.[6]
The disorders that can present with multiple scaly plaques are pityriasis rosea (typical collarette of scale at the margin of the lesions), secondary syphilis (involvement of palms, soles and mucosa), psoriasis vulgaris (positive Auspitz sign and coarse nail pitting), psoriasiform drug rash (temporal relation with intake of a drug), subacute cutaneous lupus erythematosus (history of photosensitivity and oral ulcer) and Reiter's disease (associated with joint pain and/or swelling). Furthermore, none of these conditions have any cutaneous infiltration, as was present in our case.
The histolopathological hallmark is a perivascular and/or periadnexal or a dense diffuse/interstitial or nodular infiltration of leukemic cells involving the dermis and subcutis. Immunohistochemistry stains are mandatory to characterize the immunophenotype of tumor cells. Detailed analysis of the peripheral blood picture, bone marrow smears and genetic profile are crucial for the diagnosis of the specific type of leukemia.[2] Leukemia cutis is a marker of poor prognosis and indicates that the disease course is aggressive and most patients (85%) die within 4 months after diagnosis.[7]
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. |
Rao AG, Danturty I. Leukemia cutis. Indian J Dermatol 2012;57:504.
[Google Scholar]
|
| 2. |
Cho-Vega JH, Medeiros LJ, Prieto VG, Vega F. Leukemia cutis. Am J Clin Pathol 2008;129:130-42.
[Google Scholar]
|
| 3. |
Wang L, Du F, Zhang HM, Wang HX. Evaluation of a father and son with atypical chronic myeloid leukemia with SETBP1 mutations and a review of the literature. Braz J Med Biol Res 2015;48:583-7.
[Google Scholar]
|
| 4. |
Raj A, Rai R, Rangarajan B. Exfoliative dermatitis with leukemia cutis in a patient with chronic myeloid leukemia: A rare association. Indian J Dermatol Venereol Leprol 2011;77:208-10.
[Google Scholar]
|
| 5. |
Lane SW, Fairbairn DJ, McCarthy C, Nandini A, Perry-Keene J, Kennedy GA. Leukaemia cutis in atypical chronic myeloid leukaemia with a t(9;22) (p24;q11.2) leading to BCR-JAK2 fusion. Br J Haematol 2008;142:503.
[Google Scholar]
|
| 6. |
Wong TY, Suster S, Bouffard D, Flynn SD, Johnson RA, Barnhill RL, et al. Histologic spectrum of cutaneous involvement in patients with myelogenous leukemia including the neutrophilic dermatoses. Int J Dermatol 1995;34:323-9.
[Google Scholar]
|
| 7. |
Singhal M, Singh S, Kumar R, Raina V. Extensive cutaneous manifestations: Presenting feature of chronic myelocytic leukemia in second blast crisis. Indian J Dermatol 2010;55:265-7.
[Google Scholar]
|
Fulltext Views
1,770
PDF downloads
811





