Translate this page into:
Linear IgA bullous dermatosis due to vancomycin and cutaneous necrosis due to terlipressin in the same patient
2 Department of Pharmacy, Hospital del Sureste, Arganda del Rey, Madrid, Spain
3 Department of Gastroenterology, Hospital del Sureste, Arganda del Rey, Madrid, Spain
4 Department of Pathology, Hospital del Sureste, Arganda del Rey, Madrid, Spain
Correspondence Address:
María Castellanos-González
Calle Sánchez Pacheco, 75, 1C, 28002, Madrid
Spain
| How to cite this article: Castellanos-González M, Marzal-Alfaro MB, Díaz-Sánchez A, Martos MG. Linear IgA bullous dermatosis due to vancomycin and cutaneous necrosis due to terlipressin in the same patient. Indian J Dermatol Venereol Leprol 2016;82:723-726 |
Sir,
Dermatologists are used to dealing with adverse drug reactions but the increasing use of the new-generation medications have unveiled novel, less-known entities some of which may be of a severe nature.
A 54-year-old man with Child-Pugh C alcoholic cirrhosis was admitted with acute variceal bleeding. An upper gastrointestinal endoscopy with variceal band ligation was performed and he was started on treatment with vancomycin. He received vancomycin for 3 days. Subsequently, the patient developed a type 1 hepatorenal syndrome and hence, terlipressin was initiated and vancomycin stopped considering the risk of nephrotoxicity.
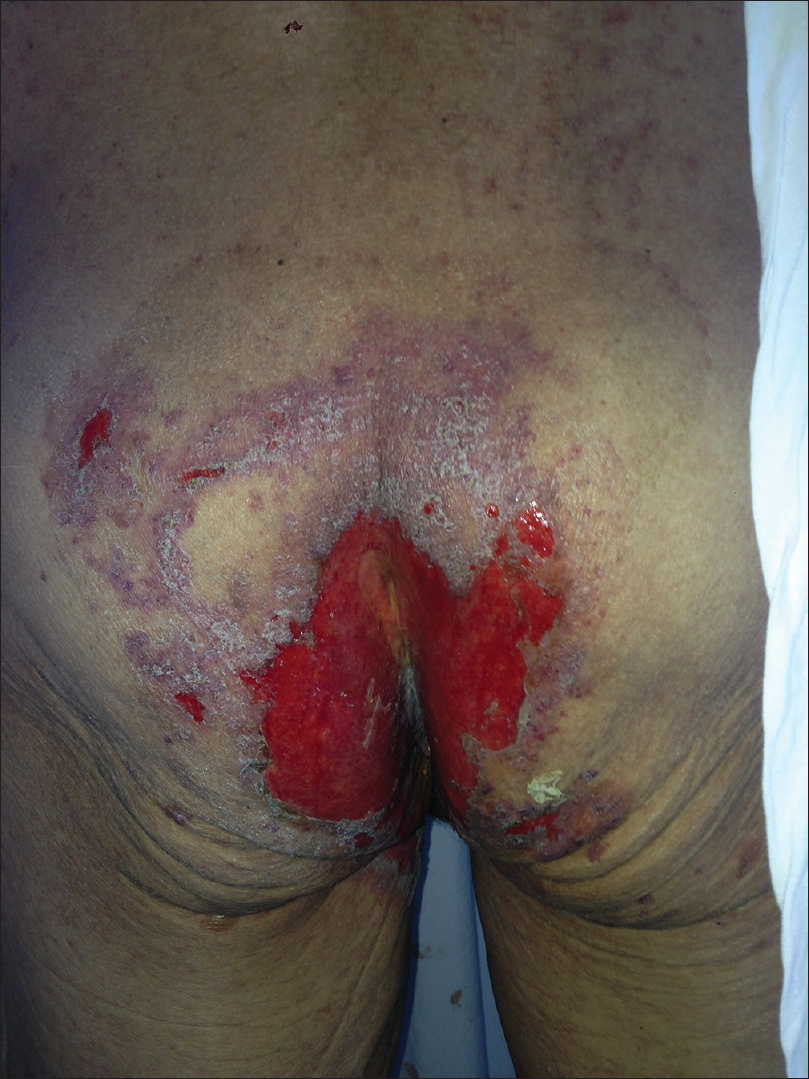
Two weeks following admission, the patient developed discrete and grouped tense bullae on his trunk, soles and feet. A large denuded area was observed on the buttocks and erosions were also seen in the oral mucosa [Figure - 1]. The possibility of toxic epidermal necrolysis syndrome was considered and a skin biopsy was performed. We decided to continue terlipressin in view of the underlying hepato-renal involvement and systemic corticosteroids were started for the cutaneous eruption. Meanwhile, the skin biopsy showed subepidermal cleavage with neutrophilic inflammatory infiltrate and direct immunofluorescence revealed intense linear deposition of IgA at the basement membrane zone [Figure - 2]. Thus, a diagnosis of linear IgA bullous dermatosis due to vancomycin was made. As vancomycin had been previously stopped, we did not change the treatment and the patient improved with systemic steroids.
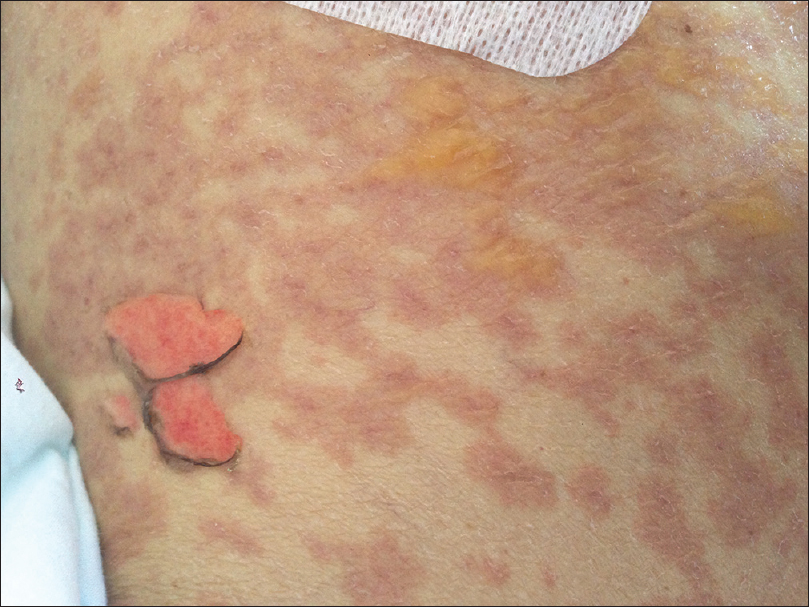 |
| Figure 1a: The patient presented big denuded areas that resembled toxic epidermal necrolysis |
 |
| Figure 1b: The patient presented big denuded areas that resembled toxic epidermal necrolysis |
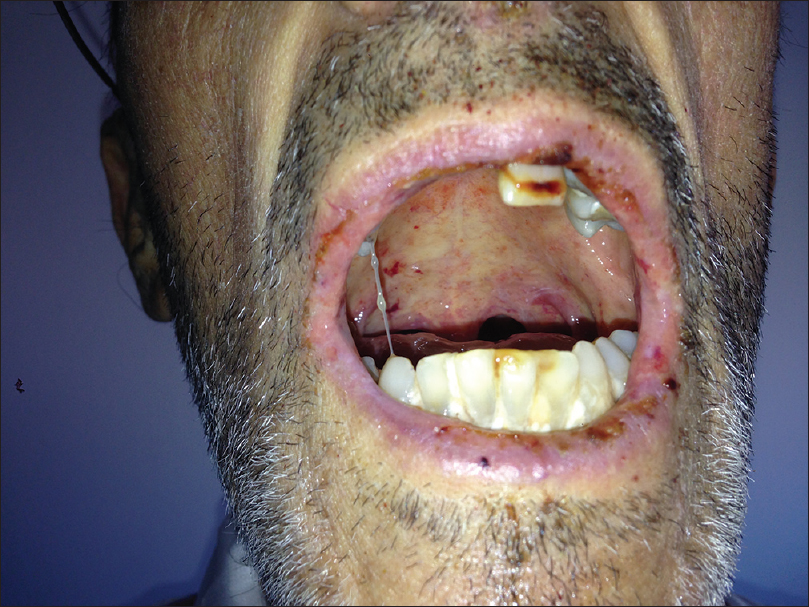 |
| Figure 1c: The oral mucosa was completely affected with hemorrhagic crusts on lips and denuded and erosive areas |
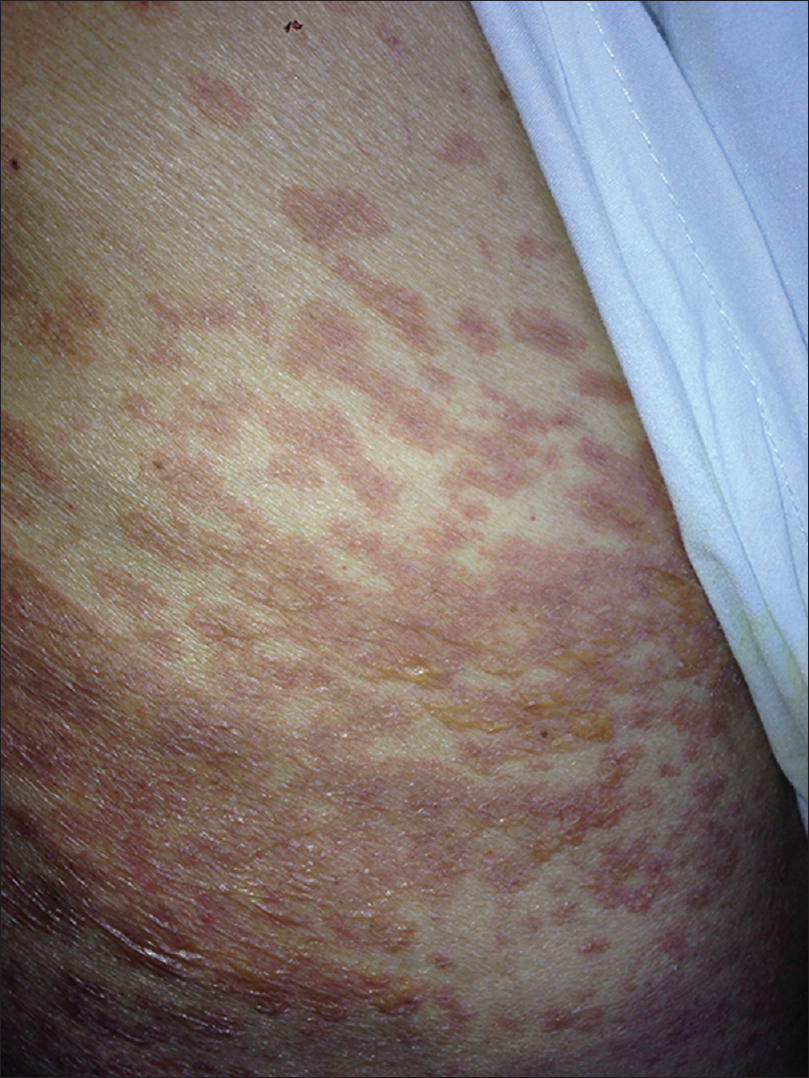 |
| Figure 1d: Some serous, confl uent bullae were observed in trunk |
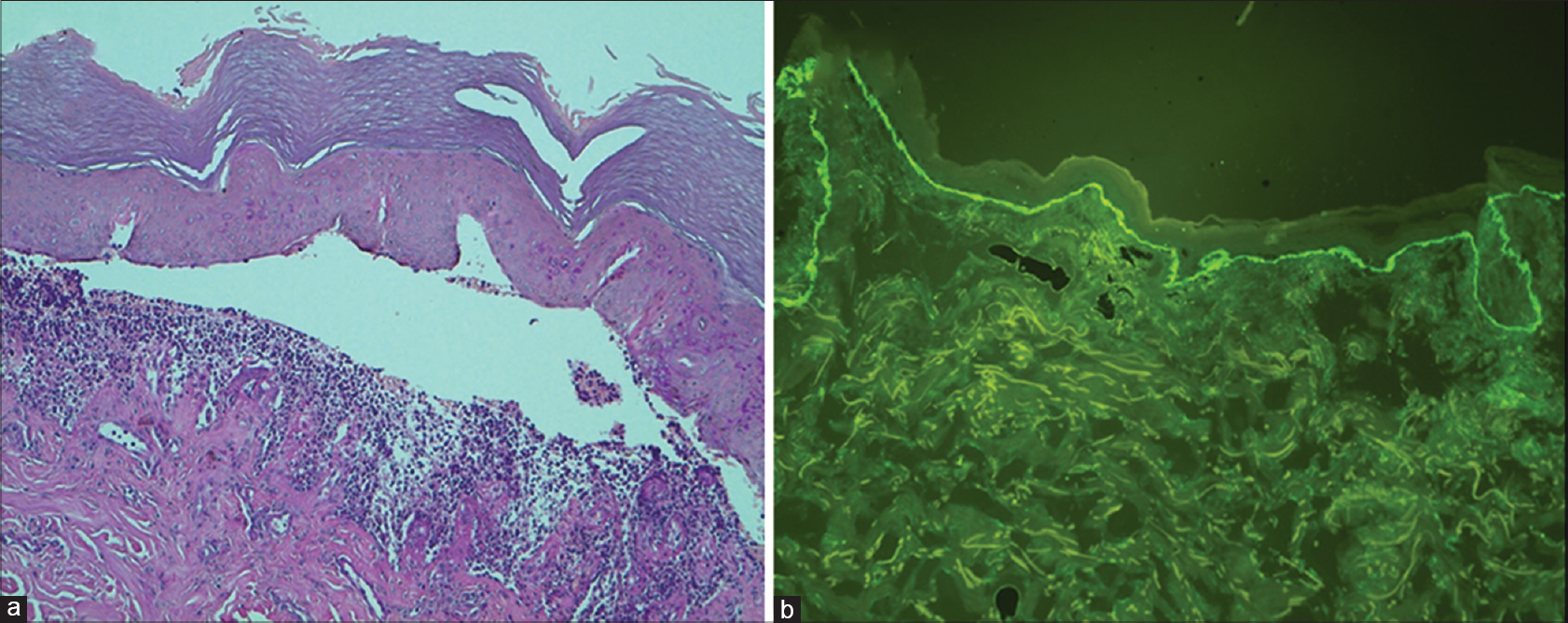 |
| Figure 2: (a) Subepidermal cleavage with mixed infl ammatory infi ltrate including neutrophils and eosinophils (H and E, ×100). (b) IgA immunofl uorescence stain. The direct immunofl uorescence testing of perilesional skin showed intense linear deposition of immunoglobulin A at the basal membrane zone (x100) |
One week later, the patient developed generalized discomfort accompanied by oval, violaceous and confluent papules on the lower limbs and large necrotic areas with sharp demarcation on the thighs, ankles and toes that further extended in the next 2 days [Figure - 3]. Investigations including an echocardiogram, immunological analysis, antinuclear antibodies, antineutrophil cytoplasmic antibodies, IgG, A, M, complement studies and cryoglobulins were normal or negative. Levels of proteins C, S and platelet count were decreased, probably due to the underlying hepatic failure. Anticardiolipin and anti-beta2 glycoprotein IgA antibodies were normal. Blood cultures were sterile. A new skin biopsy showed detachment of the necrotic epidermis, intense spongiosis and red blood cell extravasation with perivascular infiltration of mononuclear cells in the deep dermis without signs of thrombotic vasculopathy or vasculitis. The diagnosis of skin necrosis induced by terlipressin was made and the medication was stopped. Unfortunately, although his skin lesions dramatically improved after drug discontinuation, hepatorenal syndrome worsened and he died 5 days later.
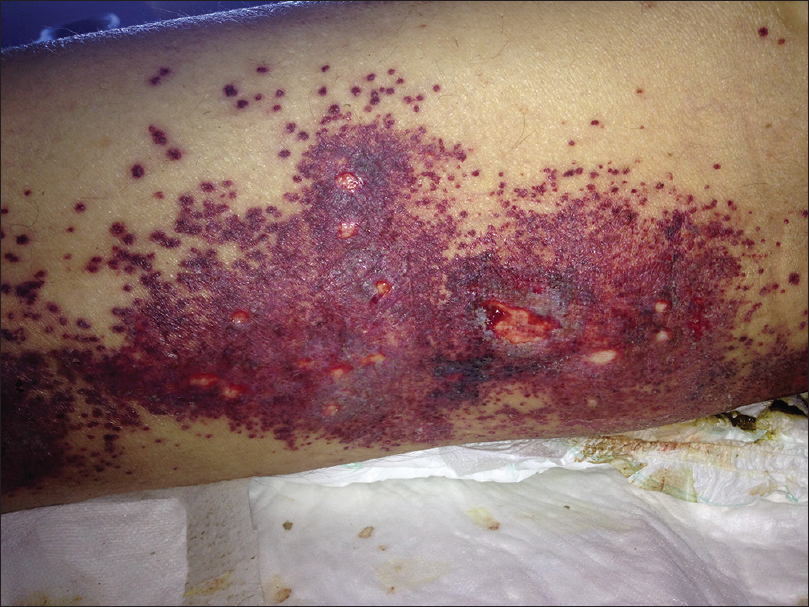 |
| Figure 3a: Violaceous, confl uent papules and necrosis areas in lower limbs were developed 48 hours after terlipressin was started |
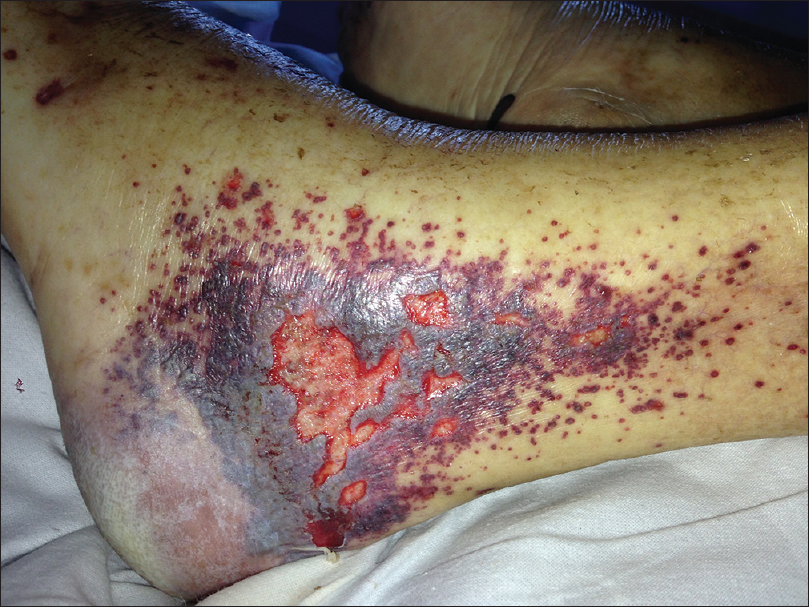 |
| Figure 3b: Violaceous, confl uent papules and necrosis areas in lower limbs were developed 48 hours after terlipressin was started |
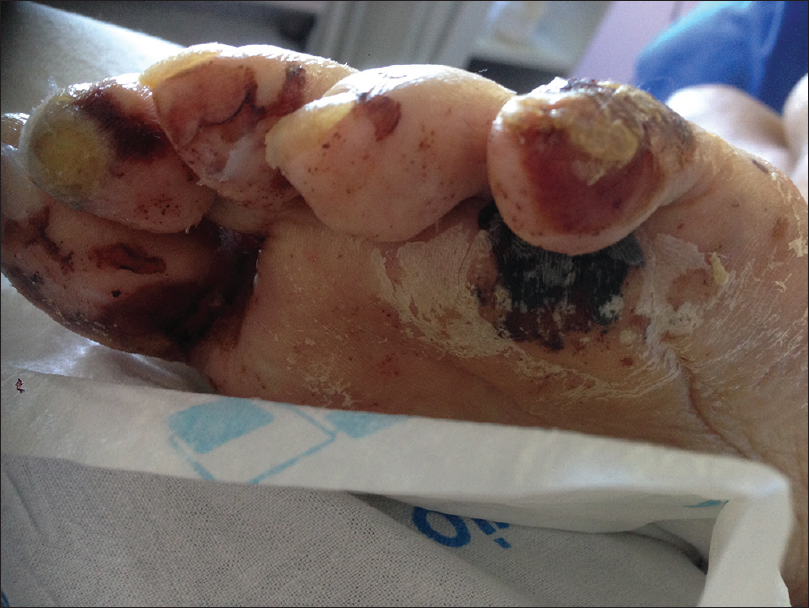 |
| Figure 3c: Acral areas such as toes were also affected |
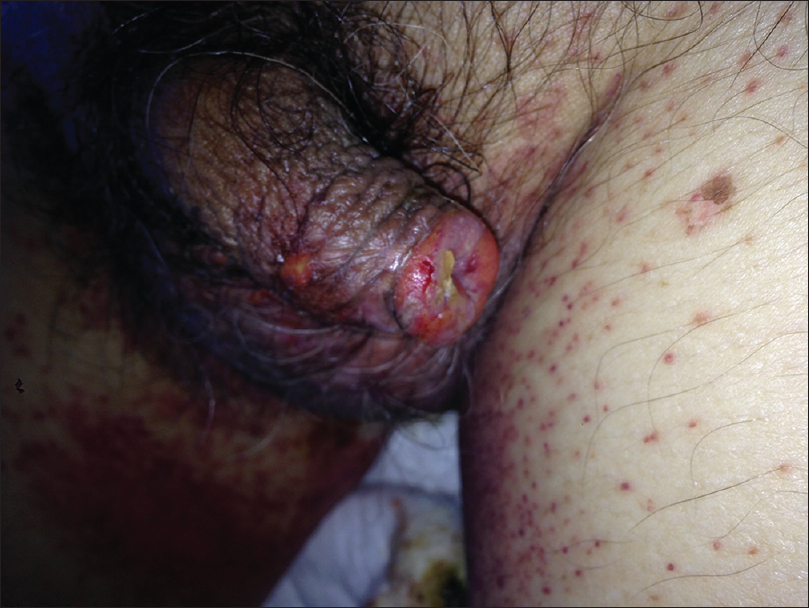 |
| Figure 3d: Although the mucosa oral was spared, genital area was severely affected with small ulcers and pain |
Terlipressin is a non-selective V1 arginine-vasopressin analogue with a potent and prolonged vasoconstrictor effect. It is widely used in the management of hepatorenal syndrome in cirrhotic patients. Some of its well-known adverse effects include paleness, increased blood pressure and headache. However, less common but potentially life-threatening complications of tissue ischemia including terlipressin-induced extensive skin necrosis have been recently reported.[1]
Linear IgA dermatosis is an autoimmune bullous disorder characterized by subepidermal blister formation and linear and homogenous deposition of IgA in the basement membrane zone. Although uncommon, the clinical presentation may resemble toxic epidermal necrolysis, as occurred in our patient. Other medications considered to be associated with this entity include penicillin, ceftriaxone and metronidazole, however, vancomycin has been most consistently implicated.[2]
There are only a few cases of skin necrosis caused by terlipressin reported till date.[3] All reported patients suffered from cirrhosis due to different etiologies. Terlipressin dosages varied from 0.5 mg to 2 mg every 4–6 h. The route of administration was intravenous. The onset of skin necrosis varied between 1 and 14 days after the initiation of drug treatment. There is only one reported pediatric case, a 2-month-old infant who presented with progressive ischemia of the right lower limb and intestinal ischemia.[1],[2],[4],[5]
The ischemic complications can be attributed to its vasoconstrictive effect compromising tissue oxygenation. The estimated frequency of this adverse effect is 5%.[4] Despite having a selective action on the splanchnic circulation, terlipressin also has systemic effects causing ischemic manifestations in various tissues. This complication may be related to the unique distribution of the target receptor of terlipressin which is located in the smooth muscles of blood vessels, mainly in the territory of the splanchnic circulation, kidney, myometrium, bladder, adipocytes and skin circulation. Thus, the skin manifestations occur along this distribution and are frequent in parts of the body with large skin surfaces such as thighs and abdomen.
In addition, there seem to be some risk factors of cutaneous necrosis such as obesity, hypovolemia and the concomitant use of pressor drugs. The severity of skin ischemia is not linked to the dose of terlipressin. However, continuous infusion has been reported as an additional risk.
We present a unique patient who presented with two different rare cutaneous adverse events in the same month. Recognizing these conditions would help clinicians to improve the management of their patients.
Acknowledgments
We would like to acknowledge J. L. Rodriguez-Peralto for the histopathological study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. |
Dellavalle RP, Burch JM, Tayal S, Golitz LE, Fitzpatrick JE, Walsh P. Vancomycin-associated linear IgA bullous dermatosis mimicking toxic epidermal necrolysis. J Am Acad Dermatol 2003;48 5 Suppl:S56-7.
[Google Scholar]
|
| 2. |
Donnellan F, Cullen G, Hegarty JE, McCormick PA. Ischaemic complications of Glypressin in liver disease: A case series. Br J Clin Pharmacol 2007;64:550-2.
[Google Scholar]
|
| 3. |
Lu YY, Wei KC, Wu CS. Terlipressin-induced extensive skin necrosis: A case report and published work review. J Dermatol 2012;39:866-8.
[Google Scholar]
|
| 4. |
Lee HJ, Oh MJ. A case of peripheral gangrene and osteomyelitis secondary to terlipressin therapy in advanced liver disease. Clin Mol Hepatol 2013;19:179-84.
[Google Scholar]
|
| 5. |
Herrera I, Leiva-Salinas M, Palazón JM, Pascual JC, Niveiro M. Extensive cutaneous necrosis due to terlipressin use. Gastroenterol Hepatol 2015;38:12-3.
[Google Scholar]
|
Fulltext Views
1,724
PDF downloads
902





