Translate this page into:
Linear IgA bullous dermatosis induced by diclofenac sodium
Correspondence Address:
Sarita Sanke
Department of Dermatology and S T D, Lady Hardinge Medical College and Associated Hospitals, Shaheed Bhagat Singh Marg, New Delhi - 110 001
India
| How to cite this article: Sanke S, Kumar A, Chander R. Linear IgA bullous dermatosis induced by diclofenac sodium. Indian J Dermatol Venereol Leprol 2018;84:496-497 |
An 18-year-old female presented with multiple, fluid-filled lesions all over the body of 5 days duration, which was preceded by intake of oral diclofenac 100mg for leg pain 3 days prior to the development of lesions. Cutaneous examination showed multiple, clear, fluid-filled as well as hemorrhagic vesicles and bullae all over the body ranging from 0.5 cm to 4 cm in diameter forming a “string of pearl” configuration [Figure - 1]. All mucosae were normal. Nikolsky's sign and bulla spread sign were negative. Histopathology from a fresh vesicle showed subepidermal blister with perivascular and interstitial inflammatory infiltrate composed of neutrophils and eosinophils.. Direct immunofluorescence from the perilesional skin revealed linear deposition of IgA along the dermo-epidermal junction. The Naranjo score for adverse drug reaction was 7 (probable drug reaction). In the absence of any other precipitating factors, constitutional features, and normal laboratory findings, a diagnosis of linear IgA bullous disease induced by diclofenac sodium was made.
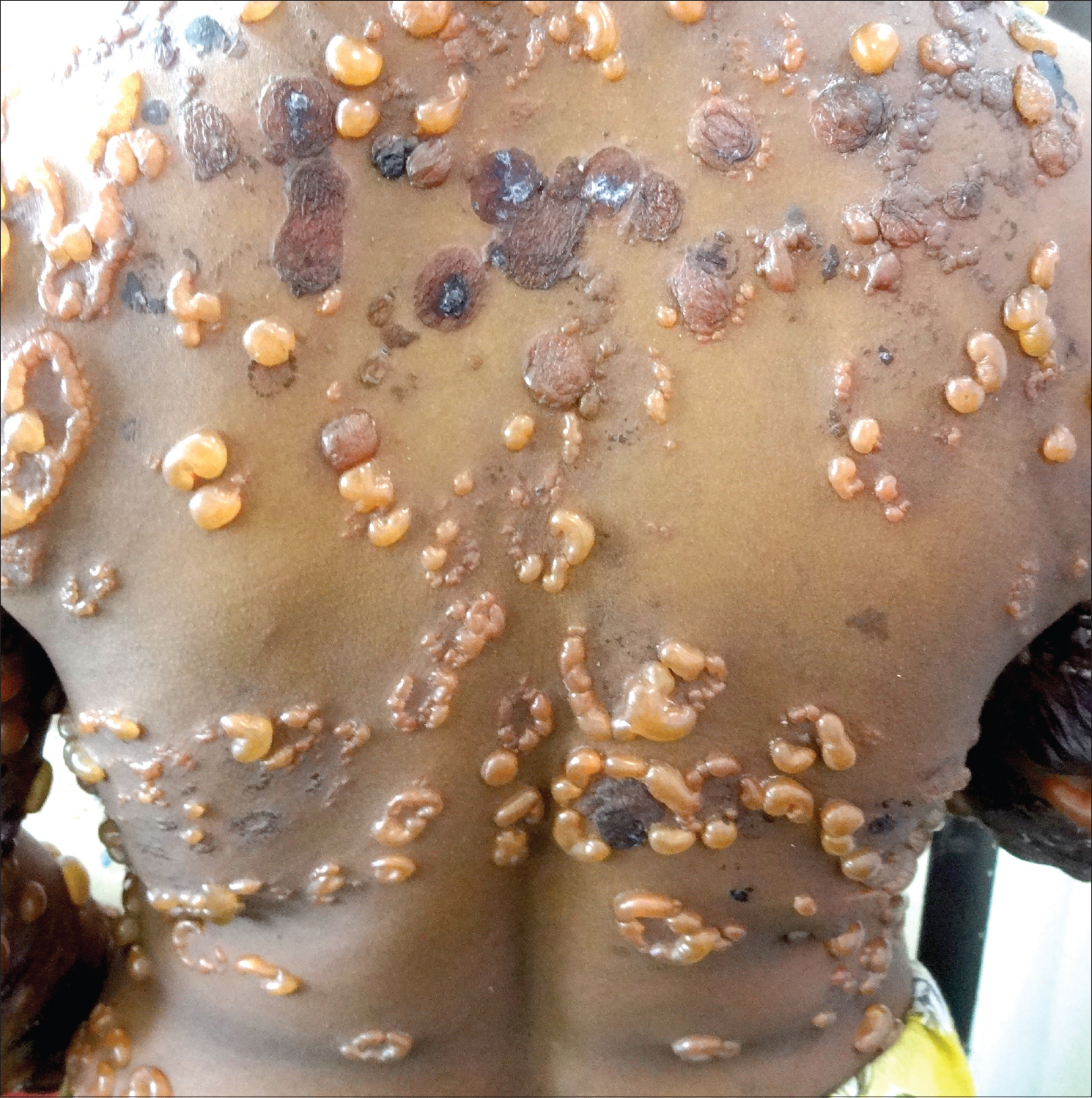 |
| Figure 1: Multiple clear to hemorrhagic fluid-filled bullae in a “string of pearl” configuration |
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Fulltext Views
1,962
PDF downloads
1,119





