Translate this page into:
Metastatic tubercular abscess associated with bone and lymph node involvement
2 Department of Pathology, Medical College and Hospital, Kolkata, West Bengal, India
Correspondence Address:
Indrashis Podder
Block AD-71, Salt Lake City, Kolkata - 700 064, West Bengal
India
| How to cite this article: Sancheti K, Podder I, Saha M, Chowdhury SN, Bandyopadhyay D. Metastatic tubercular abscess associated with bone and lymph node involvement. Indian J Dermatol Venereol Leprol 2017;83:276 |
Sir,
Tuberculosis is seldom considered an etiology of subcutaneous abscess in our daily practice. Here, we report a rare case of metastatic tubercular abscess in a malnourished girl, with bone involvement and lymphadenopathy.
A 13-year-old malnourished girl presented with multiple abscesses predominantly involving her upper and lower extremities for the last 2 years. She had multiple, variably sized, subcutaneous discharging abscesses on the upper extremity, lower extremity and neck [Figure - 1] and [Figure - 2]. The lesions were fluctuant and non-tender without any sign of inflammation. Matted, enlarged lymph nodes were noted in the cervical [Figure - 3] and inguinal regions [Figure - 4]. Scars were observed at the sites of previous drainage. She started developing these abscesses on her neck and extremities which progressively increased in size. They had been drained unsuccessfully, only to reappear again. She also received multiple courses of systemic antibiotics and antifungals with no appreciable improvement. There was no history of fever, cough and breathlessness at presentation. The past history revealed an episode of pulmonary tuberculosis, five years back, along with weight loss. Family history was unremarkable. The patient had not been immunized with bacillus Calmette–Guérin (BCG) vaccine. There was pallor, and malnutrition (body mass index 15.5 kg/m2); the remainder of the general examination was normal.
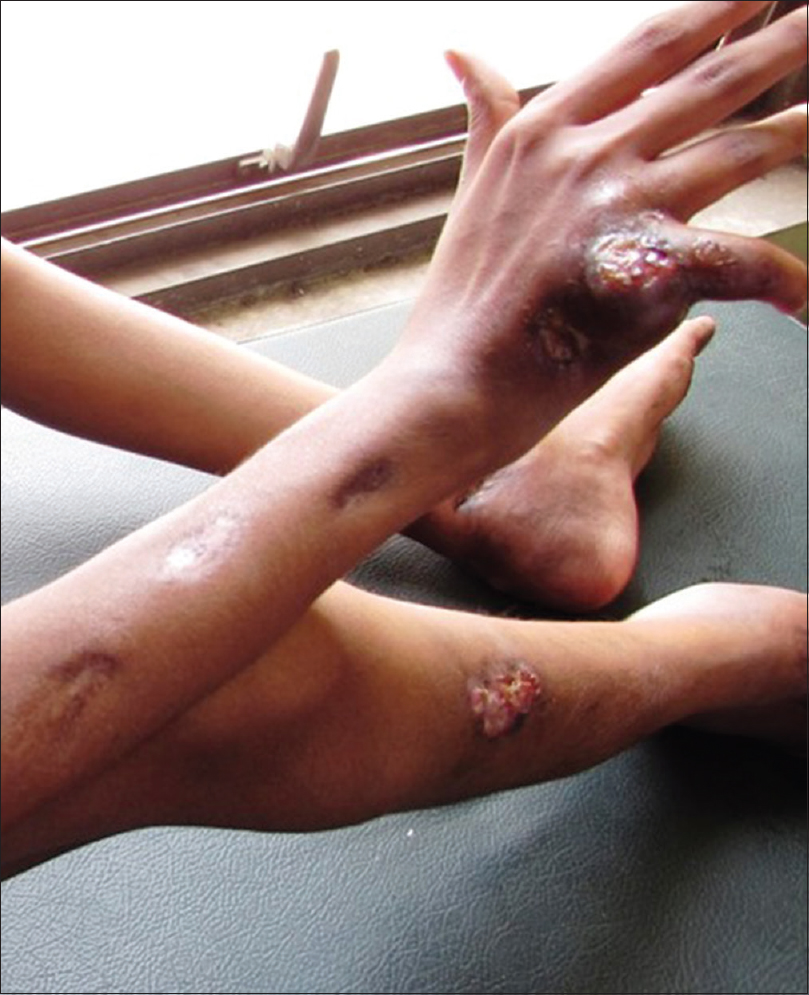 |
| Figure 1: Multiple, discharging subcutaneous abscesses on the right upper and lower limbs |
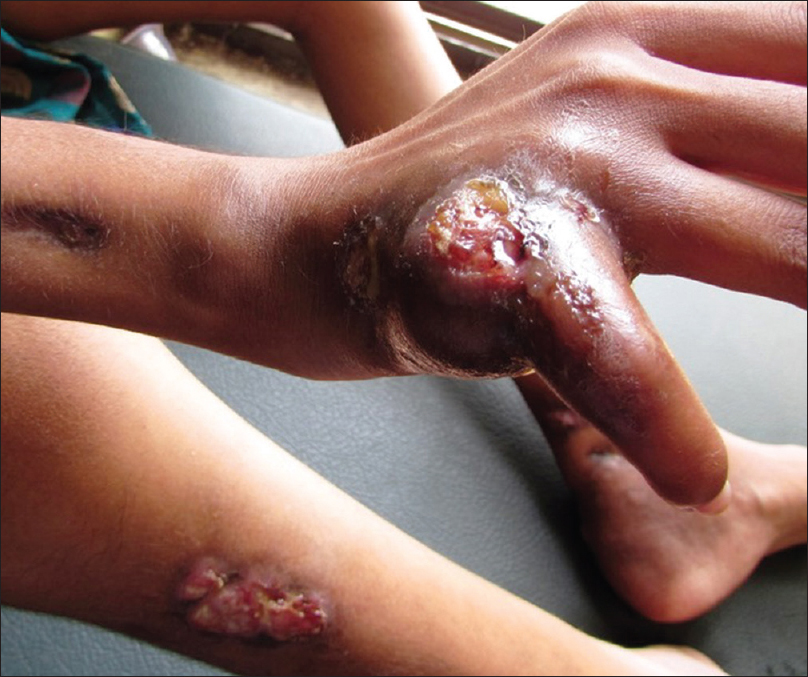 |
| Figure 2: Necrotic ulcer on the right hand |
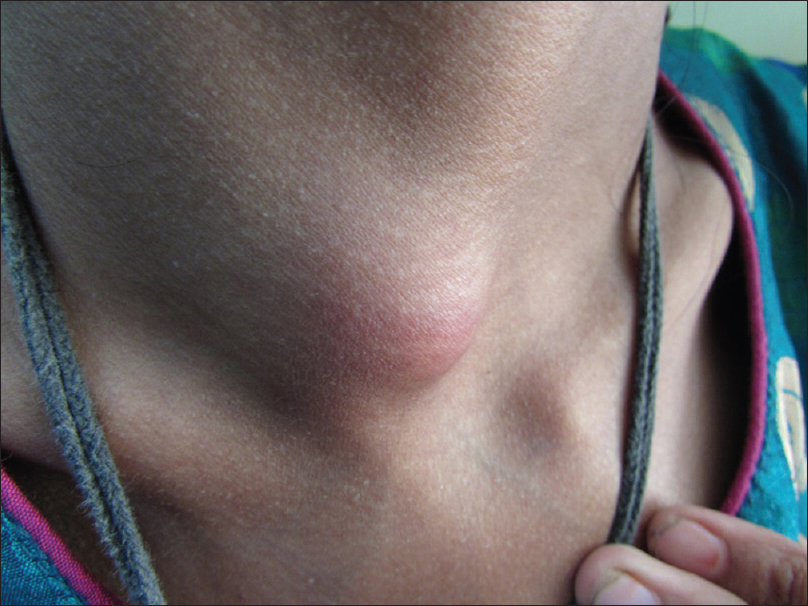 |
| Figure 3: Cervical lymphadenopathy |
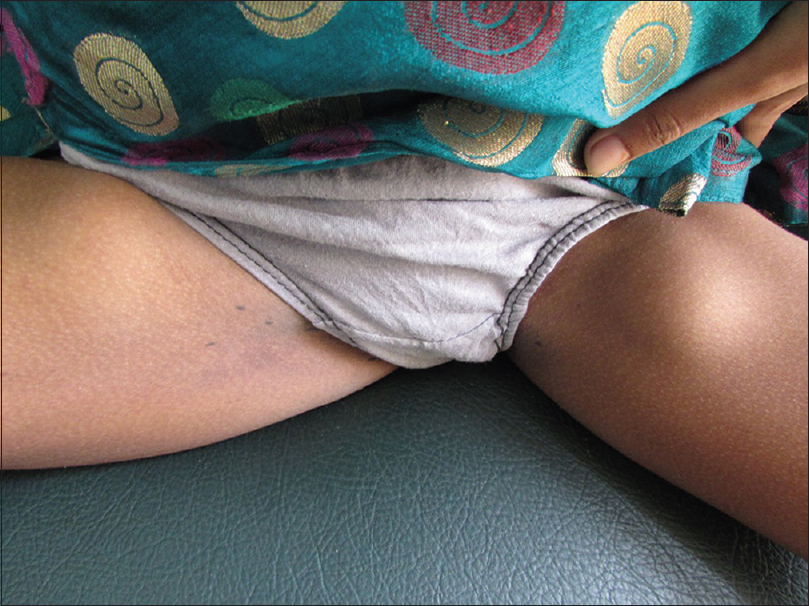 |
| Figure 4: Inguinal lymphadenopathy |
Routine blood parameters were within normal limits, except for anemia (hemoglobin 6.8 mg/dl). The patient was non-reactive for antibodies to human immunodeficiency virus 1 and 2. Chest skiagram revealed upper zone opacification and pleural effusion affecting the right lung, suggestive of a tubercular focus. Bone skiagram revealed an erosion of the medial aspect of the navicular and the medial cuneiform bone suggestive of tubercular osteomyelitis [Figure - 5]. Examination of other systems did not reveal any abnormality.
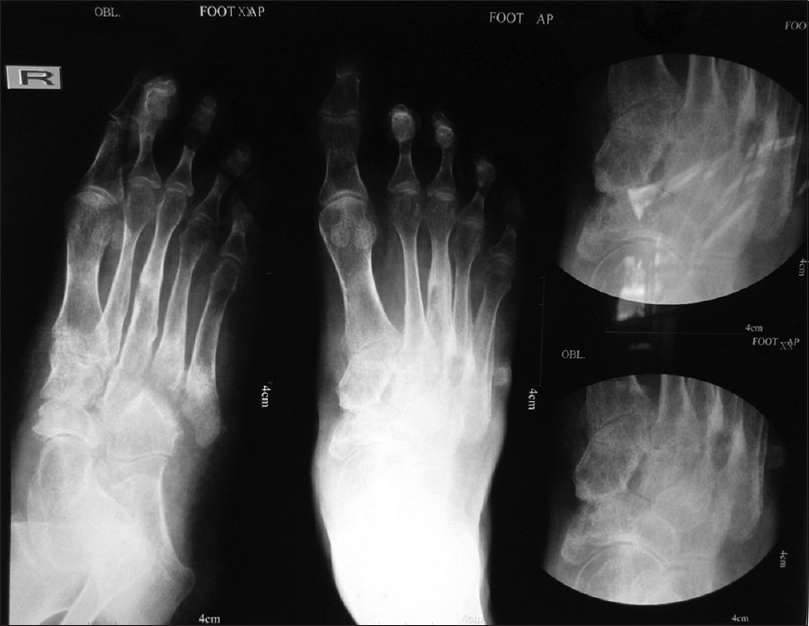 |
| Figure 5: X-ray of the right foot showing erosion of the medial aspect of the navicular and the medial cuneiform bone, suggestive of tubercular osteomyelitis |
Tuberculin skin test (Mantoux test) was positive (15 mm × 12 mm) with 5 TU purified protein derivative (PPD). Skin biopsy from the margin of abscess over the left foot showed a thickened epidermis with granulation tissue in the dermis along with a chronic inflammatory cell infiltrate and hemosiderin-laden macrophages. However, there was no granuloma, caseation necrosis or any features suggestive of a malignancy. Acid-fast bacilli were conspicuous in the discharge (Ehrlich–Ziehl–Neelsen stain) [Figure - 6]. The patient did not give consent for pus culture in Lowenstein–Jensen medium, despite repeated requests, because of economic constraints. Clinico-pathological correlation, positive tuberculin skin test and the past history of pulmonary tuberculosis led us to a final diagnosis of metastatic tubercular abscess. She was treated category 1 regimen of anti-tubercular drug therapy, after consultation with the department of pulmonary medicine, for 6 months. On follow-up after 5 months, ulcers had healed and lymphadenopathy resolved considerably [Figure - 7], [Figure - 8], [Figure - 9], thus confirming our diagnosis retrospectively. Treatment was continued for 1 more month as per protocol.
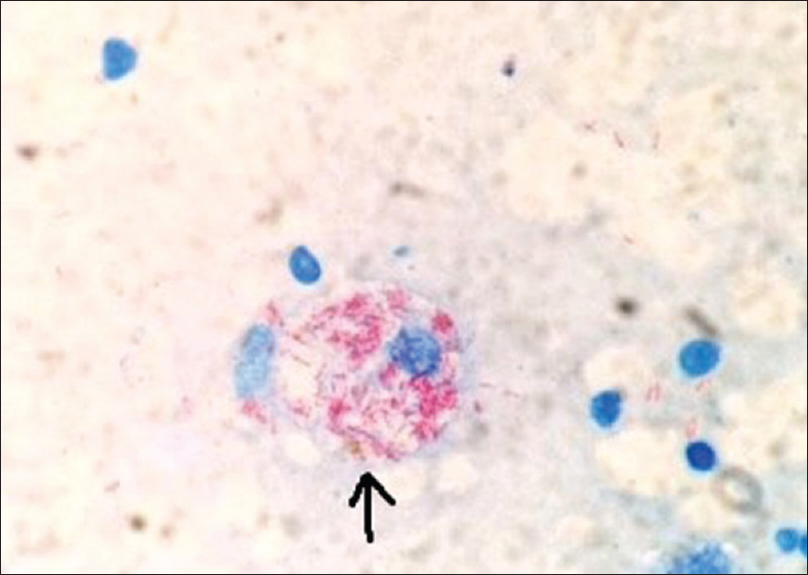 |
| Figure 6: Smear of discharge showing multiple acid-fast bacilli within a macrophage (marked by arrow) (Ehrlich.Ziehl.Neelsen, ×1000) |
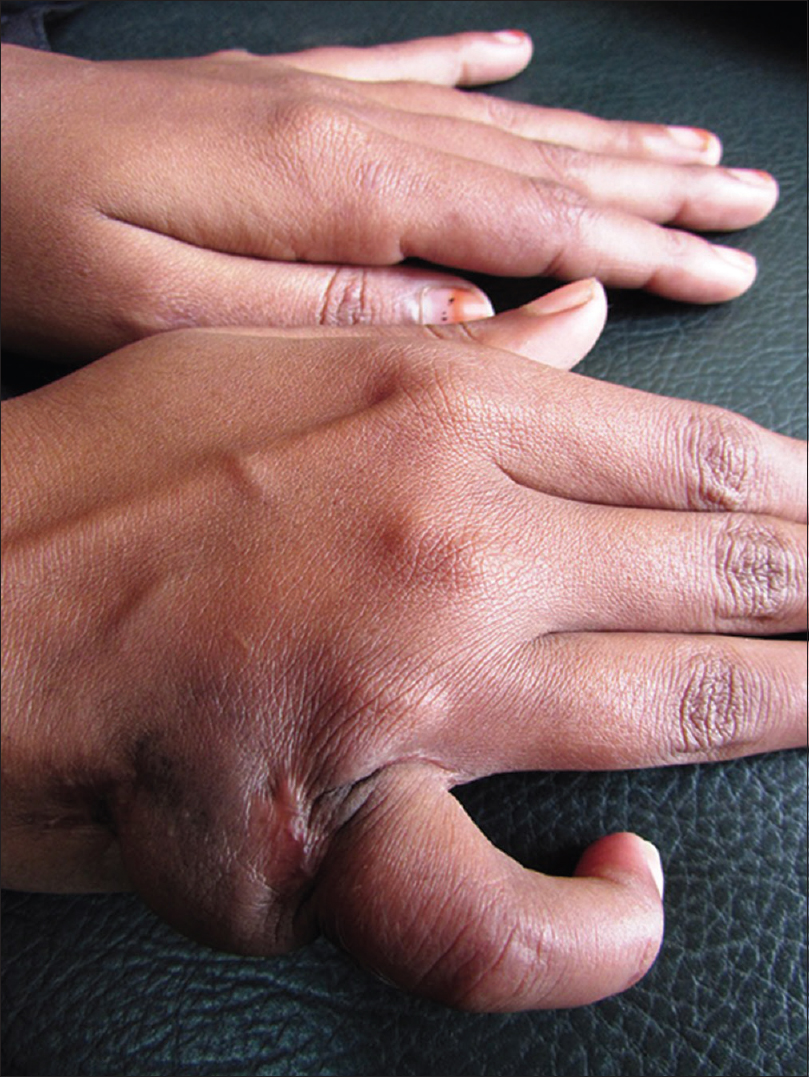 |
| Figure 7: Healed lesion on the hand |
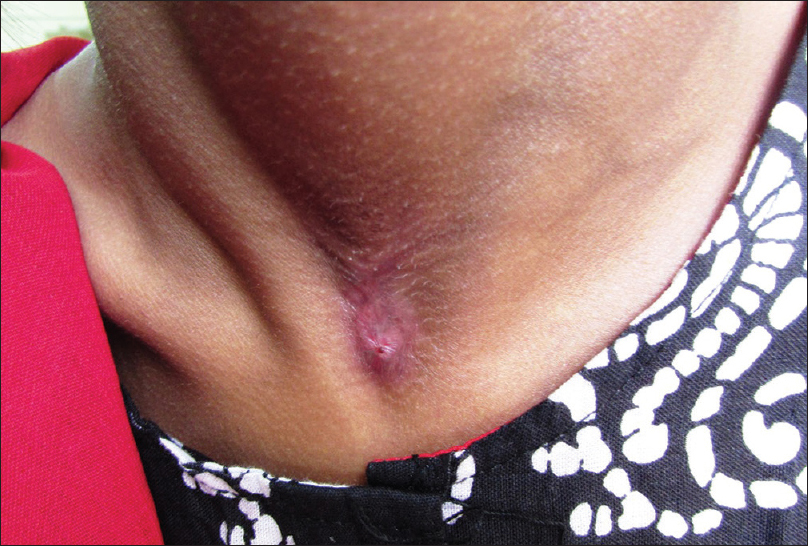 |
| Figure 8: Healed cervical lymphadenopathy |
 |
| Figure 9: Healed inguinal lymphadenopathy |
Extrapulmonary tuberculosis is seen in about one-third of all tuberculosis cases with higher preponderance in children.[1] Cutaneous tuberculosis can have a myriad of clinical presentations.[2] Lesions occurring due to exogenous inoculation of Mycobacterium tuberculosis are tubercular chancre, tuberculosis verrucosa cutis and occasionally lupus vulgaris. Endogenous cutaneous infection may lead to scrofuloderma, acute military tuberculosis, tuberculous gumma, orificial tuberculosis and lupus vulgaris. In addition, there may be cutaneous immune reactions to M. tuberculosis called tuberculids. Lupus vulgaris has been reported to be the most common form of cutaneous tuberculosis, followed by scrofuloderma, tuberculosis verrucosa cutis and cutaneous metastatic tuberculous abscess (tuberculous gumma).[3]
Metastatic tuberculous abscess occurs due to hematogenous dissemination from a primary focus during lowered immunity, resulting in single or multiple lesions. It is seen particularly in malnourished children or immunosuppressed patients, following local trauma, and in association with underlying lymphoma.[4],[5],[6] The extremities are more often affected than the trunk.[2]
Some notable differential diagnoses may be staphylococcal abscess, other mixed bacterial infections, sporotrichosis, leishmaniasis, atypical mycobacterial infections (predominantly Mycobacterium ulcerans), deep fungal infections, syphilitic gumma and leprosy. In our case, the past history of pulmonary tuberculosis, presence of tubercular focus on the chest X-ray and presence of acid-fast bacilli in Ehrlich–Ziehl–Neelsen stain helped to rule out the differentials.
The standard treatment regimen involves 2 months of 4-drugs therapy (isoniazid, rifampin, pyrazinamide and ethambutol) followed by 4 months of 2-drugs therapy (isoniazid and rifampin).[7] Various observations revealed the response of lupus vulgaris and tuberculosis verrucosa cutis to be alike with all the three regimens. The localized lesions subside completely after 4 months of therapy and the most extensive forms take 5 months to resolve. The patients with scrofuloderma responded similarly to both the triple drug regimens. The discharge, sinuses and ulcers cleared in 6 months, but the lymph nodes took longer to regress; up to 7 months in localized scrofuloderma and 9 months in more widespread scrofuloderma.[8] In our case, the lesions improved considerably after five months.
Metastatic tuberculous abscesses are usually associated with involvement of the bones of hands and feet and lymphadenopathy.[9],[10] Association has also been reported with polymyositis at different sites such as mediastinum, central nervous system and chest wall.[11],[12],[13] In our case also, lymphadenopathy (cervical and inguinal) was present, along with bony involvement of hands and feet.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. |
Darmstadt GL, Sidbury R. The skin. In: Behrman E, Kliegman RM, Jenson HB, editors. Nelson Textbook of Paediatrics. 17th ed. Philadelphia: WB Saunders Company; 2004. p. 2153-250.
[Google Scholar]
|
| 2. |
Yates VM, Rook GA. Mycobacterial infections. In: Burns T, Breathnach S, Cox N, Griffiths C, editors. Rook's Textbook of Dermatology. 7th ed. London: Blackwell Science; 2004. p. 1427-67.
[Google Scholar]
|
| 3. |
Kumar B, Rai R, Kaur I, Sahoo B, Muralidhar S, Radotra BD. Childhood cutaneous tuberculosis: A study over 25 years from northern India. Int J Dermatol 2001;40:26-32.
[Google Scholar]
|
| 4. |
Maejima H, Arai S, Ebata T, Takeda H, Kusunoki M, Katsuoka K. Tuberculous gumma associated with idiopathic thrombocytopenic purpura: Report of a Japanese female patient. J Dermatol 2007;34:86-91.
[Google Scholar]
|
| 5. |
Vidal D, Barnadas M, Pérez M, Coll P, Alomar A. Tuberculous gumma following venepuncture 2001;144:601-3.
[Google Scholar]
|
| 6. |
Kalaria VG, Kapila R, Schwartz RA. Tuberculous gumma (cutaneous metastatic tuberculous abscess) with underlying lymphoma. Cutis 2000;66:277-9.
[Google Scholar]
|
| 7. |
Bravo FG, Gotuzzo E. Cutaneous tuberculosis. Clin Dermatol 2007;25:173-80.
[Google Scholar]
|
| 8. |
Ramesh V, Misra RS, Saxena U, Mukherjee A. Comparative efficacy of drug regimens in skin tuberculosis. Clin Exp Dermatol 1991;16:106-9.
[Google Scholar]
|
| 9. |
Sezgin B, Atilganoglu U, Yigit O, Ergün SS, Cambaz N, Demirkesen C. Concomitant cutaneous metastatic tuberculous abscesses and multifocal skeletal tuberculosis. Indian J Dermatol 2008;53:149-53.
[Google Scholar]
|
| 10. |
Latsios D, Chloros D, Spyratos D, Dagdilelis L, Sichletidis L. Iliopsoas tuberculous abscess associated with cervical and axillary tuberculous lymphadenopathy. BMJ Case Rep 2011;2011. pii: Bcr0320114037.
[Google Scholar]
|
| 11. |
Silva GA, Motta RN, Carvalho Rde S, Lupi O, Azevedo MC, Ferry FR. Cutaneous tuberculous gummas in a patient with polymyositis. An Bras Dermatol 2013;88:98-101.
[Google Scholar]
|
| 12. |
Pacheco C, Silva E, Miranda J, Duarte R. Cutaneous tuberculosis as metastatic tuberculous abscess. J Bras Pneumol 2015;41:200-2.
[Google Scholar]
|
| 13. |
Agrawal A, Shanthi V, Ramakrishna BA. Tubercular cerebellar abscess in a child. J Mahatma Gandhi Inst Med Sci 2015;20:77-8.
[Google Scholar]
|
Fulltext Views
1,358
PDF downloads
1,107





