Translate this page into:
Myiasis in a neglected case of generalized erythrodermic pemphigus foliaceus
Corresponding Author:
Priyanka Patil
Department of Dermatology, OPD No. 14, 2nd Floor, OPD Building, BYL Nair Hospital, Mumbai Central, Mumbai - 400 008, Maharashtra
India
drpriyankapatil219@gmail.com
| How to cite this article: Patil P, Zambare U, Tambe S, Aderao R, Nayak C. Myiasis in a neglected case of generalized erythrodermic pemphigus foliaceus. Indian J Dermatol Venereol Leprol 2017;83:350-352 |
Sir,
Myiasis is the infestation of live vertebrates with dipterous larvae which feed on living or dead necrotic tissue of the host. Preexisting dermatological conditions and poor hygiene have been described to be predisposing factors for myiasis and it is especially common amongst neglected, dependent patients. We report an elderly man with erythrodermic pemphigus foliaceus with cutaneous and aural and ophthalmic myiasis caused by tissue obligatory larvae of the old world screw-worm fly Chrysomya bezziana.
A 65-year-old man of low socioeconomic status, neglected by his family, was brought by neighbors with redness and peeling of the skin of 3 weeks duration, foul smelling skin erosions of 2 weeks duration and worms coming out of lesions since 4 days [Figure - 1].
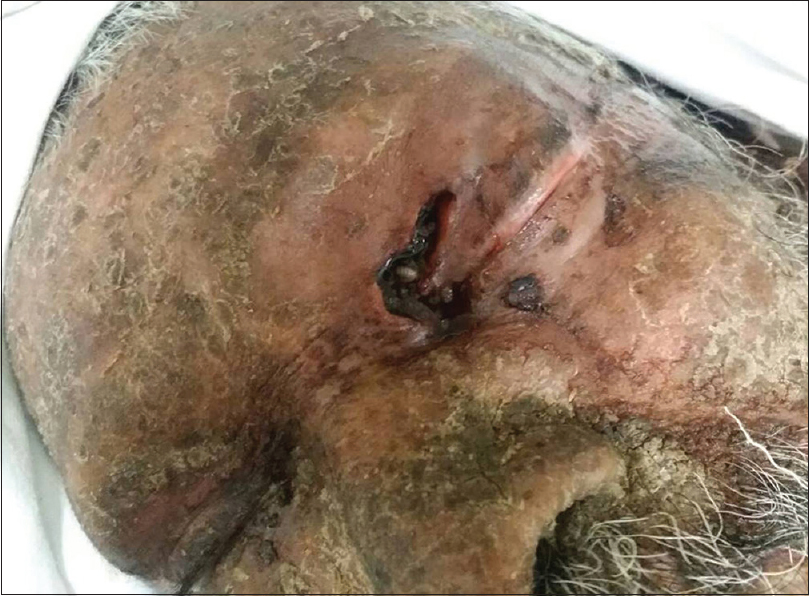 |
| Figure 1: Dermal ophthalmomyiasis, ulcer with maggots present on the left supraorbital areamaggots. |
The patient was a known case of pemphigus foliaceus and insulin-dependent diabetes mellitus since 2 years [Figure 2a] and [Figure 2b] and had received treatment including dexamethasone-cyclophosphamide pulse therapy only irregularly. He was off all treatment for over 2 months.
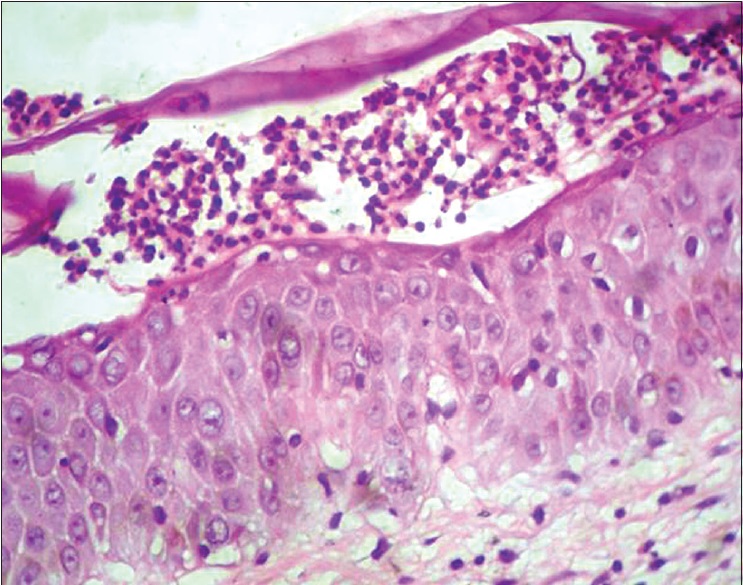 |
| Figure 2a: Biopsy from erosion revealed the absence of stratum corneum, presence of acantholytic cells and infiltrate consisting of neutrophils and eosinophils suggestive of pemphigus foliaceus (Histopathology - H and E, ×400) |
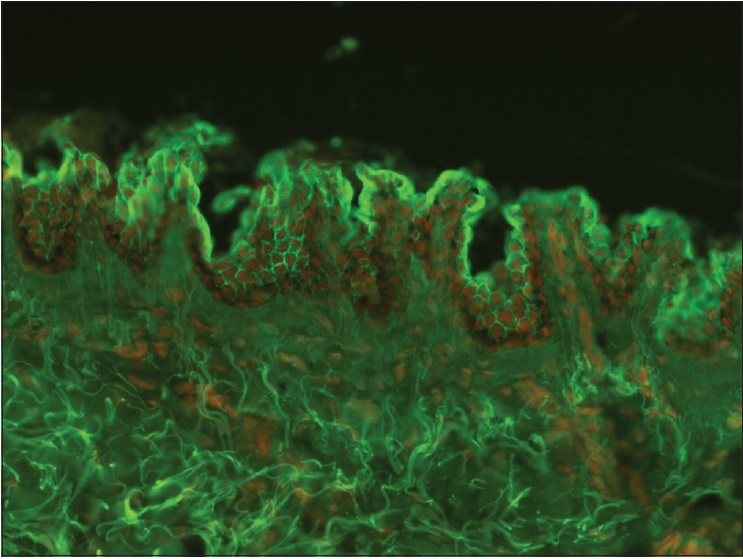 |
| Figure 2b: Direct immunofluorescence (×100) shows the presence of intercellular immunoglobulin G predominantly within the superficial layers of the epidermis suggestive of pemphigus foliaceus |
On cutaneous examination, there was erythema, scaling and erosions all over the body. Multiple ulcers measuring 1 to 4 cm in diameter with yellowish necrotic slough and maggots were present on lower abdomen, right side of the neck [Figure - 3], left supraorbital area, right hand and right thigh. The larvae extracted were whitish, 12–15 mm in size and were identified as larvae of C. bezziana species by the patterns of anterior and posterior spiracles. Patient's right ear lobe was destroyed by larvae and gangrenous changes were present on the right hand. He subsequently developed furuncular myiasis with the formation of new ulcers. Systemic examination revealed no abnormalities and patient was hemodynamically stable.
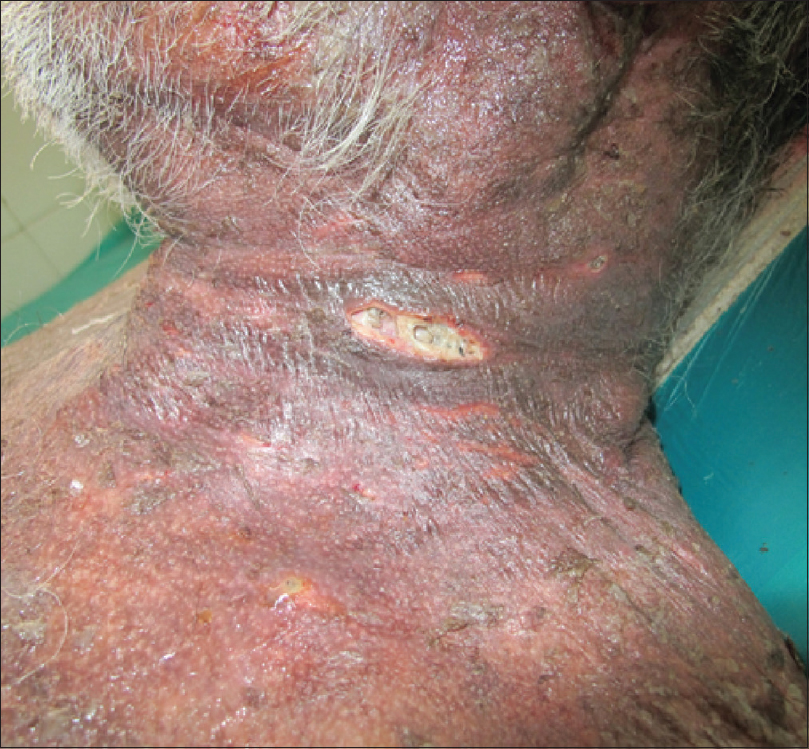 |
| Figure 3: Ulcer with maggots present on neck |
Laboratory investigations revealed mild anemia (hemoglobin - 10.2g/dl), neutrophilic leukocytosis (white blood cell count – 17,200/mm [3]), low serum total proteins (4.2 g/dl), raised liver enzymes (serum glutamic oxaloacetic transaminase/serum glutamic pyruvic transaminase - 53/43 IU) and low serum sodium levels. Urine microscopy showed the presence of 8-10 red blood cells, 15-20 pus cells and cocci. Urine culture revealed growth of Enterococcus faecalis sensitive to piperacillin-tazobactam. Arteriovenous Doppler study revealed thrombosis in the distal end of the right ulnar artery with collateral formation. Orbital computed tomography study showed periorbital soft tissue swelling. Local ultrasonography showed serpiginous hypoechogenic tracts surrounding lesions. Fundus examination was normal. Pus culture from the ulcers grew Staphylococcus aureus and Proteus mirabilis sensitive to piperacillin-tazobactam.
Gauze pieces soaked with turpentine oil were kept on the ulcers daily followed by mechanical debridement and manual removal of maggots. Patient was administered intravenous fluids, antibiotics, oral hematinics and povidone-iodine gauze dressing and emollient application on scaly areas of the body with moxifloxacin eye drops. Single dose of oral ivermectin (12 mg) was given. As patient had a secondary bacterial infection, open wounds, raised white blood cell count and gangrene of the right hand, dexamethasone-cyclophosphamide pulse therapy was deferred. Hand gangrene and thrombosis of ulnar artery were managed conservatively. The wound started healing, but unfortunately, the patient went into sudden cardiac arrest and succumbed within 10 days of admission to hospital.
Myiasis (from the Greek “myia,” meaning fly) is defined as infestation by dipterous larvae which feed on the hosts' dead or living tissues. C. bezziana infestation differs from usual maggot infestations because it buries deep into the tissue and remains attached with cuticular spines, so it is both biontophage (invade living tissue) and necrobiontophage (invade necrotic tissue) as seen in our patient, forming furuncle- like lesions with rapid development of new ulcers and also invading the orbital tissue.
Diagnosis of cutaneous myiasis is based on the morphology and identification of maggots. Deeper tissue invasion of maggots can be visualized by dermoscopy, skin ultrasound or computed tomography scan for orbital invasion.[1]
Treatment comprises manual removal of visible larvae, debridement of necrotic tissue, irrigation with antiseptic solution and daily dressing.[2] In furuncular myiasis, larval removal can be done by hydraulic expulsion technique.[3]
Various agents used to suffocate and paralyze the larvae are chloroform in olive oil, turpentine oil, petroleum jelly, liquid paraffin, beeswax, resins, glue, adhesive tape, nail polish, chewing gum, pork fat, polymyxin B sulfate ointment, ether, 1% lidocaine and pilocarpine. Essential oil of betel leaf (Piper betle) is an effective larvicidal for C. bezziana. Broad-spectrum antibiotics are implicated to prevent secondary infection. Antiparasitic drugs such as topical permethrin and semisynthetic macrocyclic lactone such as ivermectin can be used in a dose of 200 μg/kg. Other macrocyclic lactones used are moxidectin, eprinomectin and doramectin.
Myiasis has been reported secondary to dermatological conditions such as giant squamous cell carcinoma,[4] basal cell carcinoma, cutaneous melanoblastoma, Bowen's disease, following radiotherapy of squamous cell carcinoma, large perigenital seborrheic keratosis, leprosy, psoriasis, onychomycosis and herpes zoster virus infection,[5] but we were unable to find its occurrence in erythrodermic pemphigus foliaceus in literature.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient has given his consent for his images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Acknowledgment
Department of Microbiology, Topiwala National Medical College and BYL Nair Hospital, Mumbai, Maharashtra, India. Dr. Yashant Aswani, Senior Resident, Department of Radiology, Topiwala National Medical College and BYL Nair Hospital, Mumbai, Maharashtra, India.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. | Bakos RM, Bakos L. Dermoscopic diagnosis of furuncular myiasis. Arch Dermatol 2007;143:123-4. [Google Scholar] |
| 2. | Sesterhenn AM, Pfützner W, Braulke DM, Wiegand S, Werner JA, Taubert A. Cutaneous manifestation of myiasis in malignant wounds of the head and neck. Eur J Dermatol 2009;19:64-8. [Google Scholar] |
| 3. | Dybbro E, Fordyce P, Ponte M, Arron ST. Hydraulic expulsion of tumbu fly larvae. JAMA Dermatol 2014;150:791-2. [Google Scholar] |
| 4. | Yaghoobi R, Bagherani N. Chrysomya bezziana infestation in a neglected squamous cell carcinoma on the face. Indian J Dermatol Venereol Leprol 2009;75:81-2. [Google Scholar] |
| 5. | Murthy SC, Udagari MM. Myiasis complicating herpes zoster in an immuno compromised patient. Indian J Dermatol Venereol Leprol 2003;69:194. [Google Scholar] |
Fulltext Views
2,051
PDF downloads
600





