Translate this page into:
Neurocutaneous melanosis is not always a benign disease
2 Department of Pediatrics, Genetic Unit, Faculty of Medicine, Mansoura University, Mansoura, Egypt
3 Department of Biomedical Experimental and Clinical Sciences “Mario Serio,” University of Florence, Florence, Italy
4 Department of Pathology, Faculty of Medicine, Mansoura University, Mansoura, Egypt
Correspondence Address:
Yahya Wahba
Department of Pediatrics, Genetic Unit, Faculty of Medicine, Mansoura University, Mansoura, 35516
Egypt
| How to cite this article: El Mabood SA, Wahba Y, Yahia S, Sarhan MM, Mansour AK, Provenzano A, Elmahdi HS, Darwish A. Neurocutaneous melanosis is not always a benign disease. Indian J Dermatol Venereol Leprol 2020;86:335 |
Sir,
Neurocutaneous melanosis is a rare form of neurocutaneous syndrome characterized by the presence of congenital melanocytic nevi in the skin as well as proliferation of the melanocytes in the leptomeninges and the brain.[1] Patients with neurocutaneous melanosis are at risk for malignant transformation of cutaneous and leptomeningeal melanomas. The lifetime risk for development of melanoma in patients with congenital melanocytic nevi varies, being higher with those with large congenital melanocytic nevi and multiple satellites (>20 satellites).[2] In large congenital melanocytic nevi> 40 cm projected adult size, it has been estimated to be 10%–15%.[3] In 40%–60% of patients with neurocutaneous melanosis, malignant transformation of leptomeningeal tumors occurs with severe neurological insults.[4]
A 22-month-old girl was brought to the outpatient clinic of Mansoura University Children's Hospital, Egypt, with multiple hairy pigmented nevi since birth which progressively increased in size and number over time. Her back was the most involved site being covered completely with a large nevus measuring 22 cm in its largest diameter [Figure - 1]a together with multiple smaller satellites. It was first observed in the extremities and progressed to involve back and face (about 40 satellites) [Figure - 1]b. She had skin phototype III, no organomegaly, and no palpable regional lymph nodes. Skin biopsies taken from the large nevus showed mitotic index 0/mm[2], Breslow thickness 0.8 mm, and Clark level III. Abdominal ultrasonography and chest X-ray showed normal findings. No brain or whole-spine magnetic resonance imaging (MRI) screening was done during early infancy.
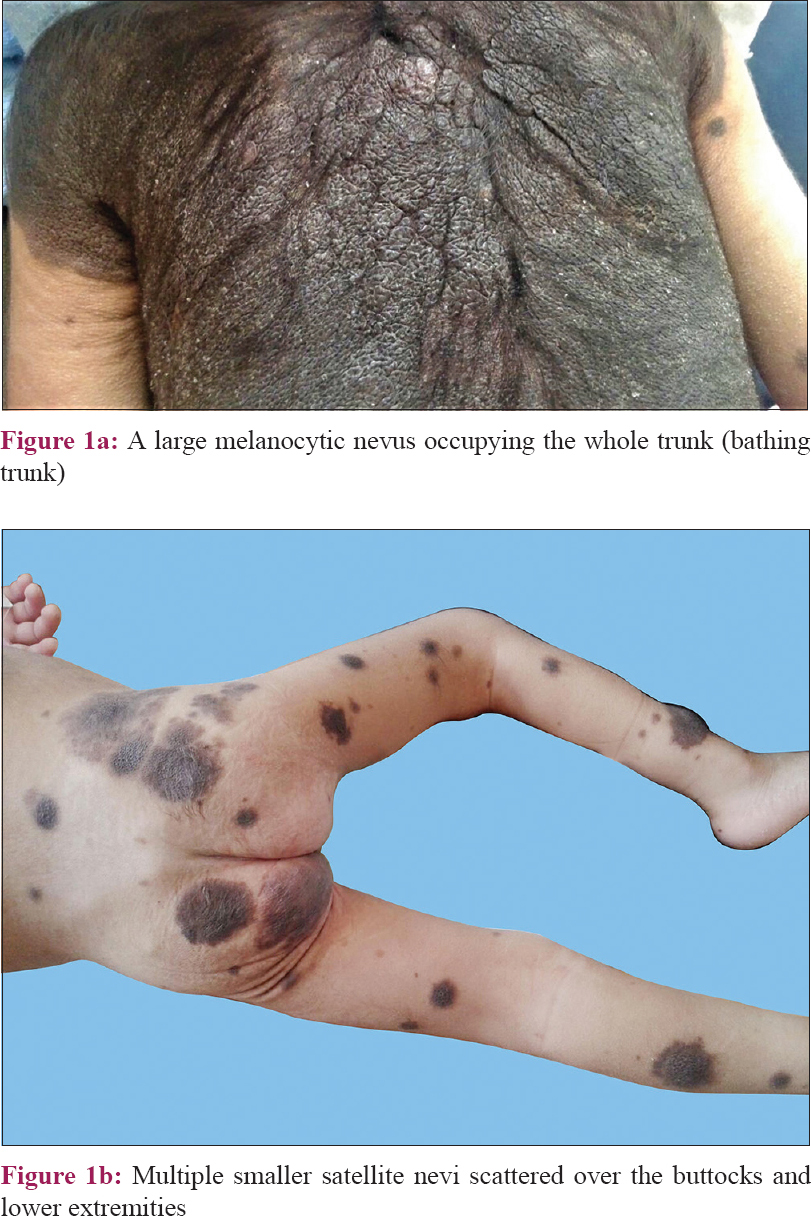 |
| Figure 1: |
Her developmental milestones were delayed as compared to her healthy peers. At the age of 18 months, she developed repeated seizures with right medial squint followed by coma. She was urgently referred to Mansoura University Children's Hospital, Egypt, where computed tomography scan and MRI brain were done showing a right cerebellar space-occupying lesion measuring 4.6 × 3.5 cm compressing the brain stem and the fourth ventricle with subsequent hydrocephalus [Figure - 2]a. MRI of the spinal cord was done revealing extensive spinal circumferential dural thickening compressing the thecal sac and the cord at all spinal levels with post contrast enhancement [Figure - 2]b. Also, cytochemical analysis and cerebrospinal fluid (CSF) culture were done which revealed normal glucose (76 mg/dL), increased protein (201 mg/dL), white blood cells (3 × 10[3]/μL), and negative CSF culture.
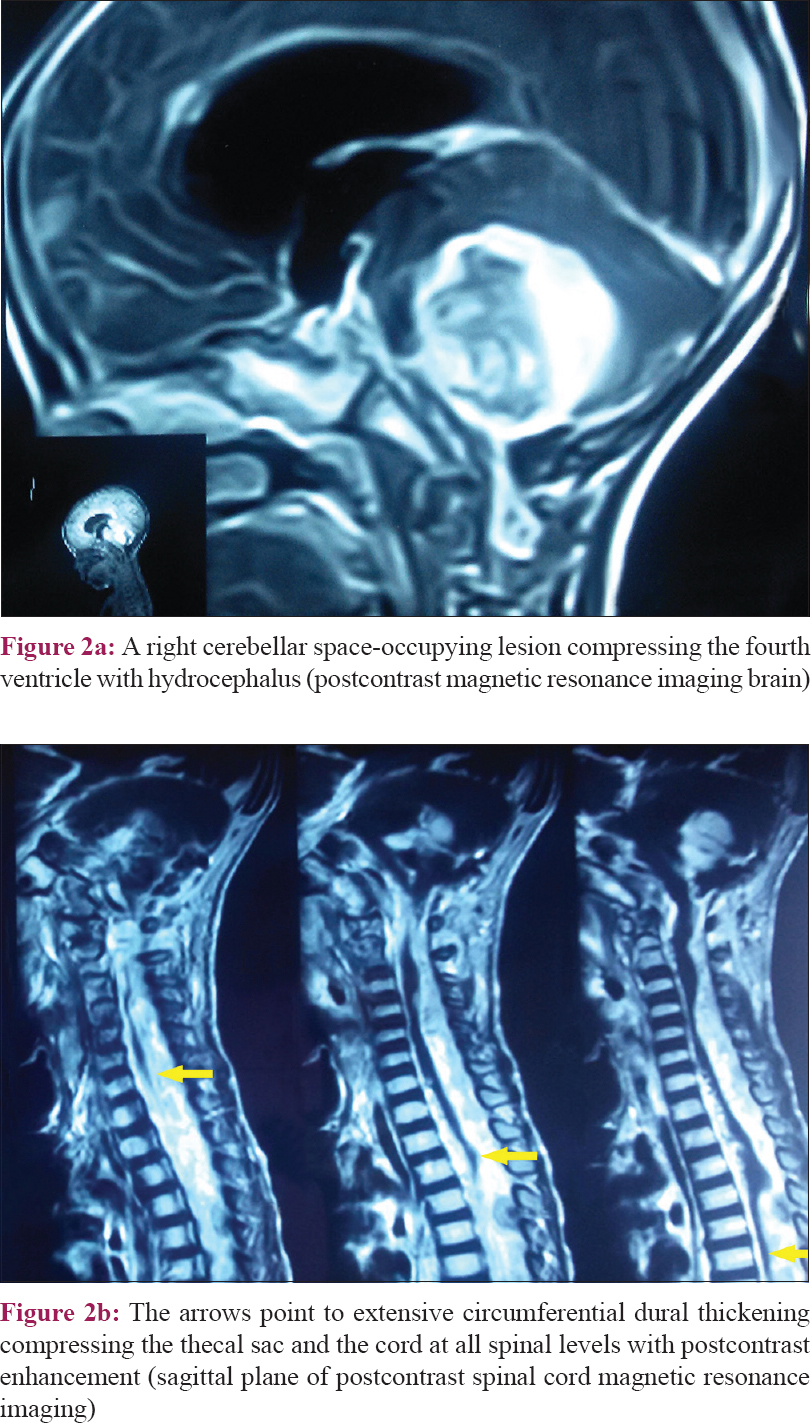 |
| Figure 2: |
As the tumor was extensively infiltrating the brain stem, it was not amenable to complete surgical resection and only subtotal excision was done by neurosurgical staff members of Mansoura University Hospital, Egypt. Pathological examination with immunohistochemical staining showed malignant round and spindle cells which focally contained melanin. A melanocyte marker, melan A, stained both small and large cells [Figure - 3]a and [Figure - 3]b. According to the clinical, histopathological, and radiological findings, the diagnosis was malignant melanoma, as a part of neurocutaneous melanosis. NRAS gene sequencing was done in collaboration with medical genetics section of Florence University, Italy, being the most frequently implicated gene, revealing a wild variant.[5]
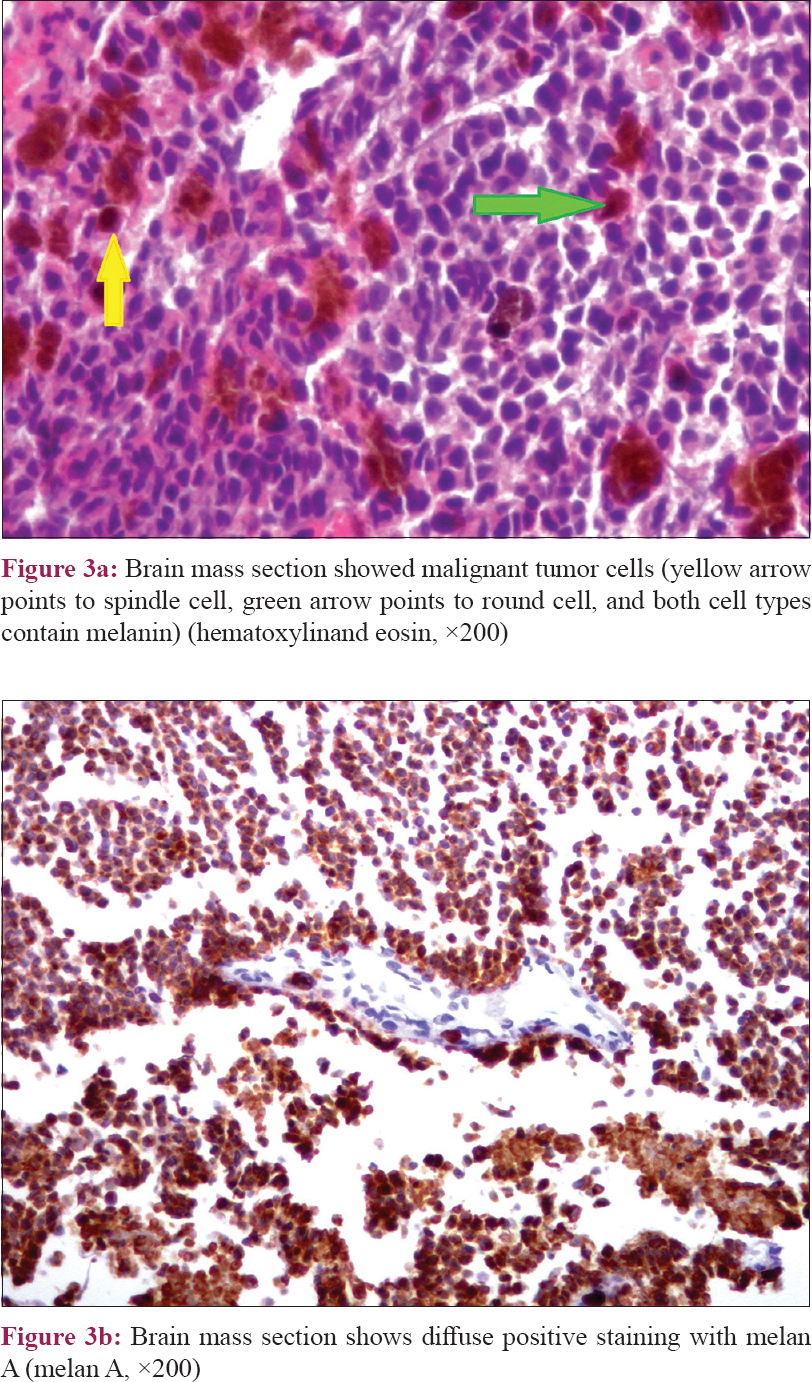 |
| Figure 3: |
The patient was refused by the radiotherapist for irradiation because of her young age and its little value for her condition. Despite its poor efficacy, she was planned to receive palliative chemotherapy aiming to prolong the survival and improve the neurological condition. She received only one cycle of 4 weekly based chemotherapy cycles consisting of intravenous vincristine 1.5 mg/m[2] on day 1, intravenous dacarbazine 200 mg/m[2] for 5 days on days 1–5, and cyclophosphamide 750 mg/m[2] continuous intravenous infusion for 2 days on days 1 and 2.[6] With regard to seizures, levetiracetam (40 mg/kg/day bid), carbamazepine (20 mg/kg/day bid), and topiramate (started orally at 1 mg/kg/day and increased up to 6 mg/kg/day bid) were used to control seizures. Unfortunately, 2 days after starting the first cycle, the patient developed progressive deterioration of her consciousness level with irregular respiration and died. She died 4 months after the diagnosis of brain melanoma.
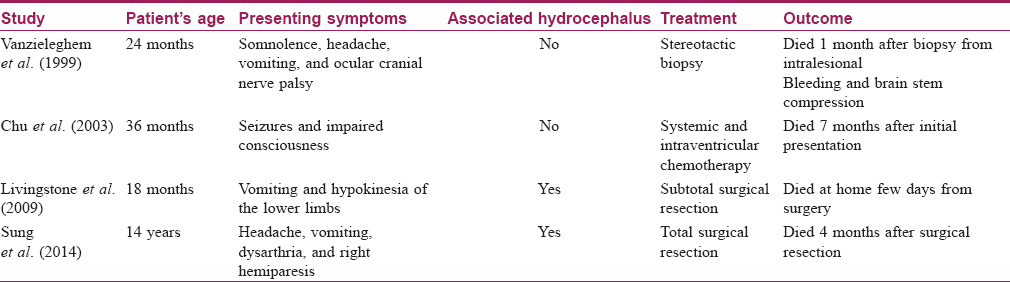
The course of neurocutaneous melanosis is quite variable; some cases have a purely benign course with normal life expectancy, while others suffer from severe neurological complications with aggressive progression and fatal outcome.[5] Neurocutaneous melanosis can affect the cerebellum, amygdala, cerebrum, medulla, pons, and spinal cord.[7],[8] There are two age peaks for presentation of complications from neurocutaneous melanosis. The first peak (most common peak) occurs during the first 3 years of life and presents with increased intracranial pressure manifestations, while the other peak occurs during the second to third decades of life in the form of headache and neuropsychiatric symptoms.[9],[10] The risk factors for neurocutaneous melanosis are the presence of giant congenital melanocytic nevi, multiple satellite nevi, male sex, and neck, head, or posterior midline location.[10],[11],[12],[13] [Table - 1] shows a summary of some reported similar cases of brain melanoma associated with NCM.[6],[14],[15],[16]
Our case showed all neurological sequences of the disease starting with developmental delay progressing to repeated seizures and hydrocephalus because of diffuse circumferential thickening of the meninges secondary to melanin deposition. Unfortunately, she had a giant trunk nevus with multiple satellites putting her at a higher risk of development of brain melanoma.[17]
Brain melanoma has very poor prognosis because of disappointing results of conventional therapy. Radical surgical resection is often impossible as the tumor commonly infiltrates the adjacent brain structures massively. Also, the tumor responds poorly to chemotherapy and radiotherapy.[6] In 2014, Küsters-Vandevelde et al. reported a trial of treatment of brain melanoma of neurocutaneous melanosis syndrome using a novel target therapy (MEK162; mitogen- activated protein kinase, MEK inhibitor) against the somatic mutation of NRAS.[18] More recent efforts are being carried out for finding targeted therapy in the future as the conventional therapeutic options have dismal results. In 2016, Patel et al. postulated that insulin-like growth factor binding proteins' downregulation enhances the proliferation and viability of neurocutaneous melanosis cells. They assumed that inhibition of insulin-like growth factor 1 receptor may lead to loss of viability of neurocutaneous melanosis which may represent in the future a therapeutic target for this rare fatal disease.[19] Our patient led a downhill course and died shortly after appearance of neurological deficits in spite of surgery and palliative chemotherapy.
We concluded that though neurocutaneous melanosis is a well-known disease, it is important for dermatologists and pediatricians to remember the risk of a potentially fatal association of giant congenital melanocytic nevi. We recommend brain and whole-spine MRI screening for infants with giant congenital melanocytic nevi, multiple medium sized congenital melanocytic nevi, or multiple satellite nevi (especially> 20 satellite nevi). Also, apart from imaging, these infants need to be followed up with serial head circumference measurement, neurological examinations, and development assessment, to pick up any abnormality early and also for repeat imaging if required. The MRI screening should be gadolinium-enhanced and should ideally be done during the first 6 months of life before myelination, as the latter may obscure the evidence of melanosis.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the legal guardian has given consent for the child's images and other clinical information to be reported in the journal. The guardian understands that the child's name and initials will not be published and due efforts will be made to conceal identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. |
Sebag G, Dubois J, Pfister P, Brunelle F, St-Rose C. Neurocutaneous melanosis and temporal lobe tumor in a child: MR study. AJNR Am J Neuroradiol 1991;12:699-700.
[Google Scholar]
|
| 2. |
Marghoob AA, Dusza S, Oliveria S, Halpern AC. Number of satellite nevi as a correlate for neurocutaneous melanocytosis in patients with large congenital melanocytic nevi. Arch Dermatol 2004;140:171-5.
[Google Scholar]
|
| 3. |
Kinsler VA, Chong WK, Aylett SE, Atherton DJ. Complications of congenital melanocytic naevi in children: Analysis of 16 years' experience and clinical practice. Br J Dermatol 2008;159:907-14.
[Google Scholar]
|
| 4. |
Ginat DT, Meyers SP. Intracranial lesions with high signal intensity on T1-weighted MR images: Differential diagnosis. Radiographics 2012;32:499-516.
[Google Scholar]
|
| 5. |
Charbel C, Fontaine RH, Malouf GG, Picard A, Kadlub N, El-Murr N, et al. NRAS mutation is the sole recurrent somatic mutation in large congenital melanocytic nevi. J Invest Dermatol 2014;134:1067-74.
[Google Scholar]
|
| 6. |
Chu WC, Lee V, Chan YL, Shing MM, Chik KW, Li CK, et al. Neurocutaneous melanomatosis with a rapidly deteriorating course. AJNR Am J Neuroradiol 2003;24:287-90.
[Google Scholar]
|
| 7. |
Barkovich AJ, Frieden IJ, Williams ML. MR of neurocutaneous melanosis. AJNR Am J Neuroradiol 1994;15:859-67.
[Google Scholar]
|
| 8. |
Foster RD, Williams ML, Barkovich AJ, Hoffman WY, Mathes SJ, Frieden IJ. Giant congenital melanocytic nevi: The significance of neurocutaneous melanosis in neurologically asymptomatic children. Plast Reconstr Surg 2001;107:933-41.
[Google Scholar]
|
| 9. |
Kadonaga JN, Frieden IJ. Neurocutaneous melanosis: Definition and review of the literature. J Am Acad Dermatol 1991;24:747-55.
[Google Scholar]
|
| 10. |
Lawrence CM. Treatment options for giant congenital naevi. Clin Exp Dermatol 2000;25:7-11.
[Google Scholar]
|
| 11. |
Makin GW, Eden OB, Lashford LS, Moppett J, Gerrard MP, Davies HA, et al. Leptomeningeal melanoma in childhood. Cancer 1999;86:878-86.
[Google Scholar]
|
| 12. |
Lovett A, Maari C, Decarie JC, Marcoux D, McCuaig C, Hatami A, et al. Large congenital melanocytic nevi and neurocutaneous melanocytosis: One pediatric center's experience. J Am Acad Dermatol 2009;61:766-74.
[Google Scholar]
|
| 13. |
Hale EK, Stein J, Ben-Porat L, Panageas KS, Eichenbaum MS, Marghoob AA, et al. Association of melanoma and neurocutaneous melanocytosis with large congenital melanocytic naevi – Results from the NYU-LCMN registry. Br J Dermatol 2005;152:512-7.
[Google Scholar]
|
| 14. |
Vanzieleghem BD, Lemmerling MM, Van Coster RN. Neurocutaneous melanosis presenting with intracranial amelanotic melanoma. AJNR Am J Neuroradiol 1999;20:457-60.
[Google Scholar]
|
| 15. |
Livingstone E, Claviez A, Spengler D, Barth H, Stark AM, Hugo HH, et al. Neurocutaneous melanosis: A fatal disease in early childhood. J Clin Oncol 2009;27:2290-1.
[Google Scholar]
|
| 16. |
Sung KS, Song YJ. Neurocutaneous melanosis in association with Dandy-Walker complex with extensive intracerebral and spinal cord involvement. J Korean Neurosurg Soc 2014;56:61-5.
[Google Scholar]
|
| 17. |
Krengel S, Hauschild A, Schäfer T. Melanoma risk in congenital melanocytic naevi: A systematic review. Br J Dermatol 2006;155:1-8.
[Google Scholar]
|
| 18. |
Küsters-Vandevelde HV, Willemsen AE, Groenen PJ, Küsters B, Lammens M, Wesseling P, et al. Experimental treatment of NRAS-mutated neurocutaneous melanocytosis with MEK162, a MEK-inhibitor. Acta Neuropathol Commun 2014;2:41.
[Google Scholar]
|
| 19. |
Patel J, Salgado CM, Múgica MR, Basu D. Insulin-like growth factor 1 receptor signaling via Akt: A general therapeutic target in neurocutaneous melanocytosis? Neuro Oncol 2016;18:142-3.
[Google Scholar]
|
Fulltext Views
4,261
PDF downloads
1,971





