Translate this page into:
Onset of psoriasis triggered by Mantoux test
2 Department of Dermatology, University College of Medical Sciences, GTB Hospital (University of Delhi), Delhi, India
3 Department of Pathology, Chacha Nehru Bal Chikitsalaya, Delhi, India
Correspondence Address:
Archana Singal
B-14, Law Apartments, Karkardooma, Delhi - 110 092
India
| How to cite this article: Khanna D, Singal A, Gupta R. Onset of psoriasis triggered by Mantoux test . Indian J Dermatol Venereol Leprol 2014;80:451-454 |
Sir,
A 16-year-old girl presented with erythematous, scaly and mildly pruritic lesions over the left forearm for one week duration. She had undergone Mantoux testing with purified protein derivative (PPD) on the forearm two weeks previously, during investigations for pyrexia of unknown origin. The tuberculin response at that time had been recorded as negative. Subsequently, a scaly lesion appeared, initially over the site of tuberculin testing and later over other areas on the forearm. She had no personal or family history of psoriasis. Physical examination revealed an erythematous plaque measuring about 3 × 3 cm with silvery-white scales, overlying the Mantoux test site [Figure - 1] and some smaller lesions confined to the left forearm showing a positive Grattage test and Auspitz sign. Scalp, palms, soles and nails were not involved. Hematological and biochemical profiles including the results of erythrocyte sedimentation rate, serum calcium, anti-streptolysin ′O′ (ASLO) and pharyngeal culture revealed no abnormality. Chest radiography and ultrasonography of the abdomen also did not reveal any focus of tuberculosis. Histopathological examination showed hyperkeratosis, elongated rete ridges with suprapapillary thinning, absent granular layer, microabscess formation and mild chronic inflammatory infiltrate composed of mononuclear cells in upper dermis corroborating the clinical diagnosis of psoriasis [Figure - 2]. In the following weeks, multiple lesions of plaque psoriasis continued to appear over other parts of the body for eight and half months, after which the patient was lost to follow up [Figure - 3].
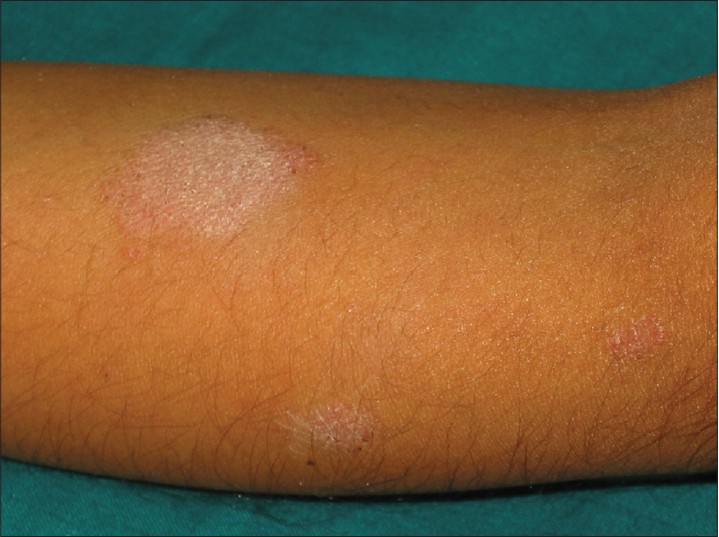 |
| Figure 1: Erythematous scaly plaque over the flexor aspect of forearm overlying Mantoux test site (note smaller lesions in vicinity) |
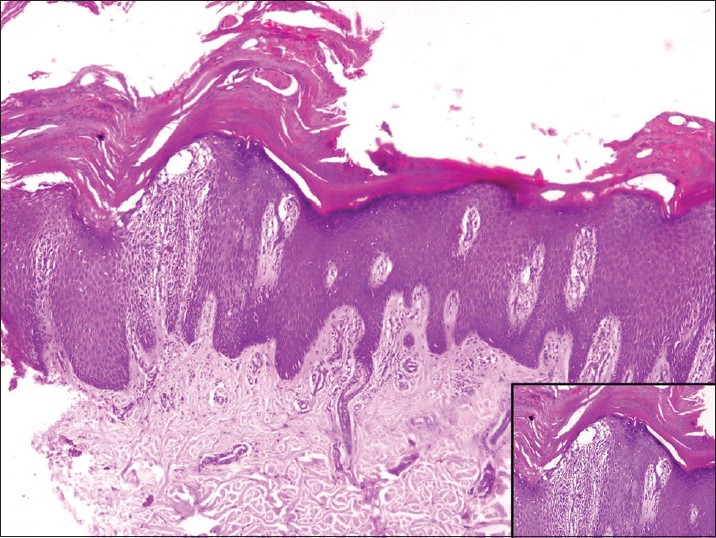 |
| Figure 2: Hyperkeratosis with parakeratosis and microabscess formation, elongated rete ridges with suprapapillary thinning, absent granular layer, marked spongiosis and mild chronic inflammation in the dermis (H and E, ×100). Inset shows parakeratosis and spongiosis in higher magnification (H and E, ×400) |
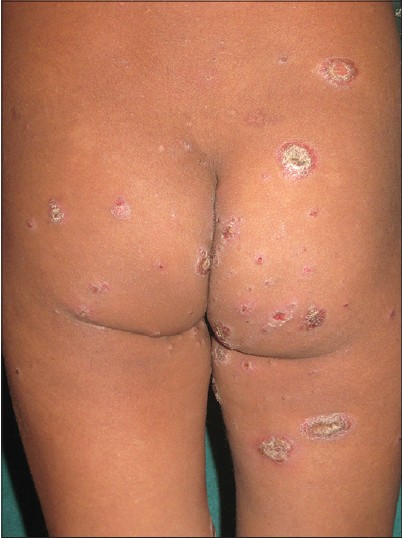 |
| Figure 3: Classical psoriatic plaques over the back and buttocks |
A dysregulated innate immune system leading to antigen driven T-cell recruitment, activation and subsequent effector cytokine release, has been hypothesized to result in inflammation and epidermal hyperproliferation in psoriasis. The role of an as yet unidentified antigen as being central to pathogenesis of psoriasis is being explored. Koebner phenomenon refers to the induction of lesions morphologically similar to a pre-existing disease by physical trauma or injury in a previously unaffected site. [1] Such trauma can be either non-infective physical insults like pressure, acupuncture, friction, burns or other cutaneous insults like tattooing, sunburn, bacterial, viral or fungal infections, and intradermal skin tests. Boyd and Neldner suggested that true koebnerization should be reproducible by a variety of insults and not just due to contact with external infective or allergic elements. [1]
Mantoux testing involves administering a standard preparation of PPD intradermally that results in a delayed type hypersensitivity reaction represented by local skin induration. Tuberculin testing like other intradermal tests can induce Koebner phenomenon simply due to the epidermal rupture it causes. Fadden et al. did not note the development of skin lesions following intradermal antigen tests in any of their Koebner-negative patients and inferred that intradermal antigen tests are no more effective in inducing the Koebner phenomenon than injury alone. [2] However, Dogan et al. found development of psoriasis lesions at one or more injection sites in five of their Koebner-negative patients. Of these, psoriasis lesions were seen at the site of injection of antigen, but not at the saline injected site in one patient, making them state that intradermal antigen tests were more effective in inducing the Koebner phenomenon than injury alone. [3] In addition, individual specific sensitivity to different antigens may be responsible for activating the cellular immune response and the disease in some patients.
A relapse of pre-existing psoriasis following Mantoux test has been previously reported. [4] We were unable to find any previous reports of the development of psoriasis in a previously unaffected patient following tuberculin testing. Koca et al. reported occurrence of guttate psoriasis-like lesions following Bacillus Calmette-Guerin (BCG) vaccination in a previously unaffected boy with a family history of psoriasis. [5] Takayama et al. demonstrated an increase in the expression of activated phospho-Stat-3 in the epidermal keratinocytes of the lesional skin in a girl developing psoriasis following BCG vaccination. [6]
The mechanism of induction of psoriasis in a previously unaffected individual following intradermal tuberculin testing needs to be addressed [Figure - 4]. Some authors have taken the extreme view that the initial presentation of psoriasis is always the result of a Koebner reaction with the unmasking of dormant psoriatic skin by some form of trauma. [7] The possibility of intradermal testing providing optimum epidermal trauma for initiation of Koebner phenomenon in a predisposed individual leading to unmasking of latent psoriasis cannot be commented upon in our patient as she had not undergone any other intradermal test. Alternatively, mycobacterial heat-shock protein may present the necessary antigenic stimulus to activate the immune response towards production of psoriatic skin lesions [Figure - 4]. A high titer of antibody to mycobacterial heat-shock protein hsp65 and other dominant mycobacterial antigens correlating with disease activity has previously been demonstrated in psoriasis patients. [8] An additive effect of both epidermal trauma and exposure to mycobacterial antigens cannot be ruled out. An exposure to mycobacterial antigens through the percutaneous route may perhaps be more effective in triggering psoriasis than through other routes.
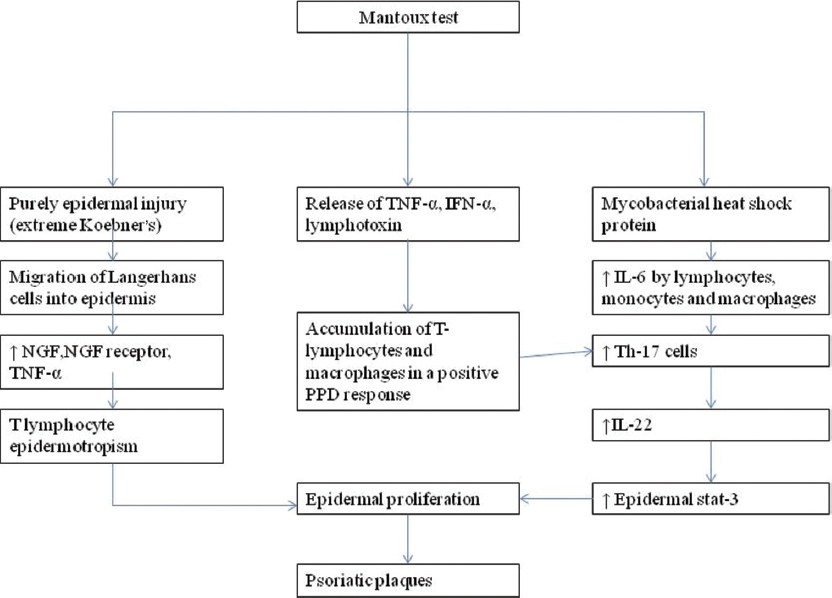 |
| Figure 4: Hypothetical mechanisms of induction of psoriasis following Mantoux testing |
| 1. |
Boyd AS, Neldner KH. The isomorphic response of koebner. Int J Dermatol 1990;29:401-10.
[Google Scholar]
|
| 2. |
McFadden JP, Powles AW, Baker BS, Valdimarsson H, Fry L. Intradermal antigen tests in psoriasis. Acta Derm Venereol 1990;70:262-4.
[Google Scholar]
|
| 3. |
Dogan B, Harmanyeri Y. Intradermal antigen tests and Koebner phenomenon in psoriasis. Int J Dermatol 1997;36:263-5.
[Google Scholar]
|
| 4. |
Piaserico S, Dan G, Peserico A. Koebner phenomenon in psoriasis induced by Mantoux test. J Eur Acad Dermatol Venereol 2009;23:1103-5.
[Google Scholar]
|
| 5. |
Koca R, Altinyazar HC, Numanoglu G, Unalacak M. Guttate psoriasis like lesions following BCG immunotherapy. J Trop Pediatr 2004;50:178-9.
[Google Scholar]
|
| 6. |
Takayama K, Satoh T, Hayashi M, Yokozeki H. Psoriatic skin lesions induced by BCG vaccination. Acta Derm Venereol 2008;88:621-2.
[Google Scholar]
|
| 7. |
Monacelli M. Koebner reaction in psoriasis. In: Farber EM, Cox AJ, editors. Psoriasis: Proceedings of the International Symposium. Vol. 99. Stanford: Stanford University press; 1971. p. 104-11.
[Google Scholar]
|
| 8. |
Rambukkana A, Das PK, Witkamp L, Yong S, Meinardi MM, Bos JD. Antibodies to mycobacterial 65-kDa heat shock protein and other immunodominantantigens in patients with psoriasis. J Invest Dermatol 1993;100:87-92.
[Google Scholar]
|
Fulltext Views
2,148
PDF downloads
1,155





