Translate this page into:
Oral isotretinoin is as effective as a combination of oral isotretinoin and topical anti-acne agents in nodulocystic acne
Correspondence Address:
Rajeev Dhir
Senior Advisor (Dermatology), INHS Asvini, Colaba, Mumbai - 400 005
India
| How to cite this article: Dhir R, Gehi NP, Agarwal R, More YE. Oral isotretinoin is as effective as a combination of oral isotretinoin and topical anti-acne agents in nodulocystic acne. Indian J Dermatol Venereol Leprol 2008;74:187 |
Abstract
Background: Due to the late introduction of oral isotretinoin, there is only a single report of the use of this drug from India. Aims: To evaluate the efficacy, acceptability and side effects of oral isotretinoin in Indian conditions and to compare the efficacy of oral isotretinoin monotherapy with that of a combination of oral isotretinoin and topical anti-acne agents. Methods: Sixty clinically diagnosed patients of nodulocystic acne were divided into two groups of 30 each. Patients in group A were administered oral isotretinoin 20 mg twice a day along with topical clindamycin (1%) and adapalene (0.1%) for 24 weeks. Patients in group B were given only oral isotretinoin. An objective clinical scoring was repeated every four weeks and patients were followed up for six months on completion of treatment. Results: Out of the 60 patients, 50 completed the treatment and 35 completed the follow-up at six months. The mean pretreatment score in group A came down from 12.8 to 1.24 and in group B from 12.4 to 1.48, thus showing a 90.55% and 88% reduction in pretreatment scores respectively. There was no statistical difference in the results obtained from the two groups. Side effects were common but minor in nature. Conclusion: Isotretinoin produces gratifying results in patients of nodulocystic acne in Indian conditions. Addition of topical antiacne agents does not alter the final outcome. This addition is well tolerated but requires careful monitoring.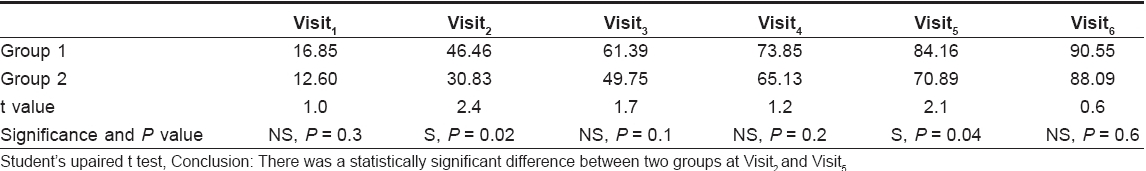






Introduction
Acne is a multifaceted dermatological disorder of the sebaceous glands, which affects 90-100% of adolescents with varying degrees of severity. Nodulocystic acne is not only the most severe form but it also has the potential of producing lifelong disfiguring scars. Isotretinoin (13- cis -retinoic acid) is an isomer of all trans- retinoic acid. It acts on all the major etiological factors involved in the pathogenesis of acne. It was approved by the US-FDA for the treatment of nodulocystic acne in 1982. Since then it has not only been extensively used but also widely publicized in North America and Europe.
As of now, there is only a single report of the use of Isotretinoin in literature from India. [1] This prompted us to conduct a formal study on the use of isotretinoin in nodulocystic acne in Indian conditions with the objectives of assessing any additional benefit from topical antiacne agents.
Methods
This is an open label, randomized study carried out at the Department of Dermatology, Indian Naval Health Service (INHS) Asvini, Mumbai, from November 2004 to April 2006 after obtaining clearance from the ethical committee of the institution. Sixty clinically diagnosed cases of nodulocystic acne were included in the study and were divided into two groups of 30 patients each. The patients in group A were administered isotretinoin 20 mg twice a day along with topical clindamycin (1%) during the daytime and adaplene (0.1%) at bed time for 24 weeks. Patients in group B were treated with the same dosage of only oral isotretinoin twice a day. Pregnant and lactating females, patients with abnormal lipid profiles, significant hepatic dysfunction and an underlying psychiatric disorder were excluded from the study. All patients were counseled and were clearly made aware of the drawbacks and limitations of the treatment. In patients below 18 years of age, counseling was done in the presence of one of the parents, while it was extended to the spouse in married patients. A written informed consent was taken at the end of counseling.
Pretreatment investigations included a routine blood count, liver function test, lipid profile and a pregnancy test for married females. A pregnancy test was repeated every four weeks while the remaining investigations were done every eight weeks. Before initiating treatment, an objective clinical scoring was done as per details given below.
Clinical evaluation (by scoring)
(i) For Comedones/Papules/Pustules
(for each)
Lesion count Score
>20 4
15-19 3
11-14 2
< 10 1
(ii) Nodules/Cysts
(for each)
Lesion count Score
>10 4
6-10 3
< 6 2
Patients were examined every four weeks and clinical scoring and side effects were recorded at each visit. This was supplemented by the physician′s global assessment as given below:
(a) Clear: 100% reduction in Pretreatment score
(b) Excellent: 75-99% reduction in Pretreatment score
(c) Good: 50-74% reduction in Pretreatment score
(d) Moderate: 25-49% reduction in Pretreatment score
(e) Poor: 0-24% reduction in Pretreatment score
Clinical photographs were recorded by a digital camera at every visit. On completion of the treatment, patients were followed up at monthly intervals for a period of six months. Statistical analysis of data obtained from the results was carried out. Patients were advised to use emollients for the lips.
Results
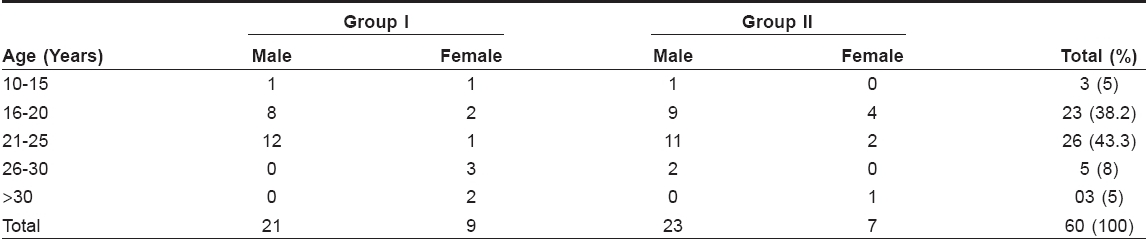
Out of the 60 patients who were initially enrolled in the study, 44 (73%) were males and the remaining were females. It was seen that out of the 16 females, only five were married. The weight of the patients varied between 50 and 65 kg. Three out of these five married couples had undergone permanent sterilization like tubal ligation or vasectomy while the other two had committed to use two different forms of effective contraception simultaneously. The majority of the patients were in the age group of 21-25years [Table - 1]. Out of the 60 patients who were included in the study, 50 completed the treatment while five patients dropped out from each group. The body weight varied from 51 to 68 kg with the median weight being 58 kg.
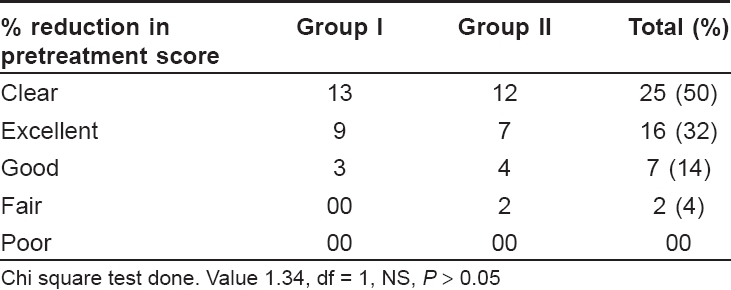
The face was the most commonly affected site in all patients (100%) followed by the back, chest and arms. The mean pretreatment scores were 12.8 and 12.4 in groups A and B, which decreased to 1.24 and 1.48 respectively at the end of treatment [Table - 2].
The physician′s global scale assessment revealed that 13 patients in group A and 12 in group B showed complete clearance of skin lesions [Table - 3] while nine patients in group A and seven in group B showed an excellent response (75-99% reduction in pretreatment scores). It was observed that the two patients who showed a moderate response (25-49% reduction in pretreatment scores) had macrocomedones with the total number of comedones being over 100. None of the patients showed a poor response.
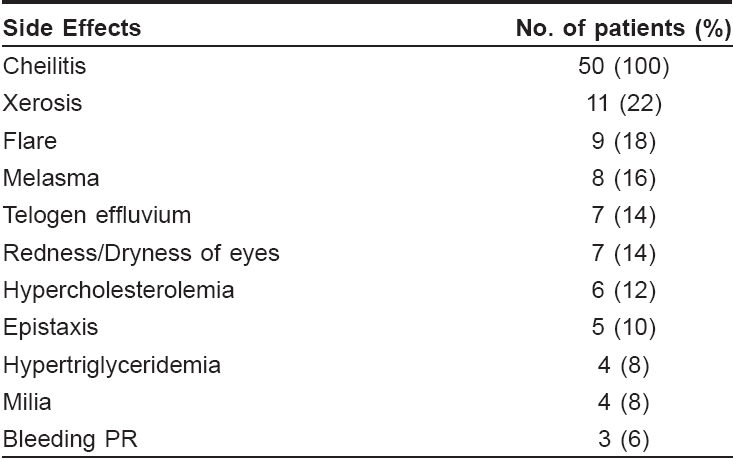
Thirty-five patients (70%) completed the six months′ follow-up, 20 in group A and 15 in group B. Out of these 35 patients, three patients had a minor relapse of Grade I or Grade-II severity acne which was managed by topical clindamycin and adapalene only. None of these patients required a second course of isotretinoin. Side effects due to isotretinoin were common but minor in nature with all the patients developing varying degrees of cheilitis [Table - 4]. It was seen that 44 out of the 50 patients developed cheilitis at four weeks, 49 at eight weeks and all 50 at 12 weeks. During follow-up, it was observed that cheilitis subsided in all the patients four weeks after discontinuing treatment. Hypercholesterolemia was seen in six (12%) patients and hypertriglyceridemia in four (8%) of the patients. These abnormalities were also transient and subsided within four weeks of stopping treatment. Surprisingly, melasma-like pigmentation was seen in eight patients, three patients in group A and five in group B. Seven (14%) patients showed redness/dryness of the eyes, five (10%) showed mild epistaxis while three (6%) showed mild bleeding per rectum (PR). Two of these three patients of bleeding PR, had underlying fissures in ano . Nine patients - six in group A and three in group B, experienced flare-ups in the form of erythema, edema and fresh papular and pustular lesions. All these flare-ups were in the first eight weeks of treatment. Treatment was not discounted in any of the patients because of side effects. There were no significant side effects due to the addition of topical antiacne agents. There was no significant difference in the pattern of side effects in the patients of two groups.
Discussion
The recommended dose of oral isotretinoin in nodulocystic acne is in the range of 120-150 mg/kg body weight. A dosage of 120 mg/kg for a 50 kg patient, would entail a treatment duration of 22 weeks and for 28 weeks for a 65 kg individual. As the weight of all the patients varied from 50 to 65 kg, a fixed dosage of 20 mg bd for 24 weeks was chosen for the purpose of overall convenience as it would not have made any substantial difference in the efficacy or side effects. [2] The epidemiological profile of this study is comparable with the results obtained by others, with a vast majority of the patients being young adults in age group of 16-29 years, the average age being 19.34 years. Mahajan et al. [3] had also reported the majority of their patients to be in the same age group with a mean age of 22 years. In our study, males outnumbered females which was in accordance with results obtained by Mahajan et al. [3] The face was the most commonly affected site (100%) followed by the back (46.66%). Ten patients dropped out of the study but none of the dropouts was related to the side effects of the drug.
A comparison of the results obtained in the two groups reveals 90.55 and 88% reduction in the pretreatment scores in groups A and B, respectively. There was no statistical difference in the results obtained in the two groups at the end of treatment, but a closer look at the mean score at each visit [Table - 5] shows relatively better results at the second (eight weeks) and fifth (20 weeks) visits. It would be pertinent to point out that this is the first study where isotretinoin monotherapy has been compared with a combination of isotretinoin and topical antiacne agents. Others have tried combinations of different systemic (but not local) antiacne agents. In a double blind, controlled study, Jones et al. [4] randomized patients of nodulocystic acne into three treatment groups of 0.5 mg/kg/wt isotretinoin, 1 g oral erythromycin daily or a combination of both and observed 73% improvement in the combination group in the total acne grade in contrast to 62% improvement with isotretinoin monotherapy. However, Marsden et al. [5] reported that a combination of 5 mg cyproterone acetate and 0.5 mg/kg/wt isotretinoin was no more effective than isotretinoin monotherapy.
In this study, patients in group B were on isotretinoin monotherapy while those in group A, were on a combination of isotretinoin and local antiacne agents. Consequently, only the results of patients in group B are being compared with the results obtained by other authors. An assessment of patients in group B using the physician′s global assessment scale, showed that 12 (48%) showed 100%, seven (28%) showed excellent (75-99%) reduction and four (16%) showed a good (51-75%) decrease in pretreatment scores. Mohammad Shahidullah et al. [6] reported an excellent response in 50.8%, good in 34.4%, fair in 12% and poor in 2.8% of their patients. The dosage used in this study ranged from 0.33-1 mg/kg body weight per day (median 0.5 mg/kg/day) for a period ranging from 1-12 months (median four months). It is possible that the relatively inadequate response seen in this study was because of the lower dose and shorter duration of treatment. In a population-based study by Hogon et al. , [7] an excellent response was seen in 83%, moderate in 11% and fair responses in 4% of the patients. These results are comparable with those obtained in our study. In a double blind, controlled study of severe nodulocystic acne, Jones et al. [8] showed 80-90% reduction in total acne lesion count at 16 weeks and maximum improvement at 20-24 weeks. They randomized patients into three treatment groups of 1 mg/kg/wt, 0.5 mg/kg/wt and 0.1 mg/1kg/wt and observed no difference in responses among the three groups. We observed 65% reduction in patients in pretreatment scoring at 16 weeks and maximum improvement at 20-24 weeks [Table - 5].
In the present study, three out of the 35 patients (8.5%) who completed follow-up showed a minor relapse at six months. Shahidullah et al. [6] reported a relapse in 5.6% of their patients within six months of discontinuation of treatment. In a long-term follow-up study (ten years) of 88 patients treated with 1 mg/kg/wt of isotretinoin, Layton [9] reported a relapse in 39% of patients of which 16% patients required oral antibiotics and 23% a second course of isotretinoin. Of the remaining 61% of the patients who were nonrelapsers, 21% required only topical therapy. They reported that isotretinoin can produce long-term remission at a dose of 1 mg/kg/day or at a cumulative dose of> 120 mg/kg.
Mucocutaneous side effects and abnormal lipid profiles were expected. All (100%) of our patients showed evidence of cheilitis and dryness of lips was observed in 90% of the patients. Melasma-like pigmentation was seen in 16% of our patients and seen after 16-20 weeks of treatment. Hyperpigmentation and melasma are the least reported side effects (1-5%) in various trials. [10] The increased incidence of side effects may be due to the greater sun exposure in Indian patients. Eighteen percent of our patients showed flare-ups within the first four weeks. All these patients had an evidence of macrocomedones and relatively higher pretreatment scores. Clock and Cunliffe [11] reported flare-ups in 6% of their cases. We observed milia in 8% of our patients, which to the best of our knowledge, has not been reported earlier.
The results of this study clearly show that oral isotretinoin produces extremely gratifying results in patients of nodulocystic acne in India. As patients of nodulocystic acne require a cumulative dosage of 120/kg/wt, a dosage of 20 mg twice a day for 24 weeks is suitable and convenient for an average Indian young adult weighing around 60 kg. However, this dose schedule may at best be used as a guideline because ultimately it has to be tailored to the requirement of the individual patient.
| 1. |
Sheth Rekha, Poonevala Vera. Isotretinoin: An Indian experience. Indian J Dermatol Venerol Leprol 2001;67:182.
[Google Scholar]
|
| 2. |
Cunliffe WJ, van de Kerkhop PCM, Caputo R. Roaccutane treatment guidelines: Results of an international survey. Dermatology 1997;194:351-7.
[Google Scholar]
|
| 3. |
Mahajan BB, Garg G. Therapeutic efficacy of intralesional triamcinolone acetonide versus intralesional triamcinolone acetonide plus lincomycin in thetreatment of nodulocystic acne. Indian J Dermatol Venerol Leprol 2003;69:217-9.
[Google Scholar]
|
| 4. |
Jones DH, Forster RA, Cunliffe WJ. A comparative study of 13-cis-retinoic acid and erythromycin in severe acne. In : Retinoid therapy - A review of clinical and laboratory research. Cunliffe WJ, Miller AJ, editors. MTP Press Ltd: Lancaster; 1984. p. 293.
[Google Scholar]
|
| 5. |
Marsden JR, Shuster S. Effect of 13-cis-retinoic acid and cyprotrene.Acetate Res 1984. p. 267.
[Google Scholar]
|
| 6. |
Shahidullah M. Isotretinoin therapy in acne vulgaris: A ten year retrospective study in Singapore. Int J Dermatolol 1994;33:60-3.
[Google Scholar]
|
| 7. |
Hogan DJ, Strand LM, Lane PR. Isotretinoin therapy for Acne: A population based study. CMAJ 1988;138:47-50.
[Google Scholar]
|
| 8. |
Jones DH, Greenwood R, Cunliffe WJ. The dose response relationship of sebum suppression to 13-cis-retinoic acid therapy in severe acne. Br J Dermatol 1983;109:366.
[Google Scholar]
|
| 9. |
Layton AM, Knaggs H. Isotretinoin for acne vulgaris-10 years laters: A safe and successful treatment. Br J Dermatol 1993;129:292-296.
[Google Scholar]
|
| 10. |
Ethan-Quan H, Nguyen Sephen, E Wolverton. Systemic retinoids. In : Comprehensive dermatologic drug therapy. Wolveron SE, editor. NB Saunders Company: p. 295-6.
[Google Scholar]
|
| 11. |
Clark SM, Cunliffe WJ. Acne flare and isotretinoin-incidence and treatment. Br J Dermatol 1995;133:26.
[Google Scholar]
|
Fulltext Views
2,119
PDF downloads
1,417





