Translate this page into:
Oral submucous fibrosis: An overview of a challenging entity
Corresponding author: Dr. Sonia Gupta, #95/3, Adarsh Nagar, Dera Bassi, Mohali - 140507, Punjab, India. sonia.4840@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Gupta S, Jawanda MK. Oral submucous fibrosis: An overview of a challenging entity. Indian J Dermatol Venereol Leprol 2021;87:768-77.
Abstract
The oral cavity is considered to be a mirror of the body’s health, as it reflects the manifestations of various systemic disorders. Most of the oral mucosa is derived embryologically from an invagination of ectoderm and thus, like other similar orifices, it may become involved in the disorders that are primarily associated with the skin. Oral submucous fibrosis is one of the commonest precancerous conditions of the oral mucosa involving any part of the oral cavity resulting in tissue scarring, dysphagia and trismus. It is a collagen-related disorder characterized by excessive fibrosis in the oral submucosa, hyalinization and degenerative changes in the muscles. This disease has become a challenging entity for dermatologists due to resemblance of its features to various mucocutaneous conditions. An improper diagnosis can lead to wrong treatment and additional complications. Dermatologists need to be aware of the characteristic features of this disease which can distinguish it from other similar conditions. This review aims to focus on the detailed aspects of oral submucous fibrosis including its historical background, etiological factors, pathogenesis, clinical features, differential diagnosis, investigations, management and future perspectives.
Keywords
Challenging
dermatology
diagnosis
mucocutaneous
oral submucous fibrosis
Introduction
The oral cavity is considered to be a mirror of the general body’s health as it reflects the manifestations of various systemic disorders.1 Most of the oral mucosa is derived embryologically from an invagination of the ectoderm, thus like other similar orifices, it may become involved in the disorders that are primarily associated with the skin.2
Oral submucous fibrosis is one of the commonest precancerous conditions of the oral cavity predominantly affecting people of South-east Asian origin.3 It is a collagen-related disorder characterized by the deposition of excessive abnormal collagen in the submucosa leading to tissue fibrosis, hyalinization and degenerative changes in the muscles.4 The unique feature of this condition is its widespread involvement in any part of the oral cavity that can result in tissue scarring, dysphagia and trismus.5
What is the need of the present review?
Oral submucous fibrosis has become a challenging entity for dermatologists, due to its resemblance to various other mucocutaneous conditions. Many times, it leads to a wrong modality of treatment due to improper diagnosis which leads to more complications. So, dermatologists need to be aware of the characteristic features of this disease which distinguish it from other similar conditions. This review focuses on the detailed aspects of oral submucous fibrosis including its historical background, etiological factors, pathogenesis, clinical features, differential diagnosis, investigations, management and future perspectives.
Historical Background and Nomenclature
Oral submucous fibrosis is well established in the Indian medical literature since the time of Sushruta (600 B.C), who described a condition named “Vidari” with clinical manifestations as “progressive narrowing of mouth, depigmentation of oral mucosa and pain on taking food.” He observed that these features were the characteristics of oral submucous fibrosis.6 Schwartz in 1952 described this disease in five Indian women settled in Kenya which he called “atrophica idiopathica mucosae oris.”7 Later, many other terminologies have been used for oral submucous fibrosis6-9 which are summarized in Table 1.
| Name of the researcher (year) | Terminology |
|---|---|
| Joshi (1953) | Submucous fibrosis |
| Lai D (1953) | Diffuse oral submucous fibrosis |
| Su IP (1954) | Idiopathica scleroderma of the mouth |
| Desa JV (1957) | Submucous fibrosis of palate and cheek |
| George AT (1958) | Submucous fibrosis of palate and mucosa membrane |
| Rao ABN (1962) | Idiopathic palatal fibrosis |
| Behl PN (1962) | Sclerosing stomatitis |
| Sirsat SS and Khanolkar VR (1962) | Submucous fibrosis of the palate |
| Pindborg JJ (1964) | Juxta-epithelial fibrosis |
| Pindborg JJ and Sirsat SM (1966) | Oral submucous fibrosis’ |
| Goleria (1970) | Subepithelial fibrosis |
| Abrol BM and Krishnamoorthy S (1970) | Idiopathic fibrosis |
| Ramanathan K (1981) | Asian sideropenic dysphagia |
| ChaturvediP (2009) | Gutkha syndrome or areca nut chewer’s syndrome |
| More C (2018) | Areca nut-induced oral fibrosis and a collagen metabolic disorder |
The term, “oral submucous fibrosis,” was given by Pindborg and Sirsat in 1966 and is in use till today. They further defined it as “an insidious chronic disease affecting any part of the oral cavity and sometimes the pharynx. Although occasionally preceded by and/or associated with vesicle formation, it is always associated with a juxta-epithelial inflammatory reaction followed by a fibroelastic change of the lamina propria with epithelial atrophy leading to stiffness of the oral mucosa causing trismus and inability to eat.”10 World Health Organization in 1978 defined this disease as, “a slowly progressive disease in which fibrous bands form in the oral mucosa, ultimately leading to the severe restriction of movement of the mouth including tongue.”11 More and Rao in 2019 defined it as “a debilitating, progressive, irreversible collagen metabolic disorder induced by chronic chewing of areca nut and its commercial preparations; affecting the oral mucosa and occasionally the pharynx and oesophagus; leading to mucosal stiffness and functional morbidity, and has a potential risk of malignant transformation.” 6
Etiology
Oral submucous fibrosis is a disease of multifactorial etiology [Table 2]. The main causative agent is “areca nut” either in raw or baked form.12 Major constituents of the areca nut are carbohydrates, fats, proteins, crude fiber, polyphenols (mainly flavonoids and tannins), alkaloids (conclusively identified; arecoline, arecaidine, guvacine and guvacoline) and minerals (most abundant; copper).13 Average daily intake of copper gets increased from 1.6 mg/day in a normal adult up to 5 mg/day in an areca nut chewer. Other contributory factors include; tobacco, chilies, nutritional deficiency, genetics, autoimmunity and infections.14-33 Recently, plasma fibrinogen degradation products have been identified in these patients which have provided a new direction for the etiopathogenesis of this disease.34
| Factor | Role | Reference number |
|---|---|---|
| Areca nut | Constant chewing causes microtrauma. Arecoline and arecaidine nitrosation causes DNA alkylation with proliferation of fibroblasts. Copper stimulates fibrogenesis through upregulation of LOX activity. Flavonoids and tannins help in cross-linking of fibers. |
4,12,13 |
| Tobacco Chewing and smoking | No evidence found with smoking. Dip, snuff, snus. |
4,14–16 |
| Chilies | Hypersensitivity reactions to capsaicin. | 1,4,17,18 |
| Nutritional (Fe, Zn, vitamins A, B, C; beta carotene) |
Result in mucosal atrophy making oral mucosa vulnerable to soluble irritants. Increased levels of iron enhance hydroxylation of proline. and lysine in the process of collagen synthesis. |
16,19–23 |
| Genetic | Overexpression, mutations, altered gene expression, polymorphism. Increased frequency of HLA-A10, HLA-B7, HLA-DR3, B3/DR3 haplotypes. Adenomatous polyposis coli gene mutation and decreased p53 expression. Familial occurrence. |
24–27 |
| Immunological | Increased expression of CD4+Ve and CD8+Ve cells on lymphocytes and Langerhans cells. Alterations in serum immunoglobulins. Increased incidence of autoantibodies. |
28–30 |
| Infections | Candida act as a risk factor in malignant transformation. Viral antigens(HSV, HPV, EBV) result in immunosuppression. Helicobacter pylori results in mucosal inflammation. |
31–33 |
| Plasma fibrinogen degradation products | FPF present in the saliva of OSMF patients behaves like thrombin that clots the exudates, and the body in response to produces more fibrinogen and its degradation products and hence increased fibrosis. | 34 |
CD=cluster differentiation, DNA=deoxyribonucleic acid, EBV=Epstein–Barr virus, FPF=fibrin precipitating factor, HSV=Herpes simplex virus, HLA=human leukocyte antigen, HPV=Human papillomavirus, LOX=lysyl oxidase, OSMF=oral submucous fibrosis
Pathogenesis
Oral submucous fibrosis is a collagen-related disorder regulated by inflammatory mediators such as cytokines and growth factors. Central to the pathogenesis of this disease is fibrosis which could be either because of increased proliferation of fibroblasts or increased collagen synthesis and decreased collagen degradation [Figure 1].
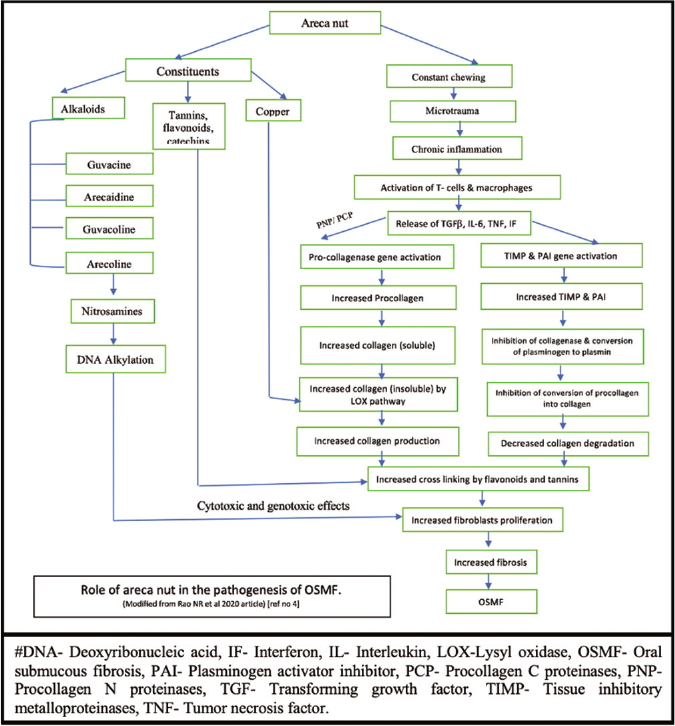
- Role of areca nut in the pathogenesis of oral submucous fibrosis
Initiation of the inflammatory response
Alkaloids and flavonoids, from the areca nut, are absorbed and undergo metabolism. Alkaloids mainly arecoline, undergoes nitrosation and gives rise to N nitrosamines which might have a highly cytotoxic and genotoxic effect on cultured human oral mucosal epithelial cells and fibroblasts.35 Also, constituents of areca nut and their metabolites act as a source of constant irritation to the oral tissues; thus the microtrauma produced by the friction of areca nut fragments facilitates the diffusion of alkaloids and flavonoids into the subepithelial connective tissue, resulting in juxta-epithelial inflammatory cell infiltration. Over some time, due to persistent habit, chronic inflammation sets in at the site, and it is characterized by the presence of activated T cells and macrophages which produce various chemical mediators of inflammation, especially prostaglandins, interleukin-6, tumor necrosis factor, interferon ά and transforming growth factor β.36
Collagen production pathway
Transforming growth factor β activates the procollagen genes, resulting in the production of more pro-collagen. It also induces the secretion of procollagen C-proteinase and procollagen N-proteinase, both of which are required for the conversion of procollagen to collagen fibrils. An important enzyme, “lysyl oxidase” plays a crucial role here causing increased cross-linking of the collagen. This enzyme is dependent on copper for its functional activity. During its biosynthesis, copper gets incorporated into it and aids in reoxidizing the reduced enzyme facilitating the completion of the catalytic cycle. Combined actions of this enzyme, flavonoids, catechins and tannins of areca nut, result in more cross-linking of the collagen fibers, further producing more collagen in an insoluble form.18
Collagen degradation pathway
Transforming growth factor-β activates the genes for tissue inhibitory metalloproteinases; thereby more tissue inhibitory metalloproteinases are formed. This inhibits the activated collagenase enzyme that is necessary for the degradation of collagen. It also activates the gene for plasminogen activator inhibitor; thus, plasmin formation is significantly reduced. Plasmin is required for the conversion of pro-collagenase into the active form of collagenase. The flavonoids of areca nut also inhibit the collagenase activity. The reduction in the activity and the levels of collagenase result in a decrease in collagen degradation.25
Fibrosis
The overall effect of both the pathways is increased fibrosis which is the hallmark of oral submucous fibrosis.
Epidemiology
According to World Health Organization, >5 million people are affected by oral submucous fibrosis globally.37 Research reveals that this disease occurs predominantly among Indians and people of Indian origin living outside India. Recent data suggests that the prevalence of this disease in India has increased from 0.03% to 6.4% in the last four decades. According to various studies conducted in different regions of India, its prevalence was 30–42% in the northern zone, 6.3–14.2% in the north-central zone, 2.7% in the eastern zone, 0.03–0.2% in the western zone and 0.6% in the southern zone.38 All these regional variations were seen due to different varieties of gutka and areca nut used by the population. Despite the government having taken many steps to ban the use of these products, the prevalence of this condition has increased.
The disease is commonly seen in the young to middle aged groups; however, it can occur at any age.39 Surprisingly, it has been observed that in recent years, school-going children are also getting affected by this disease due to their addiction to areca nut.40 It can also affect the pediatric age group.41A female predilection in one Indian study could be correlated to nutritional deficiency particularly iron.42 However, other studies reveal male predominance.43-45
Clinical Features
Signs and symptoms
Signs and symptoms of oral submucous fibrosis vary according to the stage of the disease. Various scholars have classified this disease into different stages clinically. Most recently, Passi et al.46 have categorized it into four stages clinically [Table 3]. The initial signs and symptoms of the disease are stomatitis, vesicle formation, erythematous mucosa, burning sensation, ulceration, mild blanching, melanotic mucosal pigmentation [Figure 2a], petechiae and dry mouth, followed by fibrosis which is the hallmark of this disease. Older lesions include vertical and circular palpable fibrous bands in the buccal mucosa and around the mouth opening or lips and marble-like appearance of the mucosa. In the later stages, symptoms become severe resulting in blanching involving the posterior parts like the palate and uvula [Figure 2b], broad thick fibrous palpable bands at cheeks, lips and the floor of the mouth, rigid mucosa, stiff, small, depapillated tongue with restricted movements, restricted mouth opening leading to trismus [Figure 2c], shrunken bud-like uvula (hockey stick shape), speech and deglutition defects due to the involvement of eustachian tube, nasal intonation of speech, hoarseness of voice, sunken cheeks, loss of nasolabial fold, prominent ante gonial notch, hearing impairment, severe weight loss and the sequelae of the disease leading to premalignant and malignant changes in the oral mucosa such as leukoplakia, erythroplakia and oral squamous cell carcinoma.
| Grading/staging | Clinical grading | Functional grading | Histopathological staging | Treatment | Prognosis |
|---|---|---|---|---|---|
| Grade I | Involvement of <1/3rdof the oral cavity, mild blanching, burning sensation, recurrent ulceration and stomatitis. Dryness of the mouth | Mouth opening up to 35 mm | Stage of inflammation: fine edematous collagen, congested blood vessels, abundant neutrophils along with lymphocytes with myxomatous changes in the subepithelial connective tissue | Cessation of the habit, nutritional support, antioxidants, topical steroid ointment | Excellent |
| Grade II | Involvement of 1/3rd 2/3rd of the oral cavity, blanching, mottled and marble-like appearance, fibrotic bands palpable and involvement of soft palate and premolar area | Mouth opening 25–35 mm, cheek flexibility reduced by 33% | Stage of hyalinization: Juxtaepithelial collagen hyalinization, lymphocytes, eosinophils, dilated and congested blood vessels, less fibroblastic activity. Granulation changes in the muscle layer with reduced inflammatory cells in the subepithelial layer | Habit cessation, nutritional supplement, intralesional injection of placental extracts, hyaluronidase, steroid therapy physiotherapy | Good, the recurrence rate is low |
| Grade III | Involvement of greater than >2/3rd of the oral cavity Severe blanching, broad thick fibrous palpable bands at cheeks and lips and rigid mucosa, depapillated tongue and restricted tongue movement, shrunken bud like uvula, floor of the mouth involvement and lymphadenopathy |
Mouth opening 15–25 mm, Cheek flexibility reduced by 66% | Stage of fibrosis: complete collagen hyalinization without fibroblast and edema, obliterated blood vessels, plasma cells and lymphocytes, extensive fibrosis with hyalinization from subepithelial to superficial muscle layers with atrophic, degenerative changes | Surgical treatment including band excision and reconstruction with buccal palatal flap or split-thickness graft, bilateral temporalis myotomy and coronoidectomy | Fair, the recurrence rate is high |
| Grade IV | Changes like leukoplakia, erythroplakia ulcerating and a suspicious malignant lesion | Mouth opening<15 mm or nil | Stages of malignant transformation: erythroplakia changes into squamous cell carcinoma | Surgical treatment and biopsy of a suspicious lesion | Poor, malignant transformation |
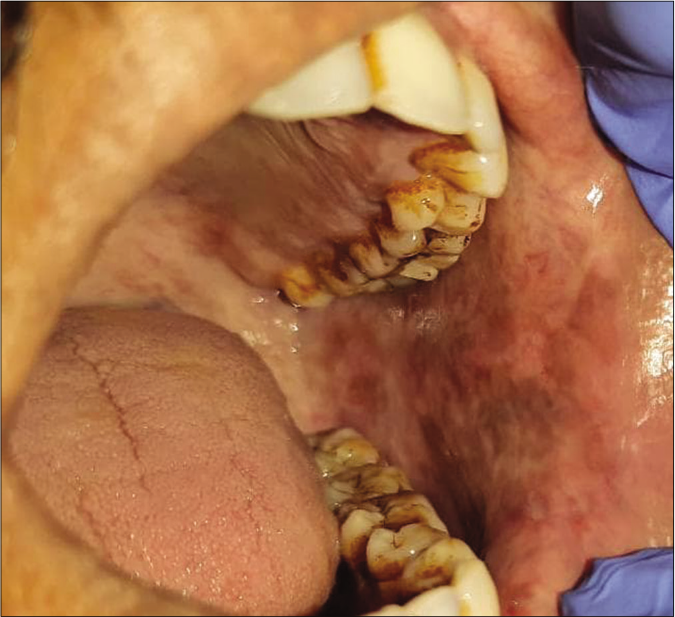
- Blanching of oral mucosa with melanotic pigmentation
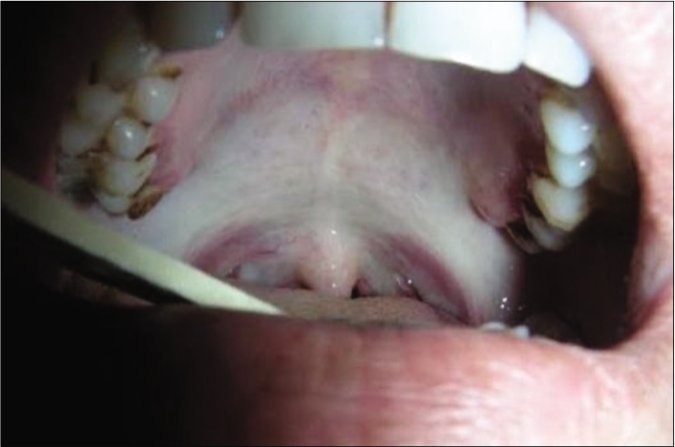
- Shrunken uvula with blanching of palatal mucosa
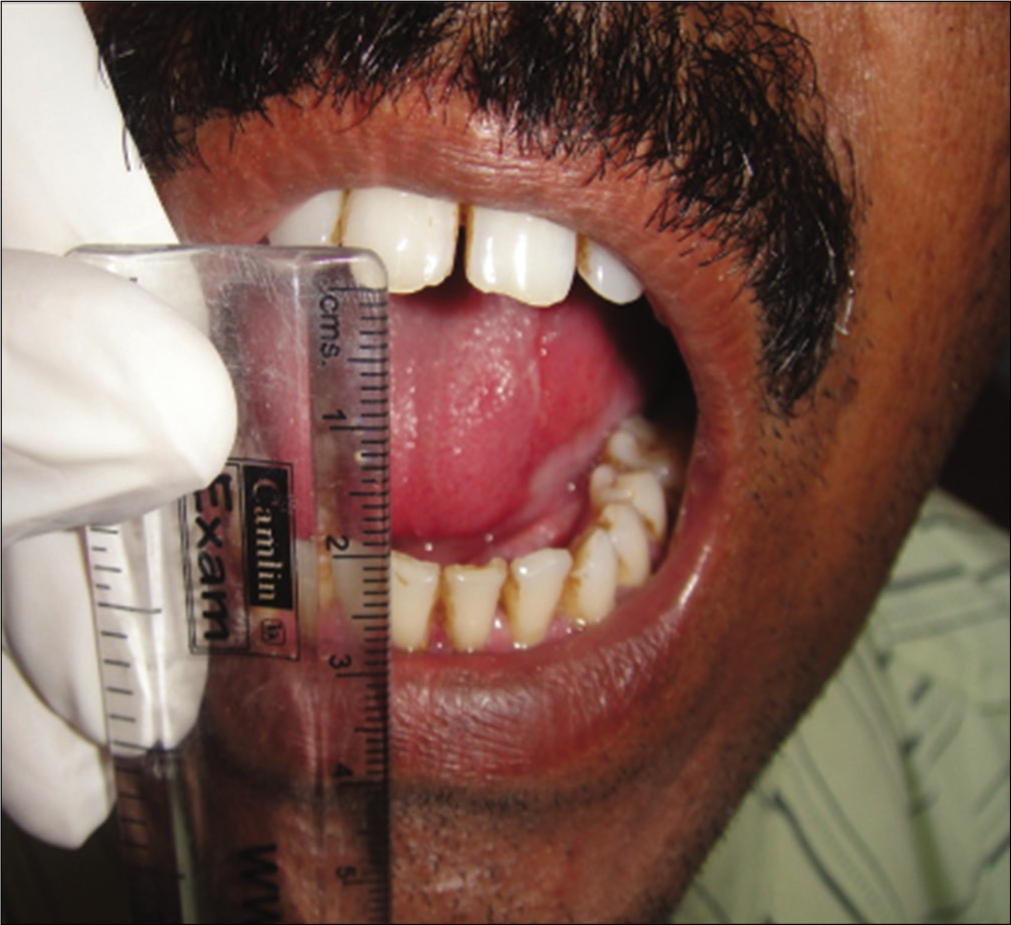
- Reduced mouth opening
Complications
Functional abnormalities
Oral submucous fibrosis may be followed by various complications such as malnutrition, speech and hearing defects, airway problems, masticatory difficulty and deafness.47
Malignant transformation
Pindborg and Zachariah in 1965 and Ramanathan et al. in 1975, described the precancerous nature of this condition.48,49 Warnakulasuriya et al. in 2007 categorized it into oral potentially malignant disorders.50
Patients with oral submucous fibrosis have been reported with a higher risk of developing carcinoma as compared to the other diseases. In a 17-year follow-up study reported in 1970, it was found that 7.6% of these cases transformed into oral squamous cell carcinoma.51 which increased up to 9% in the later studies.52,53 It has been found that areca nut products and their derivatives such as arecoline and nitrosamines can induce neoplastic changes in the oral mucosa.54 Dense fibrosis and less vascularity of the corium, in the presence of altered cytokine activity, create a unique environment for the carcinogens from the nut to act on the epithelium. These carcinogens accumulate over a long period either on or immediately below the epithelium before getting diffused into the deeper tissues; thus they get prolonged time for causing the changes in the epithelium. The dermatologist must look for dysplastic features in the surface epithelium histologically and any other associated premalignant lesion in the oral cavity of these patients. Certain biomarkers such as vimentin, plasminogen activator inhibitor-1, keratinocyte growth factor-1 and cystatin C play a crucial role in determining the malignant potential of this condition by showing increased expression in the buccal mucosa fibroblasts. Also, altered expression of p53, bcl-2, bax and ki-67 has been found in oral submucous fibrosis cases signifying an increased risk of carcinomatous changes.55-58 Mutations in tumor suppressor gene; adenomatous polyposis coli could also be potentially involved in its malignant transformation.26
Differential Diagnosis
Oral submucous fibrosis resembles many other mucocutaneous disorders either clinically or histologically, leading to difficulty in diagnosis. Thus, it is important to know those conditions and their distinguishing features [Table 4].
| Disease name | Similar features | Differentiating features |
|---|---|---|
| Scleroderma | Both are collagen-related diseases59 Clinical findings such as pale mucosa, microstomia, reduced mouth opening, facial atrophy and dysphagia are seen in both59-62 Histological features such as flat epithelium with loss of rete ridges, submucosal hyalinization, degeneration of collagen fibers, oedema present in both61 |
AN chewing associated with OSMF, not reported with scleroderma OSMF is limited up to oral cavity, but in scleroderma, extensive internal organs and skin are also involved61 Special criteria are there to diagnose scleroderma clinically63 Scleroderma predominant in blacks, while OSMF has no racial predilection64 Scleroderma is also associated with neurological disorders65 Histologically perivascular lymphocytes and thick blood vessels are mostly seen in scleroderma and not in OSMF61 |
| Amyloidosis | Homogeneous hyalinized mucosa is seen in both diseases clinically66,67 Histologically both are characterized by hyalinized stroma67 |
Amyloidosis is neither collagen or AN related Amyloidosis involves internal organs too68 Special stains like Congo red and crystal violet are used for amyloid67 In amyloidosis, hyalinization is due to deposition of amyloid, whereas in OSMF, it is assumed to be due to excessive cross-linking of collagen fibers due to the action of constituents of AN69 |
| Iron deficiency anemia | Clinical findings such as pallor mucosa along with pigmentation, blanching, burning sensation, dysphagia and atrophy of tongue seen in both.70-72 OSMF has been considered as an “Asian version of sideropenic dysphagia” which is associated with iron deficiency73 Hb and iron levels are reduced in blood in both71 |
Iron deficiency is characterized by other associated features such as Gray hair, Koilonychia, platynychia, tiredness, lethargy and breathlessness |
| Generalized fibromatosis | Histologically fibrosis and hyalinized stroma of desmoid type of fibromatosis resembles the stroma of OSMF74,75 | Fibromatoses is a tumor-like condition and OSMF not76 |
AN=areca nut, OSMF=oral submucous fibrosis
Scleroderma/systemic sclerosis
There are reports in literature of some cases in which it had become difficult to differentiate between oral submucous fibrosis and scleroderma due to the presence of similar clinical findings.59-64 Shetty et al. in 2016 reported a case of oral submucous fibrosis with facial atrophy, which was suspected to be a case of scleroderma. Later, it was revealed that the atrophy was of cerebrum and cerebellum region and neurological disorder was also associated with it.65 Except for epithelial dysplasia, all the histopathological features were the same as of oral submucous fibrosis. A final diagnosis of oral submucous fibrosis was made in that case. Panchbhai et al. in 2016 reported a case in which the patient had presented with the chief complaint of tightening of mouth, but clinically, as no features of oral submucous fibrosis were found, a final diagnosis of scleroderma was given.59 Thus, proper diagnosis can be made only if the clinician is aware of the detailed aspects of both disorders. Table 4 reflects the characteristic features of both diseases to reach an accurate diagnosis.
Amyloidosis
Amyloidosis is a group of heterogeneous disorders characterized by abnormal excessive accumulation of a specific protein known as amyloid either in localized or systematic form. In the oral cavity, amyloidosis is very rare and may appear as hyalinized mucosa, multiple soft nodules and macroglossia. Macroglossia can cause significant morbidity due to problems with breathing, talking and chewing, resulting in the need for feeding tubes and tracheostomy.66-69 Histologically, amyloidosis shows many features resembling oral submucous fibrosis.
Table 4 describes the differentiating features of amyloidosis and oral submucous fibrosis.
Iron deficiency anemia
Iron deficiency is the most common anemia in the general population. Although oral submucous fibrosis and iron deficiency anemia exist as separate conditions, yet some of the clinical findings of both mimic each other [Table 4].70-73 Coexistence of iron deficiency anemia, mucosal atrophy, dysphagia and esophageal web formation is referred to as sideropenic dysphagia/ Plummer–Vinson syndrome. This condition is at more prone to develop oral cancer.73 Oral submucous fibrosis has been considered as an “Asian version of sideropenic dysphagia” in the literature. Iron deficiency can co-exist with oral submucous fibrosis too. Lack of iron results in qualitative and quantitative defects in the oxygen and nutrient perfusion of the lamina propria and the overlying mucous membrane leading to epithelial atrophy in oral submucous fibrosis.23
Generalized fibromatosis
Fibromatosis is a group of locally aggressive, nonmetastasizing myofibroblastic neoplasms.74 It can be classified as deep (desmoid type) and superficial (palmar, plantar and penile) subtypes.75-78 Although soft tissue masses are not produced in the usual sense, the fibrosis of oral submucous fibrosis may be confused with generalized fibromatosis, especially of desmoid type.76 Desmoid-type fibromatosis can be further subdivided into extra-abdominal, abdominal and intra-abdominal variants.74 Regardless of location, the morphologic features of desmoid-type fibromatosis are relatively consistent, and characteristically, tumors are composed of the proliferation of bland fibroblasts/ myofibroblasts arranged in long sweeping fascicles admixed with thin-walled blood vessels. But there may be some histological variations in the stroma like keloidal/nodular/ hypercellular/hyalinized/staghorn pattern. The hyalinized pattern may resemble hyalinization seen in oral submucous fibrosis.75 So, both conditions need to be differentiated by correlating with their other findings [Table 4].
Investigations
Clinical history and oral symptoms are sufficient to make a clinical diagnosis of oral submucous fibrosis ; however, to evaluate the stage of progression, histopathological examination is essential.
Histopathology
Histopathologically, it is characterized by marked epithelial atrophy, loss of rete pegs and subepithelial hyalinization with abundant fibrosis and muscle degeneration. The epithelium may show dysplastic features5 [Figure 3]. Passi et al. in 2017 divided it into four histopathological stages.46 Changes occur depending on the stage of the disease [Table 3].
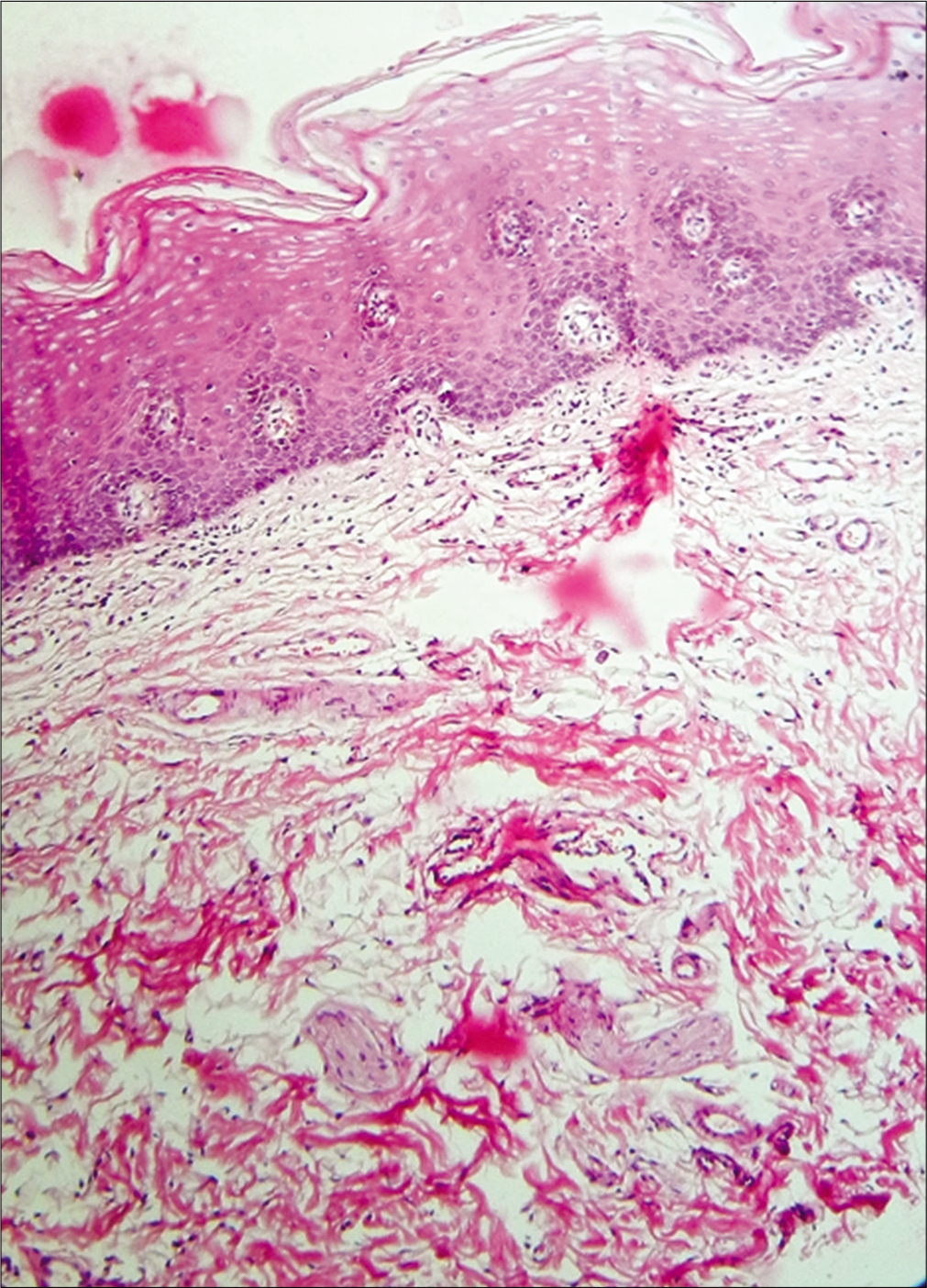
- Histopathology of oral submucous fibrosis (H and E, ×400)— Parakeratinized stratified squamous oral epithelium with the absence of rete ridges. Thickening of the spinous layer with dysplastic features like hyperchromatism, increased nuclear to cytoplasmic ratio in the basal region. Interlacing bundles of dense hyalinized collagen fibers, inflammatory infiltrate predominantly lymphocytes and fibrosed blood vessels in the connective tissue. Deeper connective shows the presence of the T.S section of the muscle fibers.
Hematological investigations
Patients of oral submucous fibrosis manifest with various hematological abnormalities such as decreased hemoglobin, T- lymphocytes count, serum iron, calcium, protein (mainly albumin), beta carotene, vitamin B complex, zinc, copper, serum antioxidants levels, lactic acid dehydrogenase IV/ II ratio; and increase in erythrocyte sedimentation rate, tissue iron-binding capacity, Immunoglobulin G, A, M, eosinophil count, serum mucoproteins, cadmium, selenium, magnesium and plasma fibrinogen degradation product levels with increased grade and stage of the disease indicating the severity of the disease.5,34,43
Immunohistochemistry
Since immune dysregulation has been observed in oral submucous fibrosis patients, various immunohistochemical markers can be used to diagnose the disease. Many studies have revealed that all cases of oral submucous fibrosis showed CD3 +ve, CD4 +ve cells in the basal layer of epithelium, juxta - epithelium and lamina propria when stained with HLA-DR antibody. Only a few cells were CD8 +ve and CD45RA +ve. There was increased expression of vimentin, plasminogen activator inhibitor-1, keratinocyte growth −1, cystatin C in buccal mucosa fibroblasts of these patients, altered expression of p53, bcl-2, bax and ki-67 markers55-58 and mutations in the adenomatous polyposis coli gene.26
Silver binding nucleolar organizer region proteins
It acts as a simple and reproducible cytological test indicative of the proliferative status of cells, particularly of hemopoietic and epithelial origin.5 Some studies reveal a progressive increase in their mean count in oral submucous fibrosis mucosa as compared to normal mucosa.79 Their counting may be useful as a predictor of the biological behavior of this disease.
Saliva as a biomarker
In some studies, it has been found that the saliva of oral submucous fibrosis patients has shown a higher level of S-100A7, salivary 8-hydroxy-2-deoxyguanosine, monoaldehyde, lactic acid dehydrogenase, total salivary protein and lipid peroxides as compared to normal individuals and decreased levels of salivary superoxide dismutase, vitamins A, C and E.80 High level of S-100A7 in the saliva of these patients indicates the risk potential of this disease.81
Grading of Oral Submucous Fibrosis
For implementing a proper treatment plan and evaluation of any disease, it becomes essential to divide it into specific grades or stages which can help in indicating the level of disease. It also helps to diagnose the disease at an early stage to prevent further complications. Various researchers have classified oral submucous fibrosis into different stages and grades depending upon the features either clinically, functionally, or histologically. And as the grades increase, the severity of the disease also increases. These parameters are of great concern in the clinical practice where the clinician can follow a proper treatment plan after accessing the grades of this disease. Most recently, Passi et al. in 2017 have divided oral submucous fibrosis into four major grades [Table 3].
Management of Oral Submucous Fibrosis
Conventional therapies used in the management of oral submucous fibrosis are empirical and symptomatic [Table 3]. The first focus is to diagnose the etiological factor responsible for the disease. History regarding habits, psychological status and stress should be evaluated. Detailed medical and family history to rule out any genetic predisposition is also necessary. The patient must be encouraged to discontinue their habit. However, cessation of habit does not necessarily lead to a complete cure. This step is beneficial in the initial stages to some extent and is recommended to prevent further progression of symptoms.82 Mild to moderate cases can be treated by the use of nonsurgical aids such as nutritional support, immunomodulatory drugs, local drug delivery,83-86 combined drug therapy, lycopene,86 physiotherapy82 and hyperbaric oxygen therapy.87,88 Ayurvedic treatment has also been tried in these patients and has shown effective results in various studies.89-93 In the moderately advanced stages with limited mouth opening and biopsy showing dysplastic changes, the release of fibrotic bands by the surgical means is necessary. However, the simple division of fibrotic bands using a blade may result in more fibrosis and disability. Since then various other surgical methods are being used, including split-thickness skin grafts, buccal fat pad grafts, microvascular free radial forearm flaps, tongue flaps and nasolabial flaps, etc., to reconstruct the defect. But these surgical procedures have a few complications such as invasive technique leading to trauma to mucosa and tissue, tissue shrinkage, delayed and defective wound healing with scarification, recurrent fibrosis and making difficulty in regular monitoring of affected mucosa for carcinomatous changes.94,95 Recently, surgeons have demonstrated the use of Laser (Light amplification by stimulated emission of radiation) in the management of oral submucous fibrosis and have found this technique to be more advantageous as compared to conventional surgical procedures resulting in minimum or no postoperative complications, healthy wound healing without scar formation and improvement in all grades of trismus irrespective of the type of laser used.96 The most common lasers used for the management of this disease are the diode laser followed by KTP-532, CO2 laser and ErCr: YSGG laser. Each of them has a specific wavelength, penetration depth and the time of application.97 However, large-scale studies are still required to investigate the efficacy and side-effects of this technology.
Stem cell therapy has also been proved to be beneficial in treating oral submucous fibrosis patients. Autologous bone marrow stem cell injections induce angiogenesis in the area of the lesion which, in turn, decreases the extent of fibrosis thereby leading to a significant increase in mouth opening.98
Overall Prognosis and Prevention
At present, oral submucous fibrosis is considered to be one of the common oral potentially malignant conditions leading to high morbidity. Early diagnosis and management are necessary to prevent further complications. Early stages may improve with treatment, but moderately advanced stages show worse prognosis. Patients must be motivated to quit their habit of areca nut chewing and be made aware of the complications of the disease. Governmental initiatives to ban such products which are the major etiological and contributory factors for the development of this condition,are necessary. Although, cessation of habit does not cure the disease completely, it may help in preventing further progression.
Conclusion
Despite having a striking clinicopathological pattern, the diagnosis of this disease is still a challenging task for the dermatologist because of the close resemblance of some of its features to other mucocutaneous disorders. Since oral submucous fibrosis is a special entity of the oral region, its many aspects may not be known to a general dermatologist. Thus, a dermatologist must be aware of overlapping features with other mucocutaneous disorders to avoid diagnostic pitfalls and subsequent inappropriate management. Moreover, due to the high rate of malignant transformation, early diagnosis of this condition is essential.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- A novel clinical protocol for therapeutic intervention in oral submucous fibrosis: An evidence based approach. J Oral Maxillofac Pathol. 2018;22:382-91.
- [CrossRef] [PubMed] [Google Scholar]
- A mouthful of epithelial-mesenchymal interactions. J Invest Dermatol. 2003;121:vii-viii.
- [CrossRef] [PubMed] [Google Scholar]
- Oral Submucous Fibrosis: A Review on Etiopathogenesis, Diagnosis, and Therapy. Int J Mol Sci. 2019;20:22.
- [CrossRef] [PubMed] [Google Scholar]
- Oral submucous fibrosis: A contemporary narrative review with a proposed interprofessional approach for an early diagnosis and clinical management. J Otolaryngol Head Neck Surg. 2020;49:1-10.
- [CrossRef] [PubMed] [Google Scholar]
- In Shafer's Textbook of Oral Pathology In: Benign and Malignant tumours of Oral Cavity. Ch. 3. (6th ed). India: Elsevier; 2009.
- [Google Scholar]
- Proposed clinical definition for oral submucous fibrosis. J Oral Biol Craniofac Res. 2019;9:311-4.
- [CrossRef] [PubMed] [Google Scholar]
- Atrophia Idiopathica (tropica) mucosae oris. Demonstrated at the eleventh international dental congress, London, 1952 (cited by Sirsat & Khanolkar) Indian J Med Sci. 1962;16:189-97.
- [Google Scholar]
- Oral submucous fibrosis-an alternative hypothesis as to its causes. Med J Malaysia. 1981;36:243-5.
- [Google Scholar]
- Oral submucous fibrosis in a 4-year-old girl. Oral Surg Oral Med Oral Pathol. 1985;59:475-8.
- [CrossRef] [Google Scholar]
- Oral submucous fibrosis with its possible effect on eustachian tube functions: A tympanometric study. Indian J Otolaryngol Head Neck Surg. 2004;56:183-5.
- [Google Scholar]
- Pathogenesis of oral submucous fibrosis. J Cancer Res Ther. 2012;8:199-203.
- [CrossRef] [PubMed] [Google Scholar]
- Chemical and analytical aspects of areca nut. Addict Biol. 2002;7:99-102.
- [CrossRef] [PubMed] [Google Scholar]
- Association of Candida species with oral submucous fibrosis and oral leukoplakia: A case control study. Ann Clin Lab Res. 2018;06:248.
- [CrossRef] [Google Scholar]
- Classification system of Oral submucous fibrosis. J Ind Acad Oral Med Radiol. 2012;24:24-9.
- [CrossRef] [Google Scholar]
- Betel nut chewing, oral premalignant lesions, and the oral microbiome. PLoS One. 2017;12:e0172196.
- [CrossRef] [PubMed] [Google Scholar]
- Estimation and comparison of copper content in raw areca nuts and commercial areca nut products: Implications in increasing prevalence of oral submucous fibrosis (OSMF) J Clin Diagn Res. 2014;8:247-9.
- [CrossRef] [PubMed] [Google Scholar]
- Raised tissue copper levels in oral submucous fibrosis. J Oral Pathol Med. 2000;29:241-8.
- [CrossRef] [PubMed] [Google Scholar]
- Oral submucous fibrosis: An overview with evidence-based management. Int J Oral Health Sci Adv. 2015;3:40-9.
- [Google Scholar]
- Clinical profile and serum beta-carotene levels in oral submucous fibrosis. Indian J Otolaryngol Head Neck Surg. 2005;57:191-5.
- [Google Scholar]
- An estimation of serum malondialdehyde, superoxide dismutase and vitamin A in oral submucous fibrosis and its clinicopathological correlation. J Oral Pathol. 2007;11:23-7.
- [CrossRef] [Google Scholar]
- Etiopathogenesis and treatment strategies of oral submucous fibrosis. J Ind Aca Oral Med Radiol. 2011;23:598-602.
- [CrossRef] [Google Scholar]
- Pathogenesis of oral submucous fibrosis: The past and current concepts. Int J Oral Maxillofac Pathol. 2012;3:27-36.
- [Google Scholar]
- Interaction of collagen-related genes and susceptibility to betel quid-induced oral submucous fibrosis. Cancer Epidemiol Biomarkers Prev. 2002;11:646-53.
- [Google Scholar]
- Molecular pathogenesis of oral submucous fibrosis-a collagen metabolic disorder. J Oral Pathol Med. 2005;34:321-8.
- [CrossRef] [PubMed] [Google Scholar]
- Adenomatous polyposis coli gene mutation and decreased wild-type p53 protein expression in oral submucous fibrosis: A preliminary investigation. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2001;92:202-7.
- [CrossRef] [PubMed] [Google Scholar]
- Familial occurrence of oral submucous fibrosis: Report of eight families from northern Kerala, south India. Indian J Dent Res. 2004;15:139-44.
- [Google Scholar]
- An immunohistochemical study of oral submucous fibrosis. J Oral Pathol Med. 1997;26:75-82.
- [CrossRef] [PubMed] [Google Scholar]
- Oral submucous fibrosis patients have altered levels of cytokine production. J Oral Pathol Med. 2000;29:123-8.
- [CrossRef] [PubMed] [Google Scholar]
- High incidence of autoantibodies in Taiwanese patients with oral submucous fibrosis. J Oral Pathol Med. 2002;31:402-9.
- [CrossRef] [PubMed] [Google Scholar]
- Prevalence of viral (HPV, EBV, HSV) infections in oral submucous fibrosis and oral cancer from India. Acta Otolaryngol. 2010;130:1306-11.
- [CrossRef] [PubMed] [Google Scholar]
- Oral submucous fibrosis and oral yeast carriage-A case control study in Srilankan patients. Mycoses. 2007;50:116-20.
- [CrossRef] [PubMed] [Google Scholar]
- Helicobacter pylori coinfection is a cofounder, modulating mucosal inflammation in OSMF. Indian J Dent Res. 2009;20:206-11.
- [CrossRef] [PubMed] [Google Scholar]
- Plasma fibrinogen degradation products in oral submucous fibrosis. J Int Soc Prev Community Dent. 2015;5:141-6.
- [CrossRef] [PubMed] [Google Scholar]
- Genotoxic effect of local & commercial areca nut & tobacco products-A review. Int J Health Sci Res. 2017;7:326-31.
- [Google Scholar]
- Oral submucous fibrosis: Review on etiopathogenesis. J Cancer Sci Ther 1. 2009;1:72-7.
- [CrossRef] [Google Scholar]
- Prevalence of oral submucous fibrosis among habitual gutkha and areca nut chewers in Moradabad district. J Oral Biol Craniofac Res. 2014;4:8-13.
- [CrossRef] [PubMed] [Google Scholar]
- Epidemiology of oral submucous fibrosis: A review. Int J Oral Health Med Res. 2017;3:126-9.
- [Google Scholar]
- The chewing of betel quid and oral submucous fibrosis and anesthesia. Anesth Analg. 2005;100:1210-3.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of areca nut chewing and maximal mouth opening in school going children in Ahmedabad. Indian J Med Paediatr Oncol. 2016;37:239-341.
- [CrossRef] [PubMed] [Google Scholar]
- Oral submucous fibrosis in paediatric patients: A systematic review and protocol for management. Int J Surg Oncol. 2019;3497136 Published online
- [CrossRef] [PubMed] [Google Scholar]
- Oral submucous fibrosis: Etiology, pathogenesis, and future research. Bull World Health Organ. 1994;72:985-96.
- [Google Scholar]
- Oral submucous fibrosis: A clinic histopathological study in Chennai. Indian J Dent Res. 2007;18:106-11.
- [CrossRef] [PubMed] [Google Scholar]
- Oral submucous fibrosis: Study of 1000 cases from central India. J Oral Pathol Med. 2007;36:12-7.
- [CrossRef] [PubMed] [Google Scholar]
- Oral submucous fibrosis: A clinicopathologic review of 205 cases in Indians. Oral Maxillofac Surg. 2011;15:15-9.
- [CrossRef] [PubMed] [Google Scholar]
- Oral submucous fibrosis: Newer proposed classification with critical updates in pathogenesis and management strategies. Natl J Maxillofac Surg. 2017;8:89-94.
- [CrossRef] [PubMed] [Google Scholar]
- An outline of existing clinical classification system for oral sub mucous fibrosis. J Res Adv Dent. 2014;3:2:72-5.
- [Google Scholar]
- Frequency of precancerous conditions in 75 Malaysian oral cancer patients. Mal J Surg. 1975;1:29-38.
- [Google Scholar]
- Frequency of oral submucous fibrosis among 100 South Indians with oral cancer. Bull World Health Organ. 1965;32:750-3.
- [Google Scholar]
- Nomenclature and classification of potentially malignant disorders of the oral mucosa. J Oral Pathol Med. 2007;36:575-80.
- [CrossRef] [PubMed] [Google Scholar]
- Cancer of the buccal mucosa; a clinical study of 650 cases in Indian patients. Cancer. 1956;9:431-5.
- [CrossRef] [Google Scholar]
- Malignant transformation in 1458 patients with potentially malignant oral mucosal disorders: A follow-up study based in a Taiwanese hospital. J Oral Pathol Med. 2007;36:25-9.
- [CrossRef] [PubMed] [Google Scholar]
- Oral submucous fibrosis: An update on pathophysiology of malignant transformation. J Oral Pathol Med. 2017;46:413-7.
- [CrossRef] [Google Scholar]
- Studies on promoting activity of Taiwan betel quid ingredients in hamster buccal pouch carcinogenesis. Eur J Cancer Oral Oncol. 1996;32:343-6.
- [CrossRef] [Google Scholar]
- Elevated vimentin expression in buccal mucosal fibroblasts by arecoline in vitro as a possible pathogenesis for oral submucous fibrosis. Oral Oncol. 2002;38:425-30.
- [CrossRef] [Google Scholar]
- The upregulation of type I plasminogen activator inhibitor in oral submucous fibrosis. Oral Oncol. 2003;39:367-72.
- [CrossRef] [Google Scholar]
- Raised keratinocyte growth factor-1 expression in oral submucous fibrosis in vivo and upregulated by arecoline in human buccal mucosal fibroblasts in vitro. J Oral Pathol Med. 2005;34:100-5.
- [CrossRef] [PubMed] [Google Scholar]
- The upregulation of cystatin C in oral submucous fibrosis. Oral Oncol. 2007;43:680-5.
- [CrossRef] [PubMed] [Google Scholar]
- Review of orofacial considerations of systemic sclerosis or scleroderma with report of analysis of 3 cases. Indian J Dent. 2016;7:134-9.
- [CrossRef] [PubMed] [Google Scholar]
- Oral manifestations of autoimmune connective tissue diseases. Indian Rheumatol. 2018;13:264-72.
- [CrossRef] [Google Scholar]
- Oral submucous fibrosis and its dermatological relation. Indian Dermatol Online J. 2014;5:260-5.
- [CrossRef] [PubMed] [Google Scholar]
- Facial atrophy in oral submucous fibrosis: An association or a coincidence. Case Rep Dent. 2016;2016:1-3.
- [CrossRef] [PubMed] [Google Scholar]
- Classification criteria for systemic sclerosis: An ACREULAR collaborative initiative. Arthritis Rheum. 2013;65:2737-47.
- [Google Scholar]
- A clinical and serologic comparison of African American and Caucasian patients with systemic sclerosis. Arthritis Rheum. 2012;64:2986-94.
- [CrossRef] [PubMed] [Google Scholar]
- Neurological manifestations in systemic sclerosis (scleroderma) J Rheumatol. 1984;11:480-3.
- [Google Scholar]
- Characteristics of orofacial amyloidosis: A case series. Ann Clin Case Rep. 2016;1:1133.
- [Google Scholar]
- Unusual presentation of oral amyloidosis. Contemp Clin Dent. 2015;6:S282-4.
- [CrossRef] [PubMed] [Google Scholar]
- Local amyloidosis in the hard palate: A case report. Oral Maxillofac Surg. 2009;13:119-22.
- [CrossRef] [PubMed] [Google Scholar]
- Orofacial manifestations of haematological disorders: Anaemia and haemostatic disorders. Indian J Dent Res. 2011;22:454-61.
- [CrossRef] [PubMed] [Google Scholar]
- Role of Haemoglobin and serum iron in oral submucous fibrosis: A clinical study. Sci World J 2012:1-5.
- [CrossRef] [PubMed] [Google Scholar]
- Plummer-Vinson syndrome. A case report and literature review. Arch Intern Med. 1995;155:2008-11.
- [CrossRef] [PubMed] [Google Scholar]
- The desmoid tumor. I. Incidence, sex-, age-and anatomical distribution in the Finnish population. Am J Clin Pathol. 1982;77:665-73.
- [CrossRef] [PubMed] [Google Scholar]
- Morphologic spectrum of desmoid-type fibromatosis. Am J Clin Pathol. 2016;145:332-40.
- [CrossRef] [PubMed] [Google Scholar]
- Submucosa precedes lamina propria in initiating fibrosis in oral submucous fibrosis-evidence based on collagen histochemistry. Oral Maxillofacial Pathol J. 2020;1:1-9.
- [Google Scholar]
- Desmoid-type fibromatosis In: Fletcher CDM, Bridge JA, Hogendoorn P, Mertens F, eds. WHO Classification of Tumours of Soft Tissue and Bone. Lyon, France: IARC; 2013. p. :727-72.
- [Google Scholar]
- The fibromatoses: A clinicopathologic classification based on 140 cases. Am J Surg Pathol. 1977;1:255-70.
- [CrossRef] [PubMed] [Google Scholar]
- Argyrophilic nucleolar organizer regions in inflammatory, premalignant, and malignant oral lesions: A quantitative and qualitative assessment. Indian J Dent Res. 2008;19:141-6.
- [CrossRef] [PubMed] [Google Scholar]
- Salivary 8-hydroxy-2-deoxyguanosine, malondialdehyde, vitamin C, and vitamin E in oral pre-cancer and cancer: Diagnostic value and free radical mechanism of action. Clin Oral Invest. 2016;20:315-9.
- [CrossRef] [PubMed] [Google Scholar]
- Differential expression of salivary S100A7 in oral submucous fibrosis. Saudi Dent J. 2019;31:39-44.
- [CrossRef] [PubMed] [Google Scholar]
- Intralesional corticosteroids as a treatment for restricted mouth opening in oral submucous fibrosis. Oral Surg Oral Med Oral Pathol Oral Radiol. 2016;122:224-31.
- [CrossRef] [PubMed] [Google Scholar]
- Pentoxifylline therapy: A new adjunct in the treatment of oral submucous fibrosis. Indian J Dent Res. 2006;17:190-8.
- [CrossRef] [PubMed] [Google Scholar]
- Management of oral submucous fibrosis by two different drug regimens: A comparative study. Dent Res J (Isfahan). 2013;10:527-32.
- [Google Scholar]
- Efficacy of lycopene in the management of oral submucous fibrosis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;103:207-13.
- [CrossRef] [PubMed] [Google Scholar]
- Hyperbaric oxygen treatment reverses radiation induced profibrotic and oxidative stress responses in a rat model. Free Radic Biol Med. 2017;103:248-55.
- [CrossRef] [PubMed] [Google Scholar]
- Keloid skin scars: The influence of hyperbaric oxygenation on fibroblast growth and on the expression of messenger RNA for insulin like growth factor and for transforming growth factor. In vitro Cell Dev Biol Anim. 2011;47:421-4.
- [CrossRef] [PubMed] [Google Scholar]
- Effectiveness of Aloe Vera and Antioxidant along with Physiotherapy in the Management of Oral Submucous Fibrosis. J Contemp Dent Pract. 2016;17:78-84.
- [CrossRef] [PubMed] [Google Scholar]
- Evaluation of efficacy of aloe vera in the treatment of oral submucous fibrosis A clinical study. J Oral Pathol Med. 2017;46:50-5.
- [CrossRef] [PubMed] [Google Scholar]
- Role of honey in modern medicine. Saudi J Biol Sci. 2017;24:975-8.
- [CrossRef] [PubMed] [Google Scholar]
- Honey as a Potential Natural Antioxidant Medicine: An Insight into Its Molecular Mechanisms of Action. Oxid Med Cell Longev. 2018;2018:8367846.
- [CrossRef] [PubMed] [Google Scholar]
- Honey in oral diseases: Ayurvedic and unani perspective. J Apither. 2016;1:55-6.
- [CrossRef] [Google Scholar]
- A systematic review of various treatment modalities for oral submucous fibrosis. J Adv Clin Res Insights. 2014;2:64-72.
- [CrossRef] [Google Scholar]
- An approach to management of oral submucous fibrosis: Current status and review of literature. Int J Curr Res. 2014;6:10598-604.
- [Google Scholar]
- Treatment outcome of diode laser for symptomatic relief in oral submucous fibrosis: A pilot study. Adv Hum Biol. 2016;6:37-41.
- [Google Scholar]
- Treatment outcomes of laser therapy in oral submucous fibrosis-a systematic review. J Oral Biol Craniofac Res. 2020;10:253-8.
- [CrossRef] [PubMed] [Google Scholar]
- Autologous bone marrow concentrate (mononuclear stem cell) therapy in treatment of oral submucous fibrosis. J Ind Acad Oral Med Rad. 2013;25:1-4.
- [CrossRef] [Google Scholar]






