Translate this page into:
Pityriasis lichenoides et varioliformis acuta following measles rubella vaccine
Correspondence Address:
Veeranna Shastry
Department of Dermatology, Venereology and Leprosy, JSS Medical College, Mysore - 570 004, Karnataka
India
| How to cite this article: Shastry V, Ranugha PS, Rangappa V, Sanjaykumar P. Pityriasis lichenoides et varioliformis acuta following measles rubella vaccine. Indian J Dermatol Venereol Leprol 2020;86:398-400 |
Pityriasis lichenoides represents a unique group of inflammatory skin disorders that include pityriasis lichenoides et varioliformis acuta, febrile ulceronecrotic Mucha–Habermann disease (a severe variant of pityriasis lichenoides et varioliformis acuta) and pityriasis lichenoides chronica.[1] Pityriasis lichenoides et varioliformis acuta is a cutaneous disorder characterized by crops of erythematous macules and papules on the trunk and flexural areas of the extremities. Reddish-brown crusted areas gradually develop and result in varioliform scarring. The etiology remains unknown. It is speculated to be an inflammatory reaction triggered by certain infectious agents, an inflammatory response secondary to T-cell dyscrasia or an immune complex-mediated hypersensitivity.[2] Few reports of pityriasis lichenoides et varioliformis acuta secondary to measles, mumps and rubella, influenza, tetanus and measles rubella vaccine have been described.[3],[4],[5],[6] Here, we describe a case of pityriasis lichenoides et varioliformis acuta following measles rubella vaccine.
A 6-year-old boy presented to the dermatology department of JSS Medical college, Mysuru with a history of reddish raised lesions over the trunk and extremities for 8 days. Initially, the lesions were asymptomatic, subsequently becoming itchy. There was no fever or constitutional symptoms. He had received measles rubella vaccine 7 days prior to the onset of lesions. He was not on any medications, and his previous vaccinations were uneventful.
On cutaneous examination, multiple, discrete erythematous papules, some with crusting, were seen bilaterally over the trunk, limbs and face. There were a few erosions over the abdomen and legs [Figure - 1] and [Figure - 2]. There were no mucosal lesions or lymphadenopathy. Systemic examination was normal.
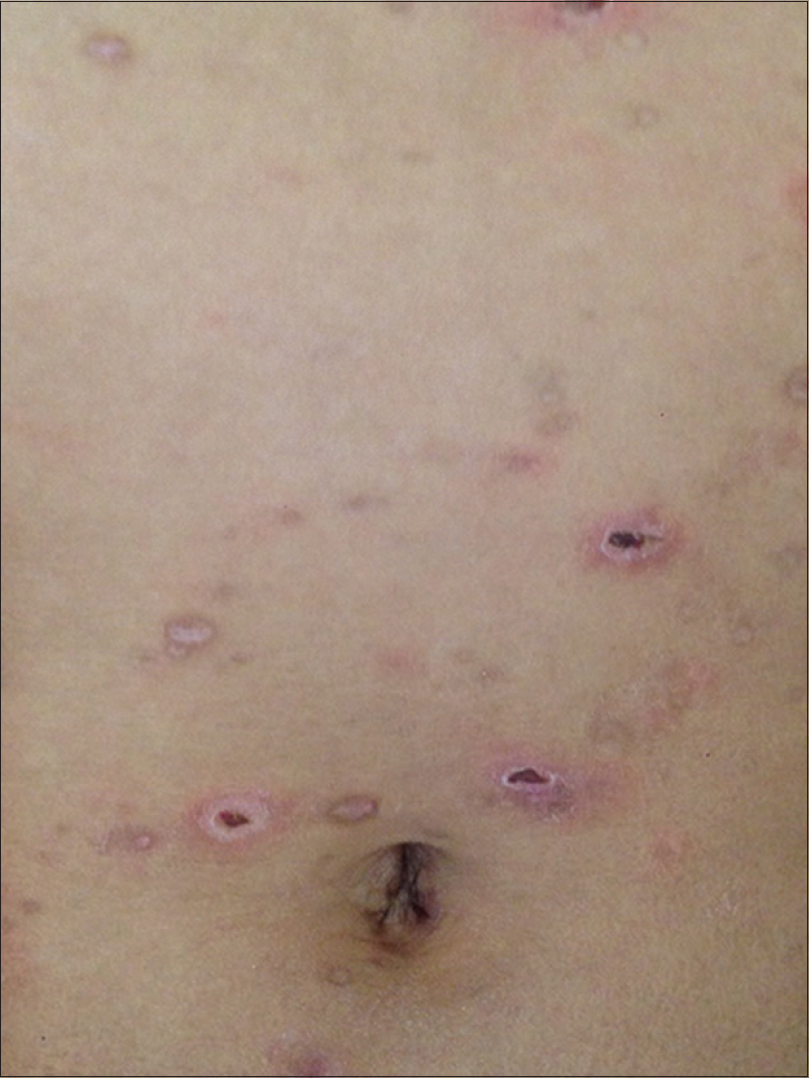 |
| Figure 1: Multiple erythematous papules with central crusting seen over the trunk |
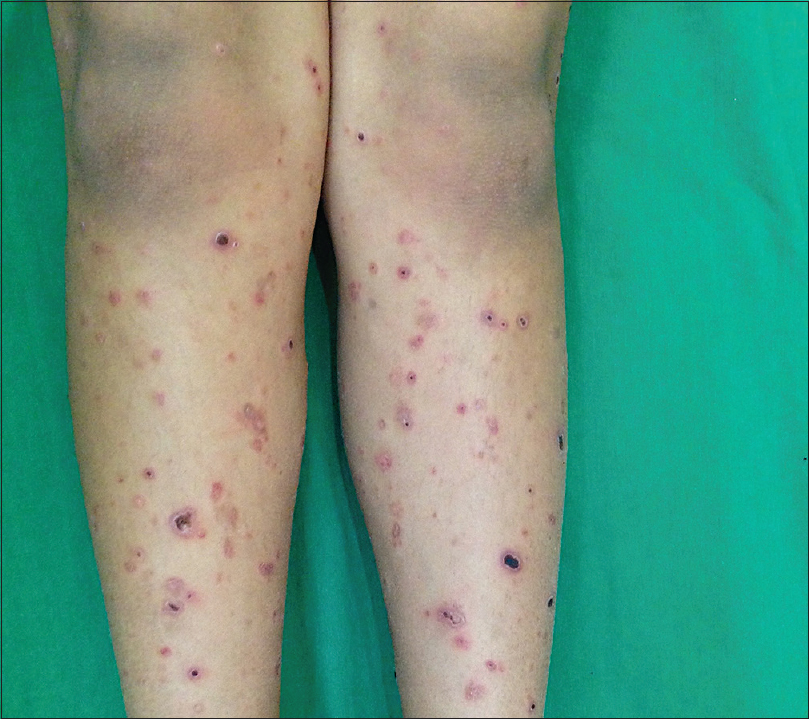 |
| Figure 2: Multiple erythematous papules with central crusting over the legs |
Based on the above findings a differential diagnosis of pityriasis lichenoides et varioliformis acuta, atypical pityriasis rosea, papulonecrotic tuberculid, varicella and lymphomatoid papulosis was considered.
Routine blood investigations were normal. Skin biopsy from an erythematous papule on the trunk showed epidermis displaying parakeratosis, necrosis of keratinocytes and basal cell vacuolar degeneration. Superficial dermis revealed perivascular and interstitial lymphocytic infiltrate. Pigment incontinence was present [Figure - 3]. Based on the clinical and histopathological features we made a diagnosis of pityriasis lichenoides et varioliformis acuta following measles rubella vaccination.
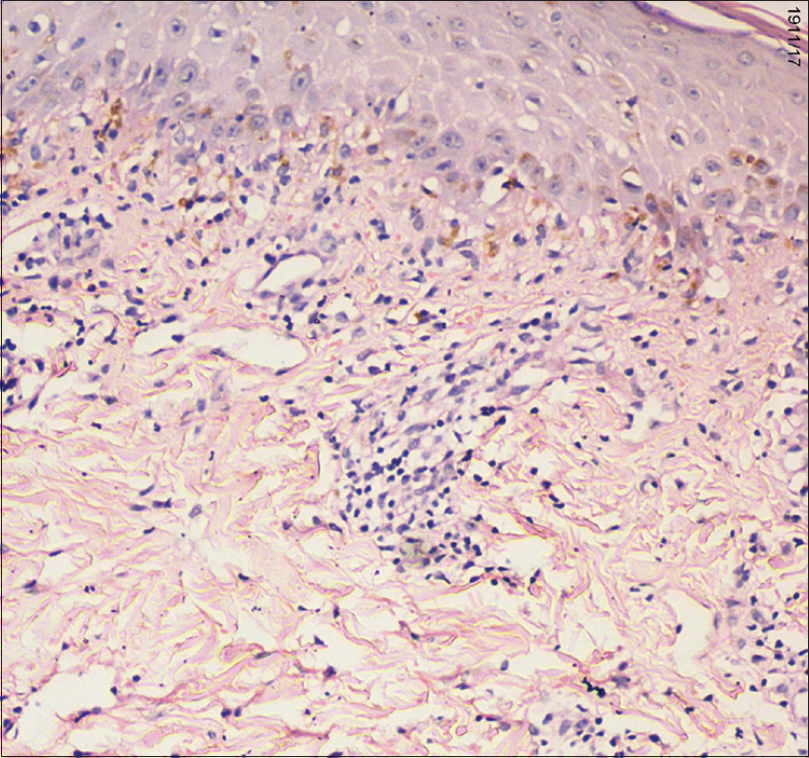 |
| Figure 3: Epidermis showing basal cell vacuolar degeneration with pigment incontinence in the dermis (hematoxylin and eosin, ×400) |
The child was started on oral azithromycin 250 mg daily and a short course of low-dose systemic steroids mention the dose of steroid for 2 weeks. Narrow-band ultraviolet-B phototherapy at 200 mJ/cm[2] thrice weekly was also given, which was gradually increased by 50 mJ/cm2 every third session until a final dose of 800 mJ/cm2. New lesions stopped appearing within 2–3 weeks of therapy and resolution was seen by 2 months. Narrow-band ultraviolet-B was continued for another 2 weeks and then stopped. The lesions healed with pigmentation along with atrophic scarring [Figure - 4].
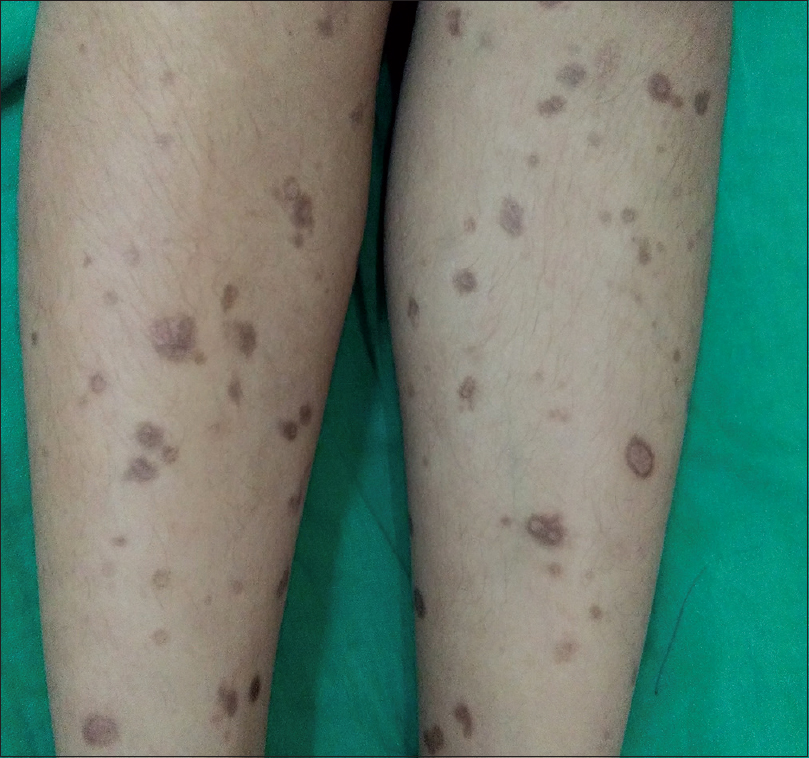 |
| Figure 4: Pigmented atrophic scarring over the legs |
Pityriasis lichenoides was first described by Neisser and Jadassohn.[5] The underlying pathogenic mechanism in pityriasis lichenoides is unknown. High antibody titres to various infectious agents such as HIV, hepatitis C, cytomegalovirus, Epstein–Barr virus, Toxoplasma gondii and Herpes simplex virus have been observed concurrently with the disease, but no definitive relation has been established.[5] Pityriasis lichenoides et varioliformis acuta following vaccination is rare. Isolated cases with tetanus, measles, measles, mumps, and rubella, measles rubella, influenza and H1N1 vaccination have been described previously.[6]
The measles rubella vaccine currently used in the immunization program is a live attenuated vaccine. The Ministry of Health and Family Welfare of India has initiated this mass vaccination campaign in the age group of 9 months to 15 years in a phased manner across the nation on February 2017. Adverse effects reported are minor and transient (local reaction, fever, rash) with no long-term sequelae. One serious but extremely rare adverse effect of measles rubella vaccine is anaphylaxis following vaccination.[7]
History of recent vaccination and typical clinical and histopathological features aided the diagnosis. Naranjo adverse drug reaction probability scale was used for causality assessment and showed a possible correlation with measles rubella vaccine and the adverse event. Measles rubella vaccine as a cause of pityriasis lichenoides et varioliformis acuta in our case was based on temporal correlation and previous similar reports of association with vaccination. Rechallenge could not be done. Some authors have proposed that the measles component of measles, mumps and rubella vaccine triggers a lymphoproliferative reaction in the skin presenting as pityriasis lichenoides et varioliformis acuta, with the virus acting as an epidermal antigenic target.[3] Whether measles or rubella component of the vaccine triggered the eruption in our case is difficult to confirm.
There are only few reports of vaccination triggered pityriasis lichenoides et varioliformis acuta worldwide. Measles rubella vaccine induced pityriasis lichenoides et varioliformis acuta has been reported from Japan.[6] Dermatologists and physicians should be aware of this rare complication associated with measles rubella vaccination.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patient has given his consent for his images and other clinical information to be reported in the journal. The patient understand that name and initials will not be published and due efforts will be made to conceal identity, but anonymity cannot be guaranteed.
Acknowledgement
Dr.Vijaya B, Department of Pathology, JSS Medical College,,
Mysore, Karnataka, India.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. |
Bowers S, Warshaw EM. Pityriasis lichenoides and its subtypes. J Am Acad Dermatol 2006;55:557-72.
[Google Scholar]
|
| 2. |
Fernandes NF, Rozdeba PJ, Schwartz RA, Kihiczak G, Lambert WC. Pityriasis lichenoides et varioliformis acuta: A disease spectrum. Int J Dermatol 2010;49:257-61.
[Google Scholar]
|
| 3. |
Gunatheesan S, Ferguson J, Moosa Y. Pityriasis lichenoides et varioliformis acuta: A rare association with the measles, mumps and rubella vaccine. Australas J Dermatol 2012;53:e76-8.
[Google Scholar]
|
| 4. |
Castro BA, Pereira JM, Meyer RL, Trindade FM, Pedrosa MS, Piancastelli AC. Pityriasis lichenoides et varioliformis acuta after influenza vaccine. An Bras Dermatol 2015;90:181-4.
[Google Scholar]
|
| 5. |
Baykal L, Arica DA, Yayl S, Altun E, Bahad S. Pityriasis lichenoides et varioliformis acuta; Association with tetanus vaccination. J Clin Case Rep 2015;5:518.
[Google Scholar]
|
| 6. |
Moriwaki M, Tanaka A, Tanaka M, Takahagi S, Hide M. Pityriasis lichenoides et varioliformis acuta after vaccination. J Cutan Immunol Allergy 2018;1:37-8.
[Google Scholar]
|
| 7. |
Introduction to measles-rubella vaccine (Campaign and Routine Immunization). Available from: http://www.searo.who.int/india/topics/ measles/measles_rubella_vaccine_guidelines.pdf?ua=1. [Last accessed on 2018 Jan 16].
[Google Scholar]
|
Fulltext Views
4,443
PDF downloads
1,304





