Translate this page into:
Rosacea in skin of color: A comprehensive review
2 Department of Dermatology, College of Medicine and Sagore Dutta Hospital, Kolkata, West Bengal, India
3 Department of Dermatology, Amrita Institute of Medical Sciences, Kochi, Kerala, India
Correspondence Address:
Rashmi Sarkar
Department of Dermatology, Maulana Azad Medical College, New Delhi
India
| How to cite this article: Sarkar R, Podder I, Jagadeesan S. Rosacea in skin of color: A comprehensive review. Indian J Dermatol Venereol Leprol 2020;86:611-621 |
Abstract
Rosacea is a relatively common inflammatory dermatosis in persons with fair skin. It is uncommonly reported in people with skin of color (darker skin tone; Fitzpatrick skin types IV, V or VI). Apart from reduced incidence, underreporting due to decreased awareness might also be a probable explanation. Rosacea commonly presents with telangiectasias and persistent facial erythema on the sun-exposed parts, which can be distressing to the patient and affect the quality of life. The diagnosis is made clinically, in the absence of any confirmatory investigation. Several treatment modalities have been employed to date with varying results. Light-based therapies should be used cautiously in the colored skin to avoid distressing pigmentation. This article focuses on the pathogenesis, clinical features, treatment recommendations and other aspects of this uncommon disorder along with a review of the literature.
Introduction
Rosacea is an inflammatory facial dermatosis with varied clinical features; the commonest being persistent facial erythema, chiefly affecting the facial convexities along with some vascular changes, inflammatory lesions, hypertrophic changes or ocular involvement. However, the clinicopathological features and pathogenesis remain unclear without any confirmatory laboratory test. The diagnosis of this condition is more difficult in people with colored skin (darker skin tone; Fitzpatrick skin types IV, V or VI), as the disease characteristics are less well-defined with hallmark erythema being indistinguishable. As a result, this condition remains underreported in the skin of color, although its incidence is increasing in this group. We have tried to present a comprehensive review of this disorder, especially in the colored skin; to aid in its early diagnosis and appropriate treatment.
Epidemiology
The exact prevalence of rosacea remains unknown; fair-skinned individuals being affected more commonly. A review of population studies has revealed a widely varying prevalence ranging from 0.1% in the Faroe Islands to 5% in Russia to 12.3% in Germany to 10% in Sweden and 22% in Estonia. These studies mainly included patients with phototypes 1–3.[1] It has been occasionally reported in patients with skin of color; however, epidemiologic data are scarce.
Although rosacea is not uncommon in the colored skin, typical clinical features are hard to detect, thus, leading to underdiagnosis and underestimation of the prevalence of rosacea among people with colored skin.[2]
In India, rosacea, especially steroid-induced one is being increasingly reported.[3] One Indian study reported that rosacea/rhinophyma accounted for almost 0.5% of all dermatology consultations indicating that it is more common than previously thought.[4] A study conducted among Saudi females with rosacea reported its highest prevalence in skin type 6 (42%), followed by skin type 4 (40%) and skin type 5 (18%).[5] In China, Taiwan, Korea and Japan, rosacea is a common disease.[6] In Tunisia, Khaled et al.[7] reported 224 cases of rosacea in a predominantly dark-skinned population, with a hospital prevalence of 0.2%. However, a study from Ghana did not report any case of rosacea.[8]
The onset is usually in middle age (30–50 years) though any age, including children, may be affected. Sexual predilection is absent; however, men often develop more severe disease with a higher risk of developing rhinophyma.
Pathogenesis
The exact pathogenesis is still unclear. A genetic component, alterations in the innate immune mechanisms, defective barrier function of skin, ultraviolet radiation exposure, microorganisms and neurological dysfunction have all been postulated to play a role.
Genetic factors
Though the higher incidence of rosacea in people of North European and Celtic descent and a familial occurrence suggests genetic predisposition, the same was not established until recently. A genome-wide association study conducted by Chang et al. identified two significant single-nucleotide polymorphisms associated with rosacea in the European population, one of them being located between the genes HLA-DRA and BTNL2.[6] Furthermore, increased expression of both HLA-DRA and BTNL2 proteins have been detected by immunohistochemistry. This indicates the biological relevance and association of these proteins supporting the inflammatory nature of this disease.
Another study conducted by Aldrich et al. on a cohort of identical and fraternal twins with rosacea stressed the role of both genetics and environment in determining the severity of this condition.[9]
Alterations in innate immunity
Elevated levels of cathelicidin and kallikrein 5 (serine proteases responsible for cleaving cathelicidin into its active form- LL 37) have been detected in skin sections of patients with rosacea. These peptides regulate and promote leukocyte chemotaxis, angiogenesis and expression of extracellular matrix components. In rosacea, toll-like receptor (TLR)-2 expression is also altered, which increases the susceptibility to various innate immune stimuli, ultimately resulting in increased production of cathelicidin and kallikrein.[10] Moreover, matrix metalloproteinases show increased expression in lesional skin and have been indirectly shown to enhance the activity of kallikrein 5, which may, in turn, lead to increased levels of IL-37.
Lymphatic and vascular markers like vascular endothelial growth factor, CD31 and lymphatic endothelium marker D2-40 have been found to be elevated in rosacea skin suggesting the role of vascular and lymphatic endothelial cells. Reactive oxygen species are also noted to be involved in the pathogenesis.[11] A study examining skin biopsy samples from healthy individuals and rosacea patients found higher levels of reactive oxygen species in lesional skin of rosacea patients.[12]
Microbes
Demodex folliculorum[13] and Helicobacter pylori[14] are the two main microorganisms implicated in the pathology of rosacea. However, their exact role is yet unclear.
D. folliculorum commonly resides in the sebaceous follicles of healthy skin and has been implicated to trigger rosacea due to inflammation within the pilosebaceous follicles. A higher density of these mites has been found on the facial skin of rosacea patients compared to healthy controls. These mites are thought to release chitin, which can activate TLR2 resulting in increased protease activity. Moreover, these mites have been found to harbor Bacteroides oleronius, a nonmotile, Gram-negative, endospore-forming bacterium which is thought to trigger the production of matrix metalloproteinase-9, tumor necrosis factor and interleukin-8.
A normal skin commensal, Staphylococcus epidermidis, has been cultured from lesional rosacea skin and these isolates have been shown to secrete virulence factors, not found on normal or uninvolved skin. These virulence factors may trigger the innate immune system to produce aberrant antimicrobial peptides.
The role played by H. pylori and other intestinal bacteria is still unclear. Numerous studies have reported an increased prevalence of H. pylori seropositivity in patients with rosacea, while others have failed to demonstrate the same. Increased number of H. pylori strains in rosacea has tested positive for the cytotoxin-associated gene 1(CAG-1), an H. pylori virulence factor; however, the exact pathomechanism remains obscure. The studies favoring the eradication of the bacteria for clinical improvement have also failed to deliver conclusive evidence.
Defective skin barrier
The skin of patients with rosacea has been shown to have decreased barrier function. The skin of affected patients have shown increased transepidermal water loss along with reduced hydration. The increased levels of a serine protease in rosacea probably worsen the barrier function.[12]
Ultraviolet radiation
Ultraviolet radiation is a known precipitating factor for rosacea. Both ultraviolet A radiation through its action on matrix metalloproteinases and collagen denaturation and ultraviolet B radiation with its effects of increasing the secretion of fibroblast growth factor 2 and vascular endothelial growth factor 2 may contribute to the hypervascularity of the skin in rosacea.[12] Ultraviolet light radiation increases reactive oxygen species in the skin, which can signal through TLR2 to propagate the kallikrein 5/cathelicidin inflammatory cascade.[15]
Neurogenic dysregulation
Rosacea skin has a significantly lower heat pain threshold than normal skin. Within the transient receptor potential family of cation channels, two subfamilies could possibly contribute to the pathogenic mechanisms of rosacea. Four vanilloid receptors (transient receptor potential [TRPV] 1–4) and one ankyrin receptor (TRPA1) have been found to be active in rosacea. The different transient receptor potentials have an impact on a wide range of functions including local immunity, vasoregulation, nociception and epidermal barrier integrity; however, their signaling pathways are incompletely understood.[16]
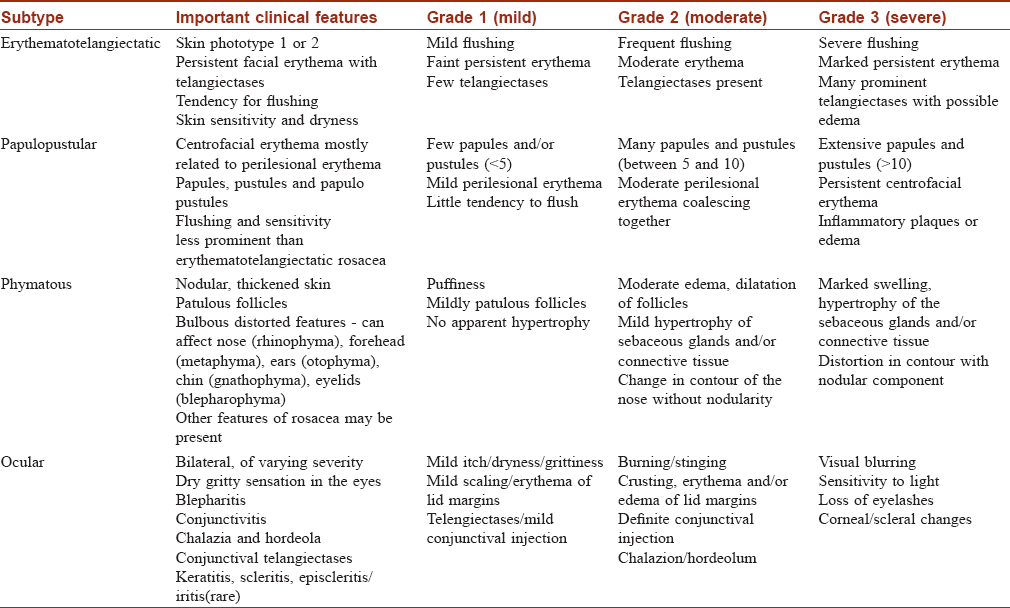
Classification and Diagnostic Criteria of Rosacea
In 2004, the National Rosacea Society Expert Committee devised a classification system to help standardize the diagnosis of rosacea.[17] The committee classified the primary and secondary characteristics of rosacea [Table - 1], with the presence of more than one primary feature being indicative of the diagnosis. Secondary features may or may not be present. There are no specific tests to diagnose rosacea to date.

According to this classification, there are four rosacea subtypes and their further variants depending on the primary and secondary features: erythematotelangiectatic rosacea [Figure - 1], papulopustular rosacea [Figure - 2], phymatous rosacea and ocular rosacea. The clinical features of the subtypes with their severity grading are tabulated in [Table - 2]. This new classification has aimed to provide a framework that will serve as a diagnostic guide for the clinicians and enable better standardization in terms of research and comparison of data.
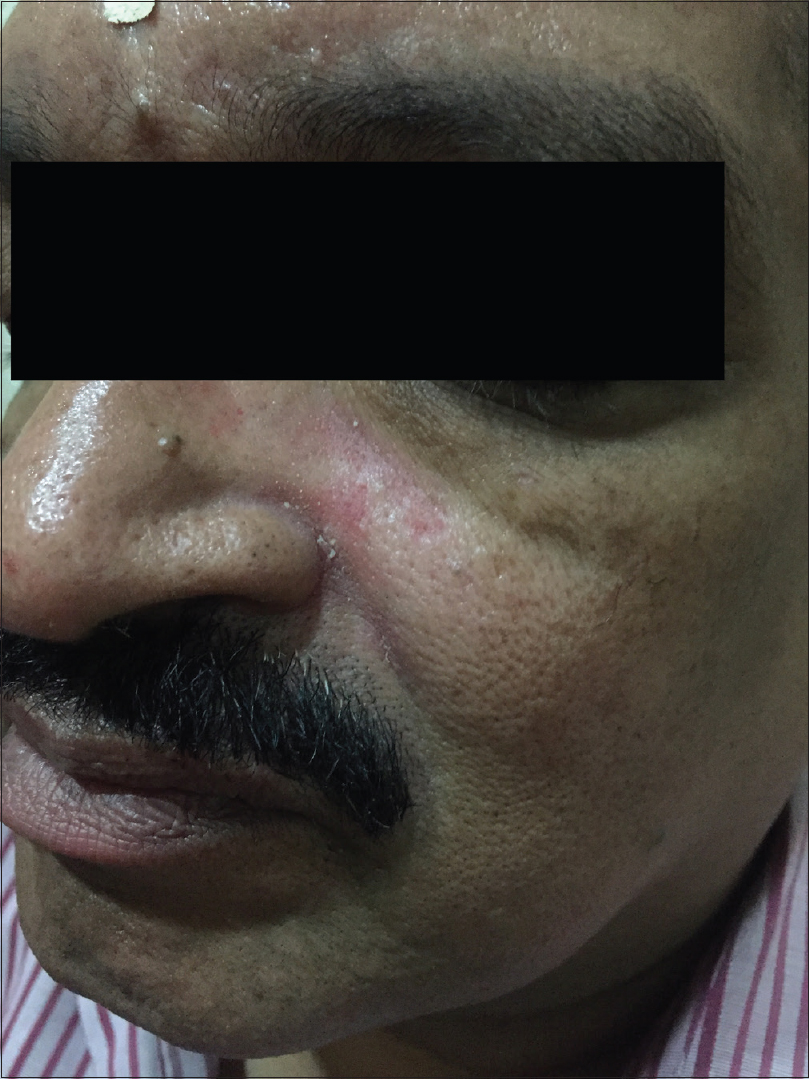 |
| Figure 1: Erythematotelangiectatic rosacea |
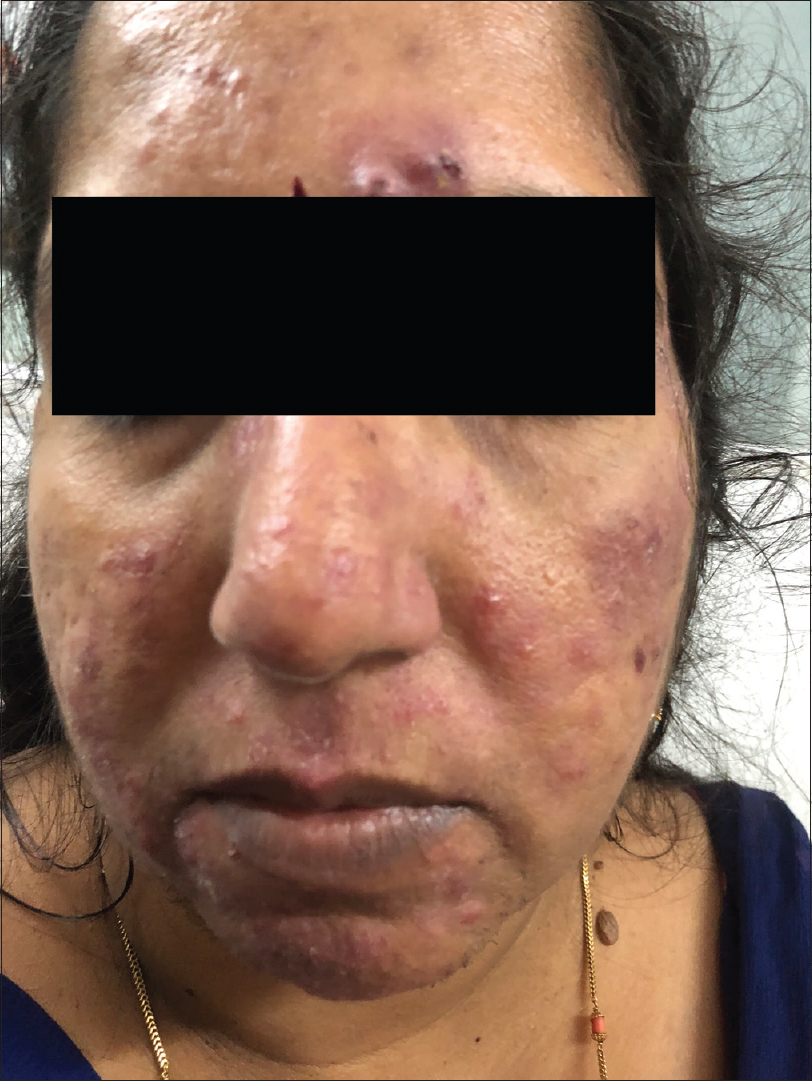 |
| Figure 2: Papulopustular rosacea |
Clinical picture in patients with skin of color
In patients of skin phototype ≥4, erythema is often difficult to ascertain, creating diagnostic difficulties as the visual characteristics of the disease are yet to be defined fully.
In a study done on Saudi women (skin phototypes 3, 4 and 5), sun exposure was identified to aggravate the disease in 72% of the patients. Pruritus was the most prominent symptom in contrast to burning or stinging as seen in Europeans or white Americans. Severe erythematotelengiectatic rosacea and papulopustular rosacea were the commonest varieties; however, severity was not related to skin type. Extrafacial sites were more commonly involved (14%). Phymatous and granulomatous types were not seen. Post-inflammatory hyperpigmentation or hypopigmentation was surprisingly not reported in any of the patients.[5]
Khaled et al. reported sun exposure to be a major triggering factor (64%) along with other thermal stimuli (25%) in Tunisian patients.[7] The leading clinical subtype was papulopustular rosacea (69%), followed by erythematotelangiectatic type (12%) and rhinophyma (3.7%)
A study from South Korea which included 168 Korean patients of skin phototypes 2, 3, 4 and 5 (but predominantly 3) noted that 50.6% and 96.4% of patients were diagnosed with papulopustular rosacea and erythematotelangiectatic subtype, respectively. The other types were rare and the degree of sun exposure was noted to have a significant correlation with the occurrence and severity of the erythematotelangiectatic variant.[18]
A study conducted in China among 586 patients evaluated the differences in epidemiological and clinical characteristics among rosacea patients according to different facial sites- full-face, cheeks, nose or perioral involvement. There were 471 (80.4%), 49 (8.4%), 52 (8.9%) and 14 (2.4%) cases in the full-face, cheek, nasal and perioral groups, respectively. Compared with the full-face group, the nasal group had more severe phymatous changes and less severe self-reported and dermatologist-evaluated grading of symptoms.[19]
In Japan, rhinophyma is uncommon when compared to the Western population and affects almost exclusively the lower half of the nose.[20]
An Indian study suggested that rosacea and rhinophyma may not be as common in the Indian population as compared to other Asian countries, however epidemiological data is scarce.[4] Steroid-induced rosacea is also being increasingly reported in India.[3]
Another study from Seoul, Korea assessed the prognosis of 234 rosacea patients according to clinical subtype. A favorable prognosis was observed in the papulopustular subtype without centrofacial erythema than with other subtypes.[18]
Rosacea is reported less commonly in the skin of color, probably because darker skin types are less prone to photodamage and additionally flushing and telangiectasia are harder to detect clinically. However, many recent studies from this part of the world including India have detected its rising prevalence in recent times, although this condition remains to be underreported. So, the authors feel that erythema and secondary symptoms like burning and stinging sensation may be more important diagnostic tools for rosacea in skin of color, compared to traditional flushing and telangiectasia, which are more evident in the lighter skin phenotypes.
Rosacea and Systemic Disease
Rosacea has a negative impact on the quality of life by inducing stigma and anxiety in patients. A nationwide cohort study by Egeberg et al. explored a possible association between rosacea and diseases of the gastrointestinal tract and found them more likely to suffer from autoimmune disorders such as Crohn's disease or ulcerative colitis.[21] A recent nation-wide cohort study from Taiwan proposed its link with inflammatory bowel disease.[22] A case-control analysis based on a large U.K. population also reported a strong association of inflammatory bowel disease with rosacea.[23] In a study from Taiwan, hypertension, dyslipidemia and coronary artery disease were found to be significantly associated with rosacea (males > females).[24] Other reported comorbidities include dementia, Alzheimer's disease and Parkinson's disease.[25]
Treatment
The treatment of rosacea is challenging for dermatologists because of multifactorial and complex etiopathogenesis. Treatment can be categorized into general and specific measures: the latter including topical, systemic and physical/light-based modalities. The different evidence-based treatment modalities of rosacea have been discussed here along with a relevant review of literature, highlighting their level of evidence and grade of recommendation.
General measures/non-pharmacologic treatments
General measures are aimed at minimizing the effect of different triggering factors to hasten improvement. The important general measures have been listed below:
Patient education
Proper patient education regarding the benign nature of the disease is essential to achieve psychological well-being. Patients must be counseled to avoid specific triggering factors like strong winds, temperature variations, exercise, spicy foods, alcohol, physical and/or emotional stress.[1] Proper drug history should be elicited to rule out aggravating drugs like niacin and topical corticosteroids.[26] These medications should be stopped at the earliest to avoid disease worsening.
Photoprotection/sunscreens
Exposure to sunlight/ultraviolet rays resulting in photodamaged skin is a well-documented triggering factor for the occurrence and/or worsening of rosacea; thus, photoprotection measures are an important component of its treatment.[1] Apart from physical measures viz. covered clothing, shade-seeking behavior, umbrellas and shades, broad-spectrum sunscreens (having sun protection factor of 30+) are recommended to achieve effective photoprotection against ultraviolet B and ultraviolet A radiation. Recent studies have also highlighted an anti-oxidant property of sunscreens by reducing LL-37 and subsequent free-radical generation.[26] However, the conventional sticky sunscreens may worsen the condition by increasing the warmth of skin by converting ultraviolet radiation into heat. Inorganic sunscreens containing zinc oxide and titanium oxide are free from this effect and thus preferred.[27] The use of sunscreens is also to be advised in women staying indoors before they go near the hot ovens for preparing food and other household chores.
Facial cleansers
Facial cleansers are needed to remove excess sebum, exfoliated keratinocytes, environmental debris and some microorganisms (Demodex, pityrosporum, etc.) to maintain a healthy and clean biofilm. However, mild cleansers must be prescribed to preserve the epidermal barrier. Selection of appropriate cleanser is essential viz. soaps, syndets and lipid-free cleansers for oily, normal and dry skins, respectively; to achieve optimum results.[27] Ideally, cleansers should be used twice daily followed by rinsing with water to clean the biofilm maintaining the normal skin barrier. Nasolabial areas, eyebrows and beard areas must be cleaned meticulously, as these sites have high sebum concentration.
Facial moisturizers
Facial moisturizers may be helpful when patients have normal or dry skin. These substances act by helping in barrier repair and minimizing transepidermal water loss.[26] However, they should not promote bacterial overgrowth (plant and animal oils like coconut oil, olive, argan and sunflower oils) or occlude the sweat ducts to cause miliaria, acneiform eruptions and facial irritation. Broadly three classes of moisturizers are available—occlusive (oily substances that retard transepidermal water loss), humectants (substances that attract water to the epidermis from underlying dermis and environment) and hydrocolloids (large molecules that cover the epidermis and minimize transepidermal water loss). A mixture of occlusive and humectant properties provide the best results in these patients.[27]
Other cosmeceuticals
Several other cosmeceuticals or botanicals are being used in rosacea because of their anti-oxidant and vasomotor properties to reduce the associated inflammation, irritation and facial redness.[27] Some of the botanicals being currently used in rosacea have been tabulated below [Table - 3].

Camouflaging agents
Several colored cosmetics are being increasingly used in persistent cases to mask the red appearance of skin (green colored cosmetics when applied on reddish skin produces a brownish tinge which mimics the normal skin color). Often these ingredients have moisturizers as vehicles to produce optimum results. Glycyrrhizin compounds, biotin and vitamins are also used as anti-redness creams.
Specific measures
Pharmacologic treatments and several procedural interventions constitute specific therapeutic measures. Pharmacologic treatments include several topical and systemic agents while different types of lasers, peels, etc., constitute the physical modalities. All these modalities have been briefly discussed below.
Topical agents
Several topical agents are effective in rosacea, as discussed below [Table - 4]. They are often combined with oral therapy to induce remission, later used alone for maintenance.

USFDA approved topical agents
Topical azelaic acid
Level of Evidence: 1++; Grade of Recommendation: A
Azelaic acid, a naturally occurring dicarboxylic acid, is a United States Food and Drugs Administration-approved topical treatment of rosacea. Recently, it has been recommended as the first-line treatment for mild to moderate inflammatory or papulopustular rosacea by the ROSacea COnsensus (ROSCO) panel of experts.[28] It acts by reducing the levels of kallikrein 5 and cathelicidin; both being important pro-inflammatory cytokines aggravating rosacea.[28] A 2011 Cochrane review showed significant improvement with azelaic acid over placebo in four randomized control trials and slight superiority over topical metronidazole in two randomized control trials.[29] Often this agent is combined with oral agents at the outset, later continued alone to maintain remission for as long as 6 months.[26] This medication is available as 15% and 20% gel/cream. Both the strengths are effective treatment options for rosacea except for the occurrence of mild irritation, especially in the colored skin. So, in the latter, 15% cream is more favorable.
Topical metronidazole
Level of Evidence: 1++; Grade of Recommendation: A
Topical metronidazole has been used for the treatment of rosacea for the last six decades. It acts by reducing the rate of ultraviolet-induced reactive oxygen species generation and also inactivating the existing reactive oxygen species.[30] A Cochrane review has demonstrated the superiority of this medication over placebo, used twice daily, in both “self-assessment by the patient” (three studies; odds ratio: 5.96; 95% confidence interval: 2.95–12.06) and physician global assessment (three studies; odds ratio: 7.01; 95% confidence interval: 3.56–13.81).[26] Two concentrations are available (0.75% and 1%); both being well-tolerated and efficacious. Dahl et al. also showed that topical metronidazole increases the remission free period following treatment.[31]
Topical sodium sulfacetamide
Level of Evidence: 1+; Grade of Recommendation: A
Topical sulfacetamide/sulfur preparations are available in several vehicles viz. lotions, creams and cleansers. The lotion/cleanser forms are used widely, and several studies, including two randomized control trials, have depicted its efficacy, especially when combined with other topical modalities.[26] This drug probably acts by exerting anti-inflammatory action, although the exact mechanism of action is unclear. Dryness, irritation and erythema may occur at the application site initially but gradually improve with time. In patients with concomitant seborrheic dermatitis, sulfur preparations are more useful.[26]
Topical α-adrenergic agonists
Level of Evidence 1+, Grade of Recommendation A
Topical α-adrenergic agonists have been recently approved by the United States Food and Drugs Administration for the treatment of rosacea associated persistent erythema. These drugs cause vasoconstriction, thus reducing the erythema. However, telangiectatic blood vessels are not affected as they lack the required receptor, paradoxically they become more prominent once background erythema is minimized. Clinical trials including four multicenter, vehicle-controlled randomized controlled trials have shown reduced erythema 30 min to 6 h following application, however, this erythema recurs within 1 month following drug discontinuation.[32],[33] Overall, this drug is safe and well-tolerated apart from sporadic cases of rebound erythema.[26] Currently, an α2-adrenergic agonist (brimonidine tartrate 0.5%) gel has been approved for once-daily usage. Another molecule, oxymetazoline (α1-adrenergic agonist), which has shown favorable treatment response is currently undergoing phase III clinical trials.[26] This drug may be used in conjunction with other therapeutic options to obtain gratifying results in persistent facial erythema.
Topical agents not approved by the United States Food and Drugs Administration
There are several other topical agents like retinoids, calcineurin inhibitors, benzoyl peroxide, permethrin and ivermectin which have shown encouraging results in sporadic case series/reports/studies but yet not approved by the United States Food and Drugs Administration. Some of these agents are briefly discussed below:
Topical ivermectin
Level of Evidence 1+, Grade of Recommendation A
Ivermectin, a macrocyclic lactone derivative with dual anti-inflammatory and anti-parasitic properties is one of the most studied medications for rosacea. It exerts its effect by acting against the Demodex mites, one of the key factors in the pathogenesis of rosacea. It also exerts anti-inflammatory effects by reducing neutrophilic migration and phagocytosis, downregulating inflammatory cytokines like IL-1b and TNF-alpha and upregulating the anti-inflammatory cytokine interleukin-10.[34] It is available as 1% cream and once-daily usage is recommended. Recently several studies (3 clinical trials) have demonstrated increased efficacy of topical ivermectin in comparison to the placebo, topical azelaic acid and topical metronidazole.[34],[35] Topical ivermectin is generally well-tolerated with a favorable safety profile except for sporadic reports of mild dryness, burning and stinging sensation, which gradually improve over time.[34]
Topical permethrin
Level of Evidence 1+, Grade of Recommendation A
Topical permethrin cream (5%) has been suggested by some authors, owing to its anti-parasitic action against Demodex mites. A randomized control trial (pilot study) has shown significant superiority to placebo, however, no added benefit has been noted on topical metronidazole.[26]
Topical retinoids
Level of Evidence 1−, Grade of Recommendation B
Topical retinoids are known to repair photodamaged skin by promoting connective tissue repair. So, these agents have been tried in rosacea to counter ultraviolet-induced photodamage. Besides, it may also exert an anti-inflammatory effect as it has been shown to downregulate TLR2 receptor expression in-vitro.[26] Two small controlled trials have shown better response in inflammatory lesions (reduced lesion count); however, no added benefit over placebo has been reported.[36] Irritation with these agents, particularly in the colored skin is to be considered carefully.
Topical calcineurin inhibitors
Level of Evidence 2+, Grade of Recommendation: C
Topical calcineurin inhibitors viz. tacrolimus and pimecrolimus have been tried by some authors in treating rosacea because of their ability to reduce T-cell activation and subsequent release of pro-inflammatory cytokines like interleukin-2. A randomized open-label study (48 patients) demonstrated efficacy similar to topical metronidazole, whereas another randomized control trial did not find it superior to placebo.[36] Paradoxically, some authors have reported the development of rosacea-like and granulomatous dermatitis with continuous use of topical calcineurin inhibitors.[37]
Topical benzoyl peroxide
Level of Evidence 2−, Grade of Recommendation D
Topical benzoyl peroxide (2.5% and 5% gel) has also been studied because of its anti-inflammatory action. A solitary case-control trial reported 5% benzoyl peroxide to be superior to placebo; however, 23% of patients dropped out of the study without completing it.[37]
Topical clindamycin
Level of Evidence 1+, Grade of Recommendation A
Two randomized control trials have shown the efficacy of topical clindamycin (1%); the first one compared it with systemic tetracycline while the later compared it with topical tretinoin (0.025%), both for 3 months. Significant clinical improvements in inflammatory and telangiectatic components were noted in both the studies. The side effect was mild in the form of facial scaling, which did not lead to discontinuation.[37]
Systemic agents
Several systemic agents have been tried in rosacea to provide a synergistic effect along with the other modalities, as given below [Table - 5].

Tetracyclines
Level of Evidence 1++, Grade of Recommendation A
Tetracyclines are the only United States Food and Drugs Administration-approved systemic therapy for rosacea. Second generation tetracyclines like doxycycline and minocycline are preferred, because of their better safety profile. They act in papulopustular/inflammatory rosacea by downregulating pro-inflammatory cytokines like kallikrein 5 and cathelicidin resulting in decreased neutrophil chemotaxis, reduced generation of toxic reactive oxygen species and inhibition of nitric oxide-mediated vasodilatation.[26] Even sub-antimicrobial anti-inflammatory dosage is effective as no bacteria is involved in the pathogenesis of rosacea. Sub-antimicrobial doses of doxycycline (40 mg per day) and minocycline (45 mg per day) have been found to be effective in papulopustular rosacea.[26],[36] The sub-antimicrobial dose is well-tolerated with fewer side effects and it also hinders the development of drug resistance.[36] Three randomized control trials have demonstrated higher efficacy of doxycycline (40 mg) over topical metronidazole or placebo.[37] A recent large, open-label, multicenter study showed significant improvement of severity and erythema in 70% patients with once-daily low-dose doxycycline monotherapy.[38] Thus, the anti-inflammatory properties of tetracyclines are beneficial in rosacea, while the antibacterial properties have a limited role at present. However, these drugs are contraindicated in pregnant or lactating women and children less than 12 years.[26]
Beta-blockers
Nonselective beta-blockers like propranolol and carvedilol have been found to reduce the incidence and severity of rosacea associated erythema in some case reports and case series.[39] These drugs block the beta-receptors on perivascular smooth muscles resulting in vasoconstriction and dramatic improvement of erythema and flushing within 2–3 weeks.[26] However, one must be aware of potential adverse effects like hypotension and bradycardia.
Isotretinoin
Level of Evidence 1+, Grade of Recommendation a (Only for Severe Varieties Like Pyoderma Faciale)
Oral isotretinoin has been used successfully in the treatment of severe, recalcitrant papulopustular rosacea (pyoderma faciale) by some authors as an off-label drug. A lower dosage (10 mg/day) has been found to be effective with minimal adverse effects.[26] Gollnick et al.[40] conducted a randomized control trial (20 patients, 12 weeks) and reported the superiority of isotretinoin to doxycycline and placebo, while an uncontrolled study (22 patients, 4 months) described improvement in 50% patients by 9 weeks.[37] It acts primarily by down-regulating monocytic TLR2 expression.[26] Side effects are tolerable in the form of dry lips and xerosis while 25% of patients may experience mild to moderate elevation of serum triglyceride levels. Notably, this drug is teratogenic and absolutely contraindicated during pregnancy; use during lactation is also to be avoided.[37]
Oral ivermectin
Level of Evidence 3, Grade of Recommendation D
A few, sporadic case reports have reported significant clinical improvement in rosacea with single or multiple doses of oral ivermectin (200 μg/kg) along with topical medications.[34] Oral ivermectin probably acts by its anti-parasitic action against Demodex mites. However, larger controlled trials are needed to establish its role.
Eradication of Helicobacter pylori
Level of Evidence 2+, Grade of Recommendation C
Although the etiologic role of H. pylori in rosacea is controversial, few sporadic studies have demonstrated the beneficial effect of helicobacter eradication regimen comprising omeprazole (40 mg/day), clarithromycin 500 mg to 1 g/day) and metronidazole (1–2 g/day), for 1–2 weeks. Two case-control studies have shown a favorable response in rosacea after 12 (76%) and 4 weeks (96%) with this regimen.[41],[42] However, a single randomized control trial failed to demonstrate any appreciable improvement compared to placebo, the caveat being the absence of metronidazole in the said regimen. Although results seem to be in favor of this regimen as an adjunct treatment, further large scale studies are necessary to validate its role.
Octreotide
Level of Evidence 3, Grade of Recommendation D
A single anectodal observation has reported clearance of rosacea lesions in three of four treated patients with octreotide, a somatostatin analog.[43]
Zinc sulfate
Level of Evidence 1−, Grade of Recommendation B
Two randomized control trials have been found dealing with zinc sulfate; in one randomized control trial significant improvement was seen in 19 patients with zinc sulfate (100 mg thrice daily) compared to placebo,[44] while the other study did not show any significant advantage over placebo (220 mg twice daily for 3 months; 44 patients).[45] Further studies are needed to corroborate this finding.
Physical and light-based therapies
Despite the availability of several medical therapies, physical and light-based modalities are being employed lately to target some clinical manifestations like telangiectasias, persistent facial erythema and phymas (especially rhinophymas), which respond poorly to the former [Table - 6].[46]

Rationale of light-based therapies/lasers
Both telangiectasias and persistent erythema occur due to dilated blood vessels in the superficial skin. The lasers and light (intense pulsed light) rays are absorbed by certain components of blood called chromophores like oxyhemoglobin, deoxyhemoglobin and methemoglobin to generate heat energy resulting in vascular destruction.[47] However, energy needs to be delivered quicker than a vessel releases its heat to surrounding tissues (i.e., more rapidly than thermal relaxation time). Thus, smaller vessels causing redness are more difficult to treat as higher energy needs to be delivered in a shorter period of time to destroy them. The different types of lasers reported to be useful in rosacea have been tabulated below [Table - 6].
Limitations
Some important adverse effects of these light-based therapies are tabulated below: [Table - 7].[46]

Emerging therapies
Apart from the current therapeutic modalities, few emerging therapies need special mention:
Serine protease inhibitors
Patients suffering from rosacea possess higher levels of the serum protease kallikrein 5 compared to the general population, which cleaves the cathelicidin precursor into LL-37, a key molecule involved in the pathogenesis of rosacea. Thus, serum protease inhibitors are being evaluated for the treatment of rosacea. Topical epsilon-aminocaproic acid, a serine protease inhibitor has been found to improve the erythema significantly in patients suffering from papulopustular rosacea within 6 weeks of starting therapy.[48] However, further studies are needed to validate its usage.
Mast cell stabilizers
Mast cells have been suggested to play a key role in the pathogenesis of rosacea by releasing cathelicidins, LL-37, matrix metalloproteinases and other inflammatory cytokines. Thus, agents blocking mast cell degranulation may have a beneficial role, at least theoretically. With this idea, a topical mast cell stabilizer cromolyn sodium (4%) has been compared with a placebo to obtain modest symptomatic improvement.[49] However, large scale studies are needed to establish these findings.
Thus, both these agents may be considered prospective therapeutic options for treating rosacea in the coming days.
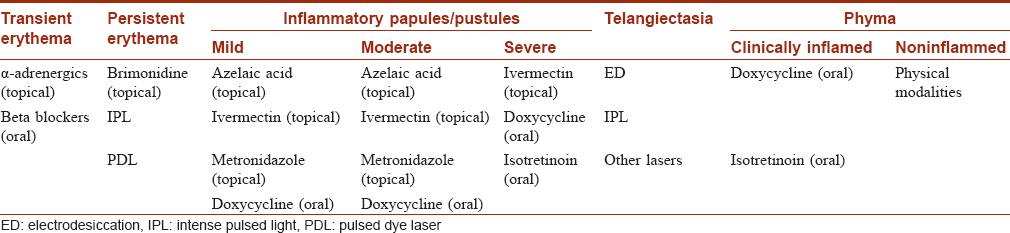
Selection of the appropriate treatment modality-consensus guidelines:
In 2014, the American Acne and Rosacea Society classified rosacea into two major types to simplify the treatment: (1) centrofacial erythema with papulopustular lesions/papulopustular rosacea and (2) centrofacial erythema without papulopustular lesions/erythematotelangiectatic variety.[40] Papulopustular rosacea may be further graded into 3 types according to severity- mild (<10 papules/pustules, mild erythema, with or without symptoms), moderate (10–19 papules/pustules, moderate erythema, with or without symptoms) and severe (>20 papules/pustules, severe erythema, with or without symptoms). Recently, a global panel of experts (ROSacea Consensus panel) has recommended a phenotype-based treatment algorithm for this condition [Table - 8].[50]
Conclusion
Rosacea remains an underreported condition in the skin of color, although its prevalence is more than what was thought previously. Epidemiological studies, especially from India are scarce and there is a need to identify clinical markers with better sensitivity to improve the diagnosis of rosacea in patients with skin of color. Like the etiopathogenic factors involved in rosacea, the treatment modalities are also multifarious and complex. The level of evidence and grade of recommendation for each treatment option is presented to aid the treating physician to select the most appropriate therapy according to the presentation.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patients have given their consent for their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. |
Berth-Jones J. Rosacea, perioral dermatitis and similar dermatoses, flushing and flushing syndromes. In: Burns T, Breathnach S, Cox N, Griffiths C, editors. Rook's Textbook of Dermatology. 7th ed.. UK: Wiley-Blackwell Publishing Ltd.; 2004. p. 2197-216.
[Google Scholar]
|
| 2. |
Al-Dabagh A, Davis SA, McMichael AJ, Feldman SR. Rosacea in skin of color: Not a rare diagnosis. Dermatol Online J 2014;20:13030.
[Google Scholar]
|
| 3. |
Bhat YJ, Manzoor S, Qayoom S. Steroid-induced rosacea: A clinical study of 200 patients. Indian J Dermatol 2011;56:30-2.
[Google Scholar]
|
| 4. |
Wollina U, Verma SB. Rosacea and rhinophyma: Not curse of the Celts but Indo Eurasians. J Cosmet Dermatol 2009;8:234-5.
[Google Scholar]
|
| 5. |
Al Balbeesi AO, Halawani MR. Unusual features of rosacea in Saudi females with dark skin. Ochsner J 2014;14:321-7.
[Google Scholar]
|
| 6. |
Chang AL, Raber I, Xu J, Li R, Spitale R, Chen J, et al. Assessment of the genetic basis of rosacea by genome-wide association study. J Invest Dermatol 2015;135:1548-55.
[Google Scholar]
|
| 7. |
Khaled A, Hammami H, Zeglaoui F, Tounsi J, Zermani R, Kamoun MR, et al. Rosacea: 244 Tunisian cases. Tunis Med 2010;88:597-601.
[Google Scholar]
|
| 8. |
Doe PT, Asiedu A, Acheampong JW, Rowland Payne CM. Skin diseases in Ghana and the UK. Int J Dermatol 2001;40:323-6.
[Google Scholar]
|
| 9. |
Aldrich N, Gerstenblith M, Fu P, Tuttle MS, Varma P, Gotow E, et al. Genetic vs. environmental factors that correlate with rosacea: A cohort-based survey of twins. JAMA Dermatol 2015;151:1213-9.
[Google Scholar]
|
| 10. |
Steinhoff M, Schauber J, Leyden JJ. New insights into rosacea pathophysiology: A review of recent findings. J Am Acad Dermatol 2013;69:S15-26.
[Google Scholar]
|
| 11. |
Gomaa AH, Yaar M, Eyada MM, Bhawan J. Lymphangiogenesis and angiogenesis in non-phymatous rosacea. J Cutan Pathol 2007;34:748-53.
[Google Scholar]
|
| 12. |
Yamasaki K, Gallo RL. The molecular pathology of rosacea. J Dermatol Sci 2009;55:77-81.
[Google Scholar]
|
| 13. |
Chen W, Plewig G. Are Demodex mites principal, conspirator, accomplice, witness or bystander in the cause of rosacea? Am J Clin Dermatol 2015;16:67-72.
[Google Scholar]
|
| 14. |
Lazaridou E, Korfitis C, Kemanetzi C, Sotiriou E, Apalla Z, Vakirlis E, et al. Rosacea and & Helicobacter pylori: Links and risks. Clin Cosmet Investig Dermatol 2017;10:305-10.
[Google Scholar]
|
| 15. |
Yamasaki K, Di Nardo A, Bardan A, Murakami M, Ohtake T, Coda A, et al. Increased serine protease activity and cathelicidin promotes skin inflammation in rosacea. Nat Med 2007;13:975-80.
[Google Scholar]
|
| 16. |
Sulk M, Seeliger S, Aubert J, Schwab VD, Cevikbas F, Rivier M, et al. Distribution and expression of non-neuronal transient receptor potential (TRPV) ion channels in rosacea. J Invest Dermatol 2012;132:1253-62.
[Google Scholar]
|
| 17. |
Wilkin J, Dahl M, Detmar M, Drake L, Liang MH, Odom R, et al. Standard grading system for rosacea: Report of the National Rosacea Society Expert Committee on the classification and staging of rosacea. J Am Acad Dermatol 2004;50:907-12.
[Google Scholar]
|
| 18. |
Bae YI, Yun SJ, Lee JB, Kim SJ, Won YH, Lee SC. Clinical evaluation of 168 Korean patients with rosacea: The sun exposure correlates with the erythematotelangiectatic subtype. Ann Dermatol 2009;21:243-9.
[Google Scholar]
|
| 19. |
Xie HF, Huang YX, He L, Yang S, Deng YX, Jian D, et al. An observational descriptive survey of rosacea in the Chinese population: Clinical features based on the affected locations. PeerJ 2017;5:e3527.
[Google Scholar]
|
| 20. |
Furukawa M, Kanetou K, Hamada T. Rhinophyma in Japan. Int J Dermatol 1994;33:35-7.
[Google Scholar]
|
| 21. |
Egeberg A, Weinstock LB, Thyssen EP, Gislason GH, Thyssen JP. Rosacea and gastrointestinal disorders: A population-based cohort study. Br J Dermatol 2017;176:100-6.
[Google Scholar]
|
| 22. |
Wu CY, Chang YT, Juan CK, Shieh JJ, Lin YP, Liu HN, et al. Risk of inflammatory bowel disease in patients with rosacea: Results from a nationwide cohort study in Taiwan. J Am Acad Dermatol 2017;76:911-7.
[Google Scholar]
|
| 23. |
Spoendlin J, Voegel JJ, Jick SS, Meier CR. A study on the epidemiology of rosacea in the UK. Br J Dermatol 2012;167:598-605.
[Google Scholar]
|
| 24. |
Hua TC, Chung PI, Chen YJ, Wu LC, Chen YD, Hwang CY, et al. Cardiovascular comorbidities in patients with rosacea: A nationwide case-control study from Taiwan. J Am Acad Dermatol 2015;73:249-54.
[Google Scholar]
|
| 25. |
Egeberg A, Hansen PR, Gislason GH, Thyssen JP. Patients with rosacea have increased risk of dementia. Ann Neurol 2016;79:921-8.
[Google Scholar]
|
| 26. |
Two AM, Wu W, Gallo RL, Hata TR. Rosacea: Part II. Topical and systemic therapies in the treatment of rosacea. J Am Acad Dermatol 2015;72:761-70.
[Google Scholar]
|
| 27. |
Draelos ZD. Cosmeceuticals for rosacea. Clin Dermatol 2017;35:213-7.
[Google Scholar]
|
| 28. |
Coda AB, Hata T, Miller J, Audish D, Kotol P, Two A, et al. Cathelicidin, kallikrein 5, and serine protease activity is inhibited during treatment of rosacea with azelaic acid 15% gel. J Am Acad Dermatol 2013;69:570-7.
[Google Scholar]
|
| 29. |
van Zuuren EJ, Kramer SF, Carter BR, Graber MA, Fedorowicz Z. Effective and evidence-based management strategies for rosacea: Summary of a Cochrane systematic review. Br J Dermatol 2011;165:760-81.
[Google Scholar]
|
| 30. |
Narayanan S, Hünerbein A, Getie M, Jäckel A, Neubert RH. Scavenging properties of metronidazole on free oxygen radicals in a skin lipid model system. J Pharm Pharmacol 2007;59:1125-30.
[Google Scholar]
|
| 31. |
Dahl MV, Katz HI, Krueger GG, Millikan LE, Odom RB, Parker F, et al. Topical metronidazole maintains remissions of rosacea. Arch Dermatol 1998;134:679-83.
[Google Scholar]
|
| 32. |
Fowler J, Jarratt M, Moore A, Meadows K, Pollack A, Steinhoff M, et al. Once-daily topical brimonidine tartrate gel 0·5% is a novel treatment for moderate to severe facial erythema of rosacea: Results of two multicentre, randomized and vehicle-controlled studies. Br J Dermatol 2012;166:633-41.
[Google Scholar]
|
| 33. |
Fowler J Jr, Jackson M, Moore A, Jarratt M, Jones T, Meadows K, et al. Efficacy and safety of once-daily topical brimonidine tartrate gel 0.5% for the treatment of moderate to severe facial erythema of rosacea: Results of two randomized, double-blind, and vehicle-controlled pivotal studies. J Drugs Dermatol 2013;12:650-6.
[Google Scholar]
|
| 34. |
Cardwell LA, Alinia H, Moradi Tuchayi S, Feldman SR. New developments in the treatment of rosacea-role of once-daily ivermectin cream. Clin Cosmet Investig Dermatol 2016;9:71-7.
[Google Scholar]
|
| 35. |
Taieb A, Ortonne JP, Ruzicka T, Roszkiewicz J, Berth-Jones J, Peirone MH, et al. Superiority of ivermectin 1% cream over metronidazole 0·75% cream in treating inflammatory lesions of rosacea: A randomized, investigator-blinded trial. Br J Dermatol 2015;172:1103-10.
[Google Scholar]
|
| 36. |
Layton AM. Pharmacologic treatments for rosacea. Clin Dermatol 2017;35:207-12.
[Google Scholar]
|
| 37. |
Rebora A, Drago F. Papulopustular rosacea. In: Williams CH, editor. Evidence-Based Dermatology. 3rd ed.. UK: Wiley Blackwell; 2017. p. 106-11.
[Google Scholar]
|
| 38. |
Del Rosso JQ, Preston NJ, Caveney SW, Gottschalk RW. Effectiveness and safety of modified-release doxycycline capsules once daily for papulopustular rosacea monotherapy results from a large community-based trial in subgroups based on gender. J Drugs Dermatol 2012;11:703-7.
[Google Scholar]
|
| 39. |
Craige H, Cohen JB. Symptomatic treatment of idiopathic and rosacea-associated cutaneous flushing with propranolol. J Am Acad Dermatol 2005;53:881-4.
[Google Scholar]
|
| 40. |
Gollnick H, Blume-Peytavi U, Szabó EL, Meyer KG, Hauptmann P, Popp G, et al. Systemic isotretinoin in the treatment of rosacea-doxycycline- and placebo-controlled, randomized clinical study. J Dtsch Dermatol Ges 2010;8:505-15.
[Google Scholar]
|
| 41. |
Yang X. Relationship between Helicobacter pylori and Rosacea: review and discussion. BMC Infect Dis 2018;18:318.
[Google Scholar]
|
| 42. |
Szlachcic A, Sliwowski Z, Karczewska E, Bielański W, Pytko-Polonczyk J, Konturek SJ. Helicobacter pylori and its eradication in rosacea. J Physiol Pharmacol 1999;50:777-86.
[Google Scholar]
|
| 43. |
Piérard-Franchimont C, Quatresooz P, Piérard GE. Incidental control of rosacea by somatostatin. Dermatology 2003;206:249-51.
[Google Scholar]
|
| 44. |
Sharique KE, Najim RA, Al-Salman HN. Oral zinc sulphate in the treatment of rosacea: A double blind placebo controlled study. Int J Dermatol 2006;45:857-61.
[Google Scholar]
|
| 45. |
Bamford JT, Gessert CE, Haller IV, Kruger K, Johnson BP. Randomized, double-blind trial of 220 mg zinc sulfate twice daily in the treatment of rosacea. Int J Dermatol 2012;51:459-62.
[Google Scholar]
|
| 46. |
Del Rosso JQ, Thiboutot D, Gallo R, Webster G, Tanghetti E, Eichenfield LF, et al. American Acne & Rosacea Society. Consensus recommendations from the American Acne & Rosacea Society on the management of rosacea, part 5: a guide on the management of rosacea. Cutis 2014;93:134-8.
[Google Scholar]
|
| 47. |
Tanghetti EA, Mirkov M, Sierra RA. Common vascular lesions. In: Raulin C, Karsai S, editors. Laser and IPL Technology in Dermatology and Aesthetic Medicine. Berlin, Germany: Springer-Verlag Berlin Heidelberg; 2011. p. 227-40.
[Google Scholar]
|
| 48. |
Two AM, Hata TR, Nakatsuji T, Coda AB, Kotol PF, Wu W, et al. Reduction in serine protease activity correlates with improved rosacea severity in a small, randomized pilot study of a topical serine protease inhibitor. J Invest Dermatol 2014;134:1143-5.
[Google Scholar]
|
| 49. |
Muto Y, Wang Z, Vanderberghe M, Two A, Gallo RL, Di Nardo A. Mast cells are key mediators of cathelicidin-initiated skin inflammation in rosacea. J Invest Dermatol 2014;134:2728-36.
[Google Scholar]
|
| 50. |
Tan J, Almeida LM, Bewley A, Cribier B, Dlova NC, Gallo R, et al. Updating the diagnosis, classification and assessment of rosacea: Recommendations from the global ROSacea COnsensus (ROSCO) panel. Br J Dermatol 2017;176:431-8.
[Google Scholar]
|
Fulltext Views
19,513
PDF downloads
3,681





