Translate this page into:
Seborrheic melanosis and dermoscopy: Lumping better than splitting
2 Skinnocence: The Skin Clinic and Research Centre; Kalyani-Escorts Hospital, Gurugram, Haryana, India
3 Department of Dermatology, College of Medicine, King Faisal University, Riyadh, Saudi Arabia
4 Institute of Dermatology, University Hospital Santa Maria Della Misericordia, Udine, Italy
5 Department of Skin and VD, Patna Medical College and Hospital, Patna, Bihar, India
6 First Department of Dermatology, Aristotle University, Thessaloniki, Greece
Correspondence Address:
Sidharth Sonthalia
Skinnocence: The Skin Clinic and Research Centre, C.2246, Sushant Lok.1, Block.C, Gurugram - 122 009, Haryana
India
| How to cite this article: Arshdeep, Sonthalia S, Kaliyadan F, Errichetti E, Jha AK, Lallas A. Seborrheic melanosis and dermoscopy: Lumping better than splitting. Indian J Dermatol Venereol Leprol 2018;84:585-587 |
Dear Sir,
We read with interest the deliberation by Verma et al., on “Seborrheic melanosis: An entity worthy of mention in dermatology literature” published in your esteemed journal.[1] Before we share our concerns, the authors deserve acknowledgement for their sincere attempt to formally describe a relatively common condition that has for long been loosely and anecdotally referred to as seborrheic melanosis.
In the absence of any accepted precedence, the definition of this condition should encompass the clinical presentation, possible etiology and the semantics of the term. To the best of our understanding, the name “seborrheic melanosis” should represent localized pockets of “melanosis,” most likely postinflammatory hyperpigmentation resulting from a “seborrhea”-related process, i.e., facial seborrheic dermatitis. Logically, these pockets may be seen in a long-standing untreated facial seborrheic dermatitis. Indeed, facial seborrheic dermatitis is a well-recognized cause for hyperpigmentation in skin of color.[2]
Postinflammatory hyperpigmentation, especially in skin of color, may result from various inflammatory and papulosquamous skin conditions, including facial seborrheic dermatitis, acne, atopic dermatitis, perioral dermatitis, psoriasis, contact allergic dermatitis, and angular cheilitis amongst others.[2] These conditions may also manifest concomitantly at overlapping facial sites. Hence, postinflammatory hyperpigmentation occurring at sebum-rich areas can be multifactorial in origin and should not be attributed majorly to facial seborrheic dermatitis. Further, we believe that the following issues regarding the study methodology merit attention:
- Although the small cohort size of 12 patients is adequate for a pilot study, the lack of clear inclusion/exclusion criteria and a sketchy methodology seem inappropriate for designating a new term worthy of publication in dermatologic literature. Moreover, deriving conclusions about the condition such as the predilection in women and in darker skin types, rarity in postmenopausal women, etc., from mere 12 patients, is at most conjectural.
- Given the explicit denotation accorded by the authors to this condition, an objectivized and better-defined systematic methodology was warranted. Categorizing 12 patients into three Fitzpatrick skin types should have mentioned the methodology (subjective analysis vs. employing Fitzpatrick skin-type scale).[3]
- Authors state that “early lesions showed localized scaly erythema of alar groove and nasolabial dyssebacia, which later progressed to develop frank pigmentation,”[1] although no time-frame of follow-up of individual patients was mentioned. In the absence of mention of any follow-up details, the authors' stated “progression of the condition” sounds implausible.
- Our most important concern is with the dermoscopic evaluation. Dermoscopic evaluation of non-nevoid conditions is still in an infantile stage, especially in skin of color. As per the adage of medical research, we must know what's normal before commenting on a condition perceived abnormal. In the absence of published literature on the normal dermoscopic findings of facial folds such as the alar groove and submental crease and lack of case–control approach in the study, stated findings of dermoscopically virginal areas in an ambiguous condition raise serious doubts. In our limited experience, even in healthy young adults not suffering from facial seborrheic dermatitis, we have clinically observed light-brown pigmentation and mild scaling in the alar grooves [Figure - 1]a and [Figure - 1]b. Dermoscopy from the alar grooves of healthy adults frequently displays mild to moderate erythema, brown globules and clods, out-of-focus linear and arborizing vessels and telangiectasias, minimal white scaling and visible as well as plugged skin pores [Figure - 1]c and [Figure - 1]d. The alar groove displays variable pigmentation and vascularity irrespective of a seborrheic dermatitic affliction.
- The labiomental crease and angles of the mouth are not typical sites of involvement in facial seborrheic dermatitis,[4] rather these areas become involved in perioral dermatitis, angular cheilitis and pigmentary demarcation line type H, respectively.[5] For example, in [Figure 4] of the original study, an overlap with angular cheilitis and perioral dermatitis is appreciable. [Figure - 2]a,[Figure - 2]b,[Figure - 2]c,[Figure - 2]d,[Figure - 2]e,[Figure - 2]f demonstrates the presence of minimal-to-moderate brown pigmentation of the labiomental folds in healthy young males without any past/current evidence of facial seborrheic dermatitis. Lack of mention of clinical/dermoscopic involvement of other typical facial seborrheic dermatitis-defining sites, especially the scalp and retroauricular region, is another pitfall.
- The “shadow” effect contributing majorly to the pigmentation of the labiomental crease (with near disappearance of the pigmentation on pursing the lips) mentioned by the authors themselves sounds contradictory. The etiology of periocular hyperpigmentation, a comprehensively studied condition with no scholastic comparison with seborrheic melanosis also involves the “tear trough” associated shadow effect.[6] Despite that, the clinico-dermoscopic evaluation of periocular hyperpigmentation has been reported only recently in an Egyptian study of 35 patients.[7] A simple dermoscopic comparison of the labiomental crease with and without stretching the skin would have been instrumental in solving this paradox
- With a presumptive background of seborrheic melanosis/facial seborrheic dermatitis relationship, the dermoscopic features were a little surprising, e.g., absence of dotted vessels in a patchy distribution typical of facial seborrheic dermatitis, and presence of linear and arborizing vessels that are typical of seborrheic dermatitis of the scalp (which was not examined).[8],[9] Moreover, linear branching vessels are seen in many facial inflammatory conditions and are not pathognomonic of facial seborrheic dermatitis.[8],[9]
- The subcategorization of the dermoscopic patterns observed in only 12 patients into three distinctive classes - vascular, pigmentation and mixed, that too for a condition being defined nebulously for the first time seems perfunctory. While pigmentation was common in all the patients, thereby qualifying for “melanosis,” the significance of vascular pattern (which may be seen normally in the alar grooves) in dermoscopic characterization and subcategorization of the condition is questionable, especially in the absence of histopathological confirmation.
 |
| Figure 1: (a) Clinical photograph of a healthy young male showing mild reddish-brown pigmentation along the right alar groove without facial seborrheic dermatitis (b) Clinical photograph of a young male displaying velvety brown-colored pigmentation along the left alar groove along with presence of multiple plugged facial pores and open comedones (c) Dermoscopic image corresponding to Figure 1a revealsdiffuse faint erythema, out-of-focus telangiectasias, linear and arborizing vessels (black arrows), plugged pores (white arrows), scattered brown globules and minimal white scaling (Escope, videodermoscope, ×20, semi-polarized mode) (d) Dermoscopic image corresponding to Figure 1b reveals diffuse faint erythema, out Figure 1: (a) Clinical photograph of a healthy young male showing mild reddish-brown pigmentation along the right alar groove without facial seborrheic dermatitis (b) Clinical photograph of a young male displaying velvety brown-colored pigmentation along the left alar groove along with presence of multiple plugged facial pores and open comedones (c) Dermoscopic image corresponding to Figure 1a revealsdiffuse faint erythema, out-of-focus telangiectasias, linear and arborizing vessels (black arrows), plugged pores (white arrows), scattered brown globules and minimal white scaling (Escope, videodermoscope, ×20, semi-polarized mode) (d) Dermoscopic image corresponding to Figure 1b reveals diffuse faint erythema, out-of-focus telangiectasias, linear and arborizing vessels (black arrows), plugged pores (white arrows), scattered brown globules and minimal white scaling (Escope, videodermoscope, ×20, semi-polarized mode)of-focus telangiectasias, linear and arborizing vessels (black arrows), plugged pores (white arrows), scattered brown globules and minimal white scaling (Escope, videodermoscope, ×20, semi-polarized mode) |
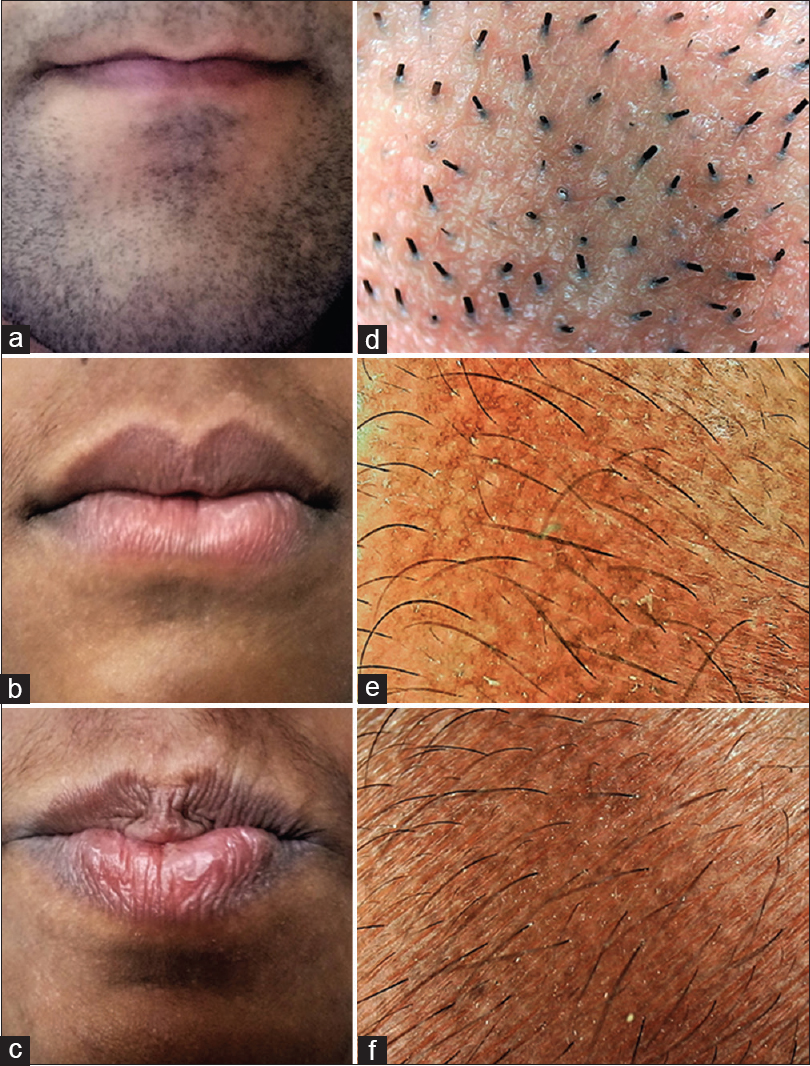 |
| Figure 2: (a) Labiomental crease and angle of the lips in a young healthy adult male without any current or recent past history of facial seborrheic dermatitis clinically showing minimal brown pigmentation (b) Clinical photograph of a young healthy adult male showing mild brown pigmentation of the labiomental crease and angle of mouth, secondary to angular cheilitis and perioral dermatitis (c) Clinical photograph of a young male showing moderate pigmentation of the labiomental crease and angle of mouth, secondary to perioral dermatitis (d) Dermoscopic image corresponding to Figure 2a reveals slightly accentuated pigment network (Escope, videodermoscope, ×20, polarized mode) (e) Dermoscopic image corresponding to Figure 2b reveals medium-brown pigment network (Escope, videodermoscope, ×20, polarized mode) (f) Dermoscopic image corresponding to Figure 2c reveals dark-brown pigment network (Escope, videodermoscope, ×20, polarized mode) |
In conclusion, although the authors must be congratulated for their maiden attempt to define this ill-understood condition and bringing forth this hitherto under-explored subject to the table, a clinicopathological correlation would have done true justice to the paper. Notwithstanding the merit in dermoscopic evaluation of any cutaneous disorder, we firmly believe that interpretation and subset categorization of dermoscopic images warrant caution and avoidance of the other highlighted pitfalls.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. |
Verma SB, Vasani RJ, Chandrashekar L, Thomas M. Seborrheic melanosis: An entity worthy of mention in dermatological literature. Indian J Dermatol Venereol Leprol 2017;83:285-9.
[Google Scholar]
|
| 2. |
Davis EC, Callender VD. Postinflammatory hyperpigmentation: A review of the epidemiology, clinical features, and treatment options in skin of color. J Clin Aesthet Dermatol 2010;3:20-31.
[Google Scholar]
|
| 3. |
Fitzpatrick TB. The validity and practicality of sun-reactive skin types I through VI. Arch Dermatol 1988;124:869-71.
[Google Scholar]
|
| 4. |
Clark GW, Pope SM, Jaboori KA. Diagnosis and treatment of seborrheic dermatitis. Am Fam Physician 2015;91:185-90.
[Google Scholar]
|
| 5. |
Tempark T, Shwayder TA. Perioral dermatitis: A review of the condition with special attention to treatment options. Am J Clin Dermatol 2014;15:101-13.
[Google Scholar]
|
| 6. |
Sarkar R, Ranjan R, Garg S, Garg VK, Sonthalia S, Bansal S, et al. Periorbital hyperpigmentation: A comprehensive review. J Clin Aesthet Dermatol 2016;9:49-55.
[Google Scholar]
|
| 7. |
Mostafa WZ, Kadry DM. Clinical and dermoscopic evaluation of patients with periorbital darkening. J Egypt Womens Dermatol Soc 2014;11:191-6.
[Google Scholar]
|
| 8. |
Errichetti E, Stinco G. Dermoscopy in general dermatology: A practical overview. Dermatol Ther (Heidelb) 2016;6:471-507.
[Google Scholar]
|
| 9. |
Lallas A, Argenziano G, Apalla Z, Gourhant JY, Zaballos P, Di Lernia V, et al. Dermoscopic patterns of common facial inflammatory skin diseases. J Eur Acad Dermatol Venereol 2014;28:609-14.
[Google Scholar]
|
Fulltext Views
5,545
PDF downloads
873





