Translate this page into:
The efficacy of 1% metronidazole gel in facial seborrheic dermatitis: A double blind study
2 Department of Dermatology, Isfahan Medical School, Isfahan University of Medical Sciences and Health services, Isfahan, Iran
3 Department of Pharmaceutics, Isfahan Pharmacy School, Isfahan University of Medical Sciences and Health services, Isfahan, Iran
4 Isfahan University of Medical Sciences, Isfahan, Iran
Correspondence Address:
Amir H Siadat
Skin Diseases and Leishmaniasis Research Center, Sedigehe Tahereh Recearch Center, Isfahan University of Medical Sciences, Isfahan
Iran
| How to cite this article: Siadat AH, Iraji F, Shahmoradi Z, Enshaieh S, Taheri A. The efficacy of 1% metronidazole gel in facial seborrheic dermatitis: A double blind study. Indian J Dermatol Venereol Leprol 2006;72:266-269 |
Abstract
Background: Seborrheic dermatitis is a common, chronic inflammatory disease of the skin, characterized by erythematous plaques that are covered with yellow, greasy, scales and may or may not be associated with itching. Although a few studies have suggested use of topical metronidazole for the treatment of seborrheic dermatitis, there is no general consensus about it. Aims: To evaluate the efficacy of 1% metronidazole gel and its vehicle alone, in the treatment of seborrheic dermatitis. Methods: This was a double-blind, prospective, clinical trial. Fifty six patients with seborrheic dermatitis were randomly allocated to either apply 1% metronidazole gel or placebo to their facial lesions, twice daily for 8 weeks. All the patients were evaluated and scored every 2 weeks for 8 weeks. The results were analyzed statistically. Results: A total of 56 patients entered this study and 53 patients completed the treatment course. There was no significant difference in terms of demographic data and lesions severity score. There were statistically significant differences in the reduction of mean severity scores between the 2 groups at the second, fourth, sixth and eighth weeks of treatment ( p < 0.05). Metronidazole gel significantly decreased mean of seborrheic dermatitis severity score from the 2nd visit ( p <0.001). Conclusion: Metronidazole gel is an effective treatment for facial seborrheic dermatitis.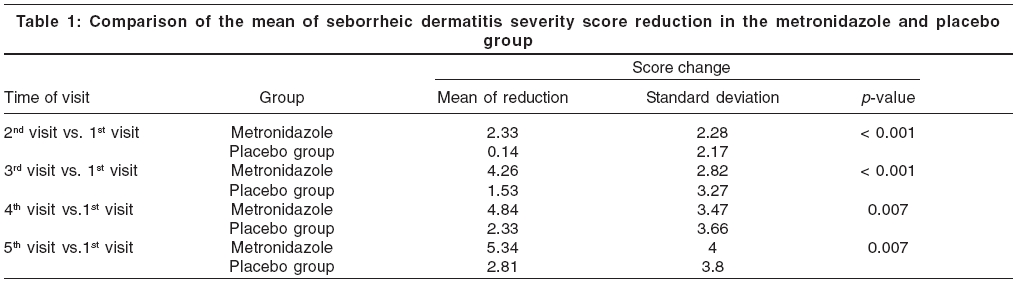

 |
 |
 |
 |
Introduction
Seborrheic dermatitis is a common, chronic inflammatory disease of the skin.[1] This disease is characterized is by erythematous plaques that are covered with yellow, greasy, scales and may or may not be associated with itching.[2] It is more common than psoriasis and involves 1-3% of the whole population and 3-5% of young adults in the USA. This disease is one of the most common dermatologic presentations in AIDS patients and 85/5% of these patients are suffering from it.[2] It has two peak ages of onset, one during first 3 months of life and the second during the 4th to 7th decades of life. It is more common in males than in females.[3] The exact cause of this disease is unknown, although many factors such as seborrhea, microbial factors (especially Malassezia furfur ), neurological disturbances, emotional stresses, physical factors, nutritional disturbances and genetic predisposition and immune deficiency,[4],[5],[6],[7] have been suggested.
Many treatments such as topical corticosteroids, 3-5% salicylic acid, topical or oral imidazoles, calcipotriol cream, ciclopirox olamine, pimecrolimus, tacrolimus and propylene glycol, have been used for treatment of this dermatosis with variable successes.
Although a few studies have suggested use of topical metronidazole for treatment of seborrheic dermatitis, there is no consensus about it.[8],[9],[10] Our objective in this study was to compare the efficacy of 1% metronidazole gel versus its vehicle alone, in the treatment of seborrheic dermatitis.
Methods
This study was a double-blind, randomized vehicle-controlled clinical trial. Our objective population was patients with seborrheic dermatitis who were older than 18 years, had not used systemic antibiotics or topical zinc, selenium or salicylic acid in the recent 2 weeks or retinoic acid in the recent 30 days or systemic antifungal or immunosuppressive drug in the recent 3 months. The patients with systemic illness and pregnant and lactating women, were also excluded. Seborrheic dermatitis of the patients had been confirmed by the dermatologist in the first visit and the severity of the lesions was determined by a similar dermatologist in the first and follow up visits. Fifty six patients with seborrheic dermatitis admitted to Skin Diseases and Leishmaniasis Research Center, were randomly allocated by random allocation software into 2 groups of cases and controls. The case group was treated with 1% metronidazole gel and the control group was treated with vehicle gel alone. Vehicle gel was composed of hydroxypropyl cellulose.
Skin Disease and Leishmaniasis Research Center ethics committee clearance, was obtained for performing this study. Written informed consent was obtained from the patients. The patients applied drugs to involved areas of the face (including nasolabial folds, dorsal site of the nose, retroauricular areas and eye brows). All of the patients were treated for 8 weeks and were followed at weeks 2, 4, 6 and 8 after starting the treatment.
For determining the severity of seborrheic dermatitis, we used the scoring system suggested by Koca et al .[8] In this scoring system, 4 different parts of the face including eye brows, retro auricular areas, dorsal side of the nose and the nasolabial folds were evaluated. Severity of erythema, scaling, pruritus and papules were evaluated and scored from 0 to 3 in each of these areas (0=absent 1 = mild 2 = moderate 3 = severe). The sum of these values is regarded as score of seborrheic dermatitis in the face. The maximum score was calculated to be 48, if all four areas were involved with the maximum severity. (4´4´3= 48).
The severity of itching was evaluated by visual analogue scale (VAS). VAS was defined as a 10 cm line, in which point 0 refers to existence of no pruritus and point 10 refers to the most severe pruritus. According to this scale, we scored the pruritus of the patients from point 1 to 3 as mild pruritus, from point 4 to 6 as moderate pruritus and from point 7 to point 10 as severe pruritus.
For comparison of seborrheic dermatitis severity between 2 groups of cases and controls, Student′s t-test and for comparison of seborrheic dermatitis severity in the same group of patients at the 2 different visits, paired t-test was used.
Results
Fifty six patients entered this study (11 males and 17 females in the metronidazole group and 7 males and 21 females in the placebo group). Fifty three patients completed the treatment course. In the metronidazole group, one male patient opted out of the study due to increased redness and pruritus and while one female case opted out due to lack of improvement in her lesions. In the placebo group, one female patient discontinued because of increase in small papules on her nose.
Age range of the participants was between 18-54 years and most of the patients in both groups were between 21-30 years old. The mean age of participants in the placebo group was 25.5 years and in the metronidazole group was 27.4 years; 53 patients completed the treatment course.
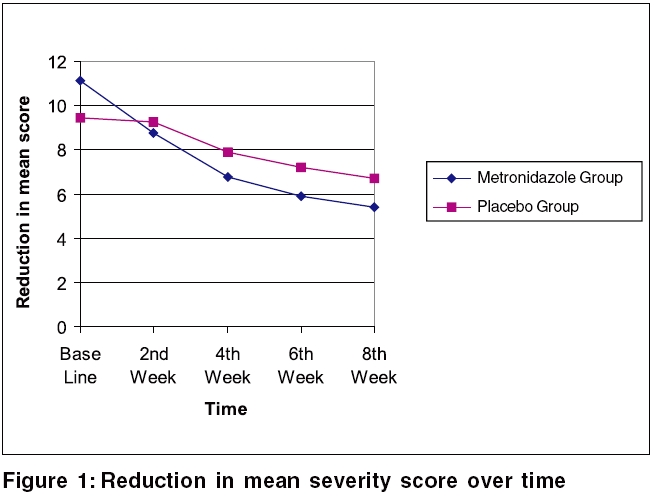
The mean severity scores at the initial visit for the placebo group and metronidazole group were 9.42 and 11.1, respectively. This difference of means at the baseline was not statistically significant ( p value > 0.05). At final visit (at 8th week) the mean severity scores for placebo and metronidazole groups were 6.74 and 5.38, respectively. There were statistically significant differences in the reduction of mean severity scores between two groups at the second, fourth, sixth and eight weeks of treatment ( p value < 0.05) [Table - 1] and [Figure - 1]. Metronidazole gel significantly decreased mean seborrheic dermatitis severity score from 2nd visit ( p value< 0.001).
Discussion
Metronidazole is a synthetic, nitro imidazole antibiotic, that acts through DNA destruction and prevention of nucleic acid synthesis, in the anaerobic organisms and cells.[11] In addition to antimicrobial effect, it has anti-inflammatory effect through inhibition of leukocyte chemotaxis. It also prevents release of inflammatory mediators from neutrophils and through that, decreases oxidative tissue damage.[8]
There are a few reports of using metronidazole gel in seborrheic dermatitis. Plewig and Jansen have suggested effectiveness of metronidazole gel in the treatment of the facial seborrheic dermatitis.[2] Parsad and his colleagues evaluated efficacy of 1% metronidazole gel in a double blind, placebo controlled study and showed that 66% of patients had marked improvement to complete clearance of their lesions in the metronidazole group versus 12% in the placebo group. They concluded that 1% metronidazole gel was effective in treatment of seborrheic dermatitis.[10]
Koca and colleges used 0.75% metronidazole gel for treatment of mild to moderate facial seborrheic dermatitis.[8] They concluded that there was no statistical significance in the mean severity score of the seborrheic dermatitis between 0.75 metronidazole gel and placebo, in the treatment of the seborrheic dermatitis.
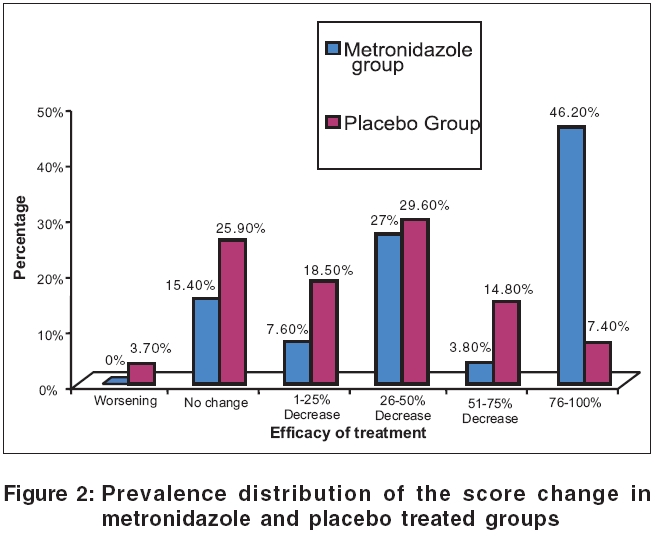
In this study, at the end of the eight weeks. As a whole, 50% of patients in the metronidazole group showed good to excellent response. In the placebo group, only 2 patients out of 27 patients showed excellent response and 4 patients showed good response and as a whole, only 22.2% of patients in the placebo showed good to excellent response.
At the end of the 8th week, 26.9% of the patients in the metronidazole group and 26.9% of the patients in the placebo group showed 26-50% improvement, that is regarded as poor response. 23% of the patients in the metronidazole group and 48.1% of the patients in the placebo group showed 0-25% response that was regarded as failure of treatment [Figure - 2].
It should be pointed out that in the placebo group, after 4 weeks of the treatment, the mean score of the disease showed statistically significant improvement that was possibly related to psychological effect of placebo or the effect of the repeated face-washing as instructed to patients or the real effects of the gel alone, itself.
According to results of this study, metronidazole gel is more effective in treatment of seborrheic dermatitis than placebo and it can be used along with other therapies as an effective, appropriate and available treatment for facial seborrheic dermatitis.
| 1. |
Gunduz K, Inanir I, Sacar H. Efficacy of terbinafine 1% cream on seborrhoeic dermatitis. J Dermatol 2005;32:22-5.
[Google Scholar]
|
| 2. |
Plewig G, Jansen T. Seborrheic Dermaitis. In : Freedberg IM, Eisen AZ, Wolff K. Fitz Patrick's Dermatology in General Medicine. 6th ed. New York: Mc-Graw Hill; 2003. p. 1199-204.
[Google Scholar]
|
| 3. |
Faergemann J. Management of seborrheic dermatitis and pityriasis versicolor. Am J Clin Dermatol 2000;1:75-80.
[Google Scholar]
|
| 4. |
Pityrosporum ovale (Malassezia furfur) as the causative agent of seborrhoeic dermatitis: New treatment options. Br J Dermatol 1996;134:12-5.
[Google Scholar]
|
| 5. |
Parry ME, Sharpe GR. Seborrhoeic dermatitis is not caused by an altered immune response to Malassezia yeast. Br J Dermatol 1998;139:254-63.
[Google Scholar]
|
| 6. |
Tollesson A, Frithz A, Stenlund K. Malassezia furfur in infantile seborrheic dermatitis. Pediatr Dermatol 1997;14:423-5.
[Google Scholar]
|
| 7. |
Hay RJ, Graham-Brown RA. Dandruff and seborrhoeic dermatitis: Causes and management. Clin Exp Dermatol 1997;22:3-6.
[Google Scholar]
|
| 8. |
Koca R, Altinyazar HC, Esturk E. Is topical metronidazole effective in seborrheic dermatitis? A double blind study. Int J Dermatol 2003;42:632-5.
[Google Scholar]
|
| 9. |
Iudica AC. Does treatment with topical metronidazole improve seborrheic dermatitis? J Fam Pract 2001;50:492.
[Google Scholar]
|
| 10. |
Parsad D, Pandhi R, Negi KS, Kumar B. Topical metronidazole in seborrheic dermatitis-a double-blind study. Dermatology 2001;202:35-7.
[Google Scholar]
|
| 11. |
Quan LT. Metronidazole. In: Wolverton SE. Comprehensive dermatologic drug therapy. Philadelphia: WB Saunders Co; 2001. p. 484-6.
[Google Scholar]
|
Fulltext Views
6,597
PDF downloads
1,013





