Translate this page into:
Treatment of spider angioma with copper vapour laser radiation (578 nm) under dermoscopy control
-
Received: ,
Accepted: ,
How to cite this article: Ponomarev IV, Topchiy SB, Andrusenko YN, Shakina LD. Treatment of spider angioma with copper vapour laser radiation (578 nm) under dermoscopy control. Indian J Dermatol Venereol Leprol 2023;89:122-5.
Sir,
Spider angioma, also known as spider naevus or spider telangiectasia, is a common benign vascular lesion resulting from dilating arterioles associated with the feeder’s vessel.1 Multiple spider angiomas may be associated with advanced liver disease, thyrotoxicosis or pronounced hormonal changes. The prevalence of spider angioma has been reported up to 15% in adolescents and adults. Patients with spider angioma are mostly looking for treatment due to cosmetic concerns. Sclerotherapy, cryotherapy, fine-needle electrocautery, pulsed dye laser, potassium titanyl phosphate or neodymium-doped yttrium aluminium garnet lasers have been used to treat spider angioma.2 Current treatment modalities of spider angioma can lead to recurrences, hyperpigmentation, hypopigmentation, pain, ulceration and scar formation.3
The pulsed dye laser treatment has shown fair results in the treatment of spider angioma, but 36% of patients have reported purpura and recurrences.3,4 According to data concerning numerical modelling of the photothermal effect of pulsed dye laser on microvessels, its radiation has failed to obturate the feeder vessel reliably in spider telangiectasia due to insufficient fluency and absorption of its irradiation by oxyhemoglobin.4 So far, in high blood velocity vessels, the blood flow does not shut off before erythrocytes expand beyond the vascular lumen via pores in the overheated vessel wall, promoting the occurrence of purpura in vessels with high-flow blood velocity.4 The copper bromide laser at 578 nm has demonstrated high efficacy for the treatment of spider angioma and other telangiectasias in four patients. Table 1 illustrates the comparison of different laser systems for the treatment of spider angioma.
| Laser mode | Number of treatments | Side effects |
|---|---|---|
| Pulsed dye laser 585 nm3 | 1–7 | Purpura |
| Copper bromide laser 578 nm5 | 3 | Mild atrophy |
| Potassium titanyl phosphate laser 532 nm8 | 1.4 ± 0.8 | 3.4% |
The copper vapour laser radiation at 578 nm has been used to treat the port-wine stain, telangiectasia and pyogenic granuloma.6,7 Copper vapour laser seems to be very promising to treat spider angioma. In this study, we report successful treatment of spider telangiectasia with dermoscopic confirmation of the complete elimination of dilated vessels using copper vapour laser.
A total of 17 patients (five males and 12 females), with Fitzpatrick skin type I–III, aged 12–30 years old, with medium-size spider angioma (up to 8 mm), were treated from January to October 2020 at the Medical Center LaNezh (Vologda, Russian Federation) and Laser department of the Health Institute (Kharkiv, Ukraine). The clinical characteristics of patients with spider angioma are indicated in Table 2.
| Number of patients | 17 |
| Number of spider angiomas | 22 |
| Age in years | 12–30 |
| Size in mm | 2–7 |
| Duration in months | 1–18 |
| Dermoscopic pattern | Star 20/22 Network 2/22 |
The lesions were located on the face and trunk [Figures 1–3]. The dermoscopic evaluation revealed irregular arborizing or branched dilated blood vessels around the core vessel confirming the diagnosis [Figures 2a and 4a].
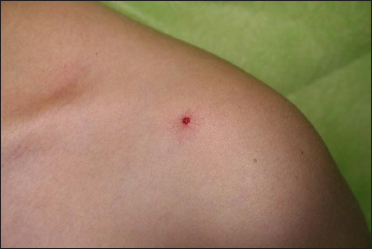
- Before treatment: Spider angioma on the left upper part of the chest in a 11 year-old-boy
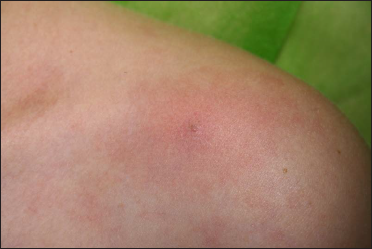
- Immediately after copper vapour laser treatment (average power - 0.7 W, exposure time - 0.2 s)
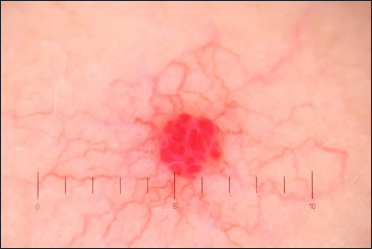
- Before treatment: Dermoscopy (×10) of spider angioma. The focus is symmetrical, boundaries are clear and the lobular structure is characteristic of eruptive angioma.
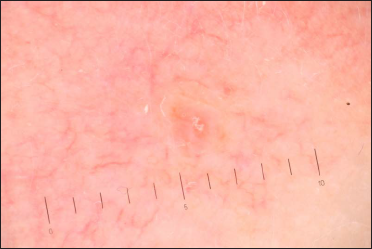
- Immediately after copper vapour laser treatment. Dermoscopy (×10) of spider angioma: the focus has changed and acquired a white-grey tint due to coagulated vessels and cavities.
Informed consent was taken after discussing the risks and benefits of laser treatment. The lesions were treated with copper vapour laser (Yakhroma–Med, Lebedev Physical Institute of Russian Academy of Sciences). The treatment was performed without anaesthesia. The laser settings were as follows: average power of 0.5–0.65 W at yellow (578 nm) wavelength, the exposure time was 0.1–0.2 s and the diameter of the light spot on the skin was 1 mm. The treatment endpoint using a multiple–stacking–pass technique was assumed when the treated area acquired a greyish tint or blanching [Figures 1b, 2b, 3b and 4b].
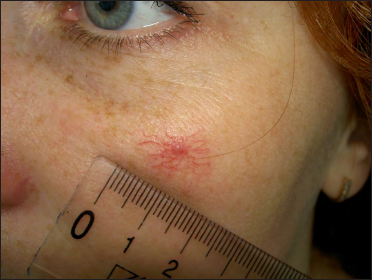
- Before treatment: Spider angioma on the right cheek in a 25-year-old woman
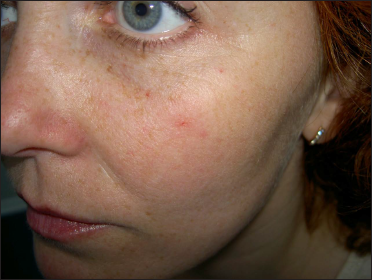
- Two weeks after copper vapour laser treatment: Spider angioma on the right cheek in a 25-year old woman
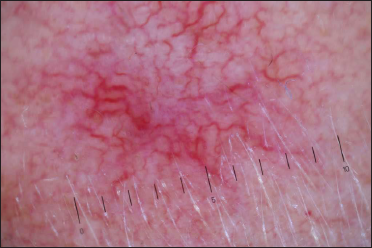
- Dermoscopy (×10) of spider angioma before treatment
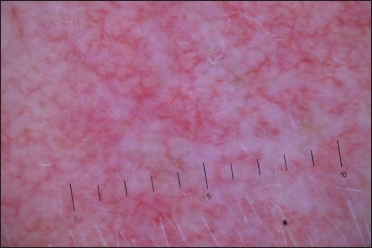
- Dermoscopy (×10) of spider angioma after tracing the “legs” surrounding the core vessel. The focus has changed dramatically and acquired a white tint due to obliterated ectatic vessels and cavities.
In Figure 5, we have demonstrated the spider angioma at various steps of laser treatment. Spider angioma is usually treated by tracing the “legs” surrounding the core vessel at an average power of 0.55 W; the exposure time being 0.12 s [Figure 5b]. Following, the power is increased up to 0.65 W, the exposure time increased up to 0.2 s, and the core vessel is sealed [Figure 5c]. The core vessel should be fully obliterated. Treating the radial dilated arterioles legs initially reduces the flow to the central core. The diminished blood flow to the core larger vessel contributes to complete photothermic obliteration of telangiectatic vessels.
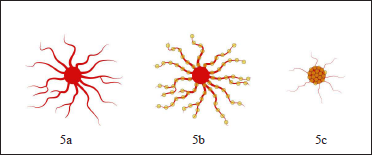
- The spider angioma treatment steps: Before treatment (5a), tracing the “legs” surrounding the core vessel (5b), power increases and the core vessel is sealed (5c). Yellow spots indicate the position of the laser pulses during the treatment.
The removal of spider angioma was carried out in a single session. No bleeding or erythema occurred post-treatment. After the procedure, the irradiated skin was treated with a 0.05% chlorhexidine gluconate solution and then epithelializing Bepanthen cream twice a day. Dermoscopy (×10) was used to check if there were some dilated vessels remaining after the laser irradiation. [Figures 2b and 4b].
The irradiated skin healed with crusting and exfoliation for two weeks [Figure 3b]. The treated spider angioma was evaluated 6–8 weeks after treatment. There were no recurrences for 24 months post-laser treatment. The treatment with copper vapour laser provided the complete removal of pyogenic angioma and clearance without adverse effect in any case. Copper vapour laser radiation provided complete removal of spider angioma by the appropriate vascular obliteration due to the high absorption of 578 nm radiation by oxyhaemoglobin in arterioles.
Our study demonstrates an excellent safety profile of copper vapour laser when treating spider angioma without purpura or pigmentary changes with good cosmetic results. Dermoscopy may detect subtle changes in the microvascular structure of the skin during the laser treatment that the naked eye cannot observe and thus improves the results of laser treatment to prevent relapses.
The limitation of the study is the lack of comparative assessment of the optimal copper vapour laser settings for patients with different skin phototypes.
Acknowledgement
The authors wish to thank Dr. Nataliya Melnikova, Medical Center LaNezh, Vologda, for supporting this study.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms.
Financial support and sponsorship
Nil.
Conflict of interest
There are no conflicts of interest.
References
- Treatment of superficial vascular lesions with the KTP 532-nm laser: experience with 647 patients. Lasers Med Sci. 2014;29:267-71.
- [CrossRef] [PubMed] [Google Scholar]
- Recurrence of spider naevi following treatment with 585 nm pulsed dye laser. J Plast Reconstr Aesthet Surg. 2007;60:668-71.
- [CrossRef] [PubMed] [Google Scholar]
- Comparative numerical analysis and optimization of blood vessels heated using various lasers. Laser Physics. 2018;28:096003.
- [Google Scholar]
- An evaluation of the copper-bromide laser for treating telangiectasia. Dermatol Surg. 1996;22:551-7.
- [CrossRef] [PubMed] [Google Scholar]
- Treatment of pyogenic granuloma in children with copper vapor laser radiation (578 nm) Indian J Dermatol Venereol Leprol. 2021;87:856-8.
- [CrossRef] [PubMed] [Google Scholar]
- Numerical modeling and clinical evaluation of pulsed dye laser and copper vapor laser in skin vascular lesions treatment. J Lasers Med Sci. 2019;10:44-9.
- [CrossRef] [PubMed] [Google Scholar]
- Single-center experience with potassium titanyl phosphate (KTP) laser for superficial cutaneous vascular lesions in face. J Cosmet Laser Ther. 2016;18:428-31.
- [CrossRef] [PubMed] [Google Scholar]





