Translate this page into:
Ultrasonography for assessing the disease activity of sclerodermoid lupus erythematosus panniculitis
-
Received: ,
Accepted: ,
How to cite this article: Cheng CY, Huang YL, Lee MC, Hu S. Ultrasonography for assessing the disease activity of sclerodermoid lupus erythematosus panniculitis. Indian J Dermatol Venereol Leprol 2021;87:146-146.
Sir,
Lupus erythematosus panniculitis is an uncommon cutaneous manifestation of lupus erythematosus. The coexistence of lupus erythematosus panniculitis and localized scleroderma is an extremely rare manifestation. Herein, we report a case of sclerodermoid lupus erythematosus panniculitis in which ultrasonography was used to monitor the therapeutic response.
A 43-year-old woman presented to our clinic with tender thickened skin lesion on the right thigh of 10 months duration. Physical examination revealed a brownish indurated plaque with peripheral erythema and epidermal atrophy on the posterolateral aspect of her right thigh [Figure 1a]. She also had malar rash [Figure 1b]. Ultrasonography (Acuson X150TM, Siemens Medical Solution USA, Mountain View, CA) demonstrated thickening of dermis and subcutaneous layer (4.4 mm and 16.3 mm, respectively). The subcutis was homogenously hyperechoic with cloudy appearance [Figure 1c]. The color Doppler also revealed increased blood flow [Figure 1d].
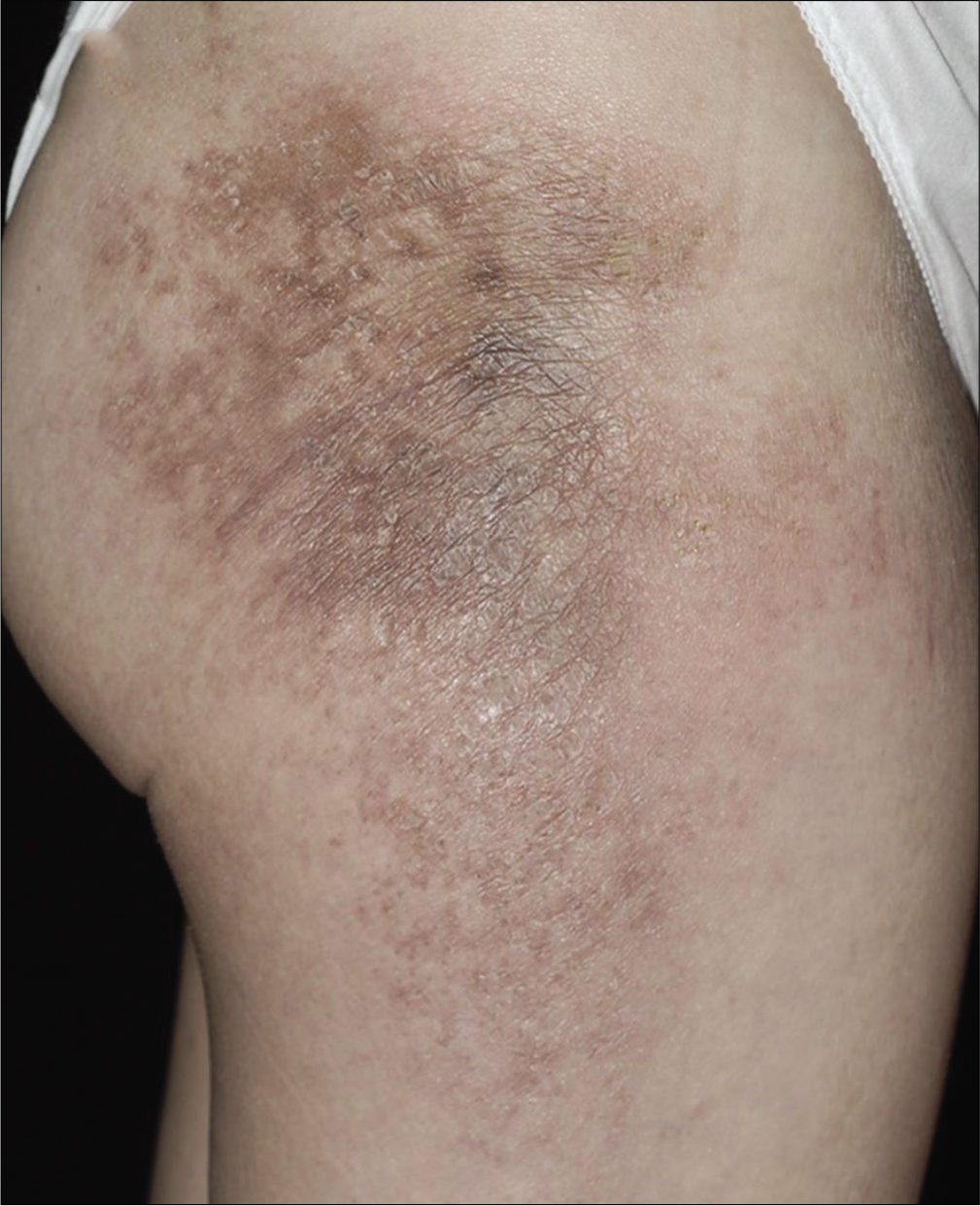
- Brownish indurated plaque with peripheral erythema on the posterolateral aspect of the right thigh
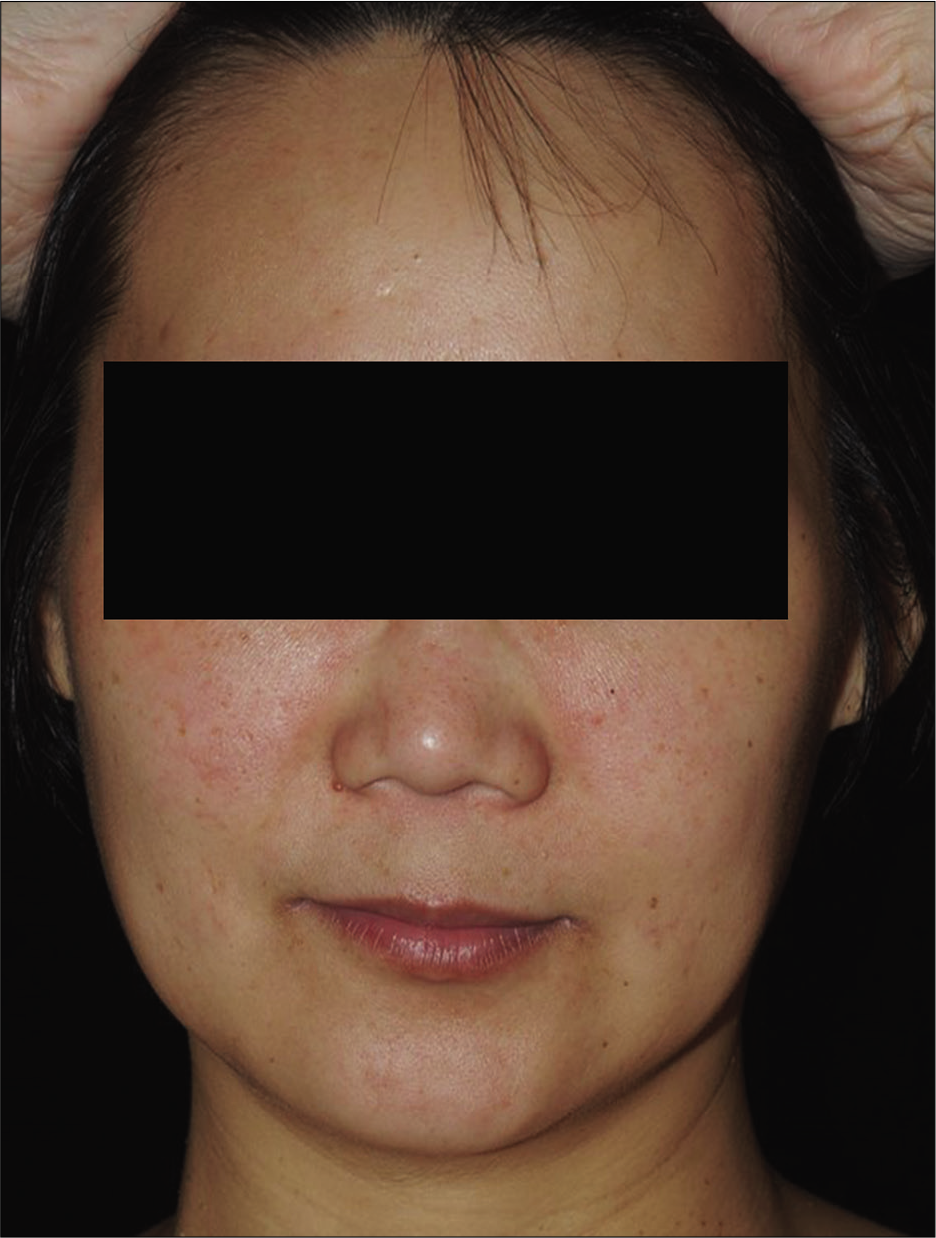
- Malar rash was noted on bilateral cheeks
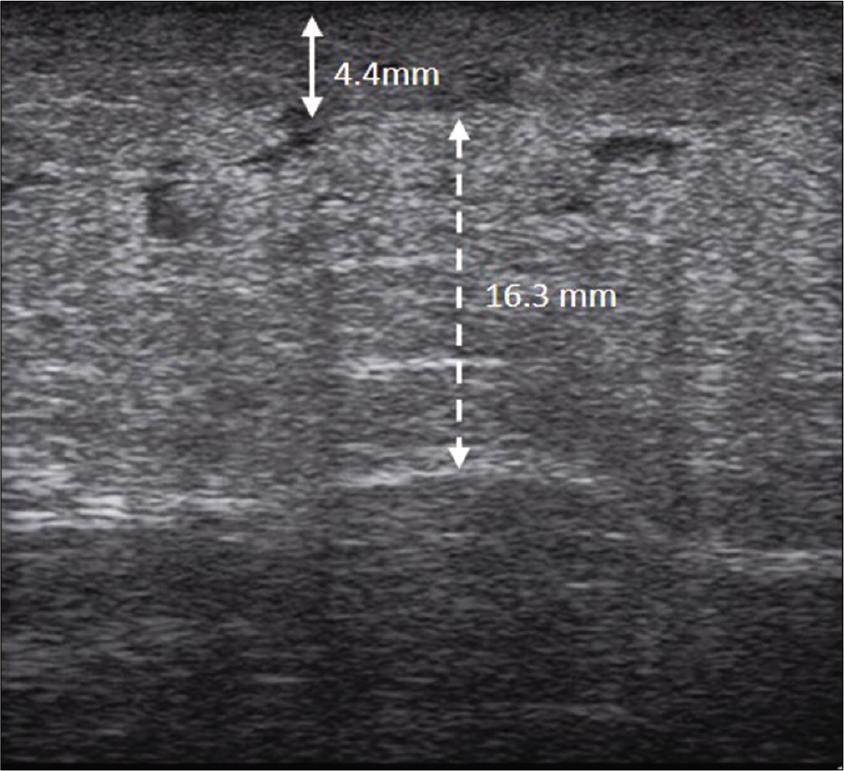
- Ultrasonographic image showing thickened dermis and subcutaneous layer, and homogenously hyperechoic subcutis with a cloudy appearance. The thickness of the dermis and the subcutaneous layer was 4.4 mm (solid double arrow line) and 16.3 mm (dotted double arrow line), respectively
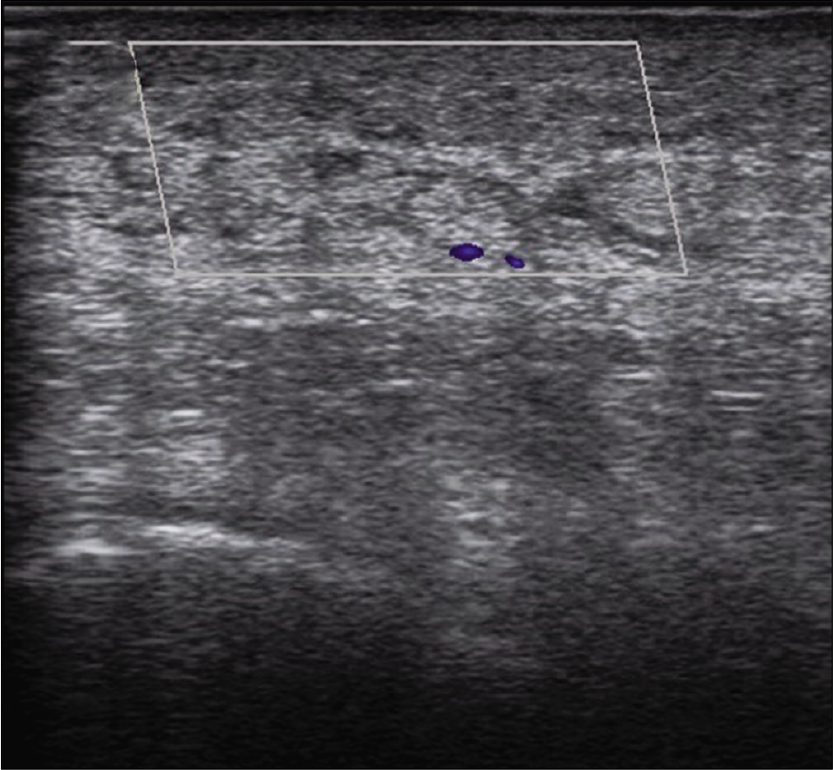
- Increased blood flow was noted in color Doppler examination
The thickness of dermis and subcutis of the contralateral nonlesional area on her left thigh was 1.0 mm and 13.0 mm, respectively [Figure 1e]. A skin biopsy taken from the plaque on her right thigh showed hyperkeratosis, mild acanthosis, melanin incontinence, thickened basement membrane and dense compact sclerosis of the dermis and subcutaneous layer with lymphocytic infiltration of vessels, sweat glands and fat lobules [Figures 2a-c]. Lupus band test was positive [Figure 2d]. The diagnosis of sclerodermoid lupus panniculitis was made based on the clinicopathological findings.
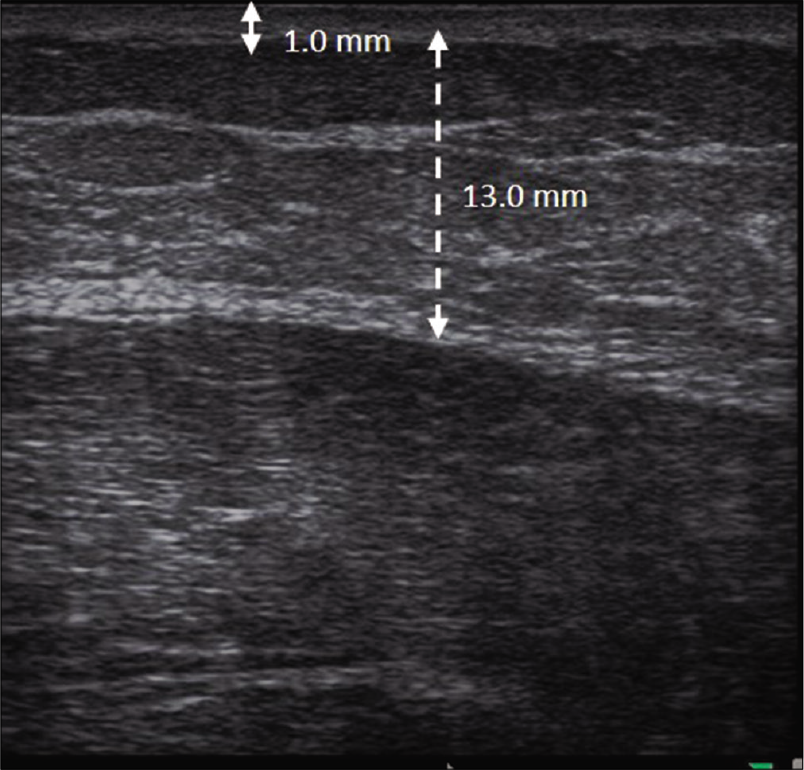
- The thickness of the dermis and subcutaneous layer of the contralateral non-lesional area was 1.0 mm (solid double arrow line) and 13.0 mm (dotted double arrow line), respectively
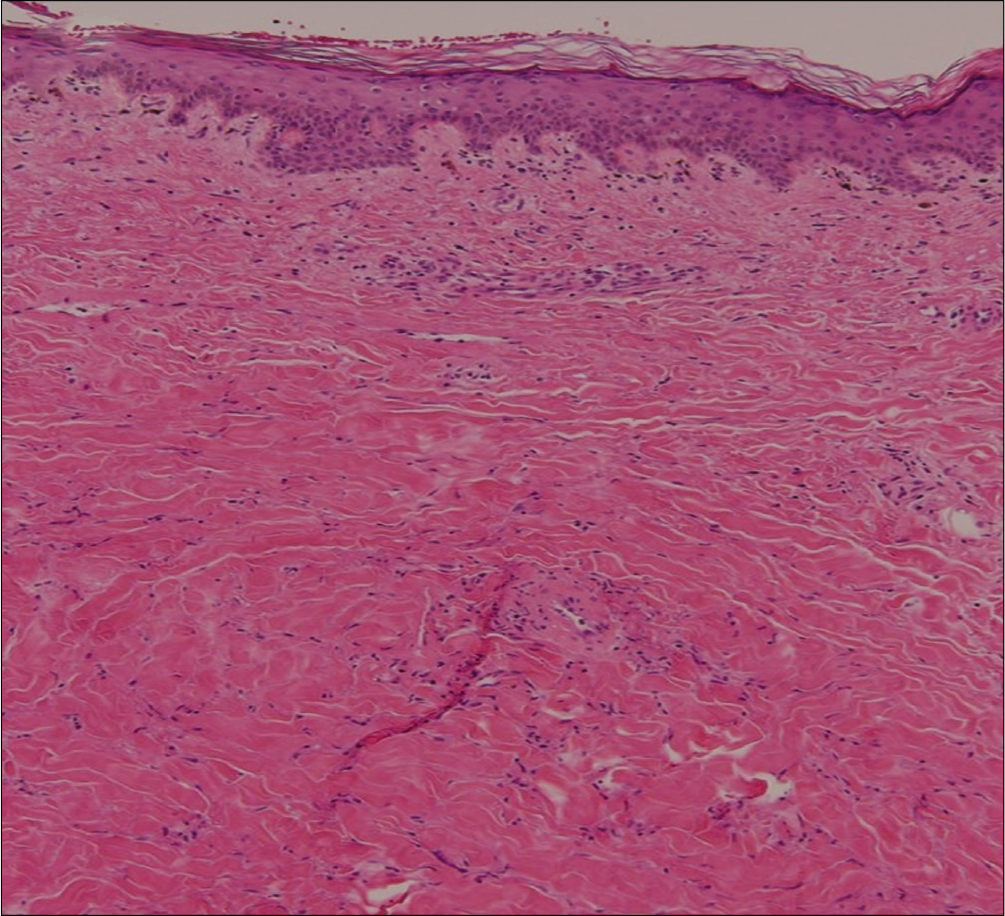
- The histopathology showing melanin incontinence, dense compact sclerosis of the dermis, periadnexal and perivascular lymphocytic infiltration (H and E, ×100)
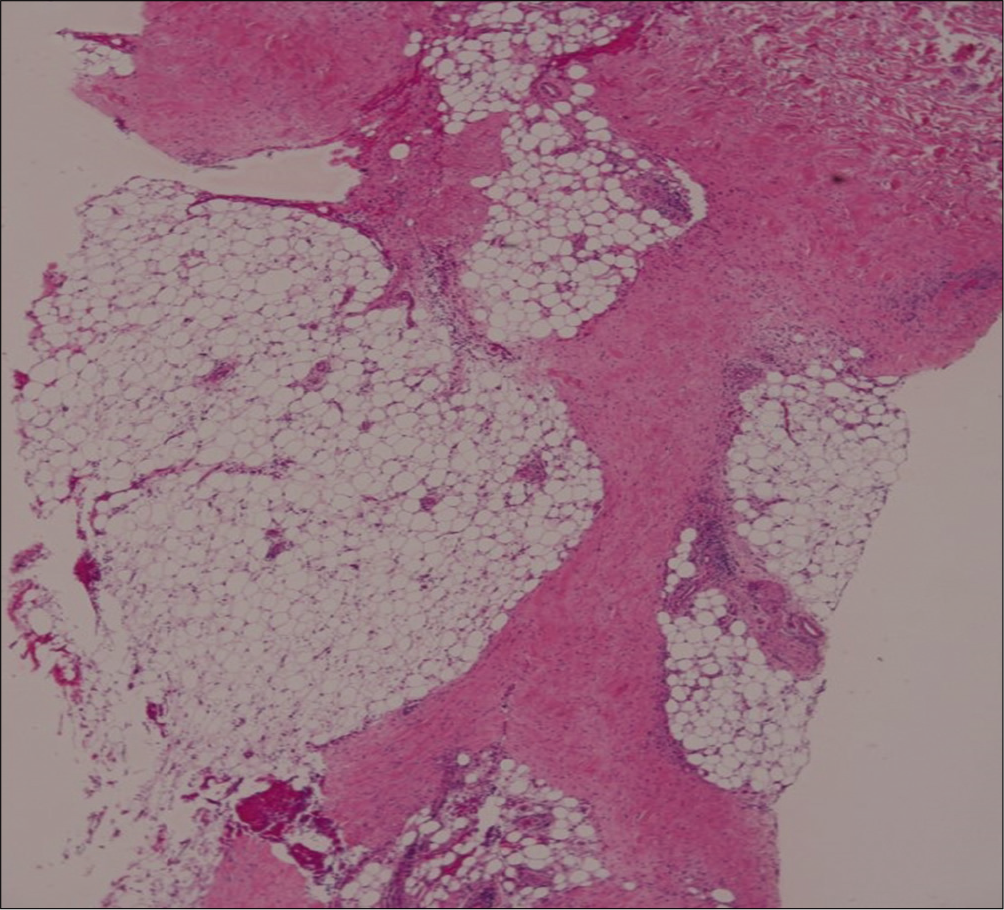
- Marked septal fibrosis with lymphocytic infiltration in subcutaneous tissue (H and E, ×100)
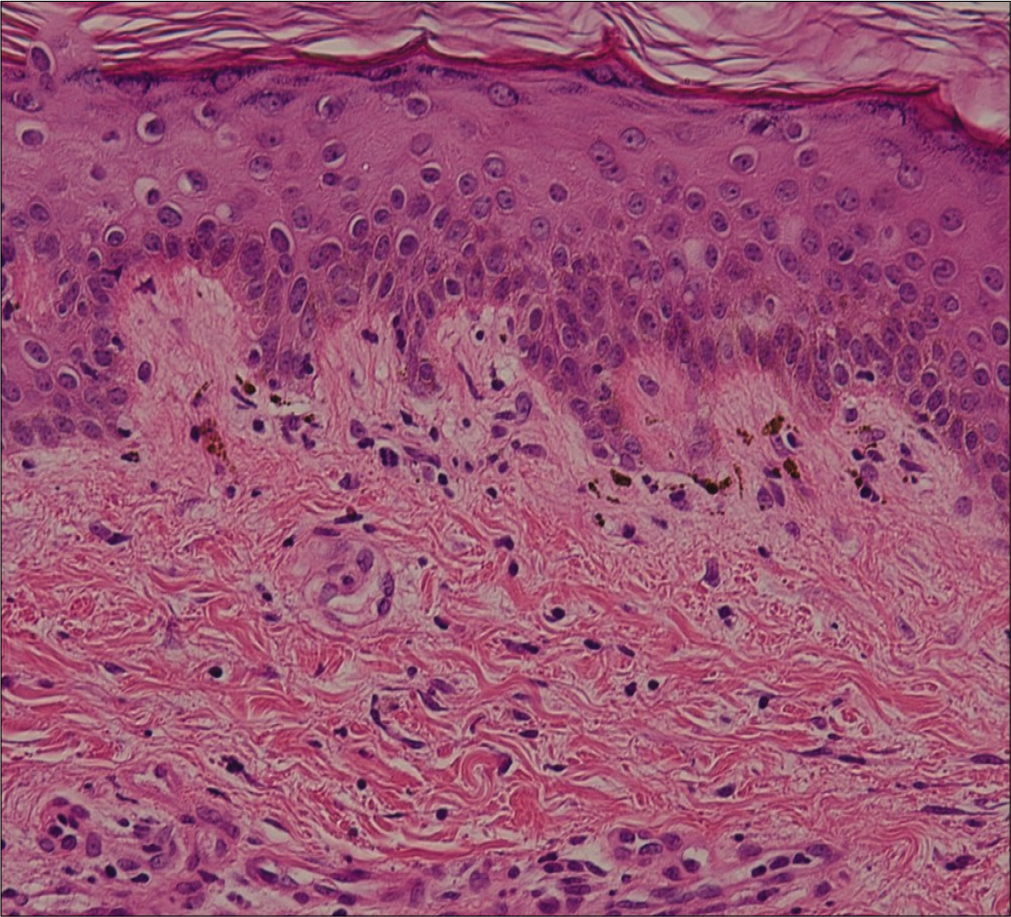
- Melanin incontinence and thickened basement membrane. (H and E, ×400)
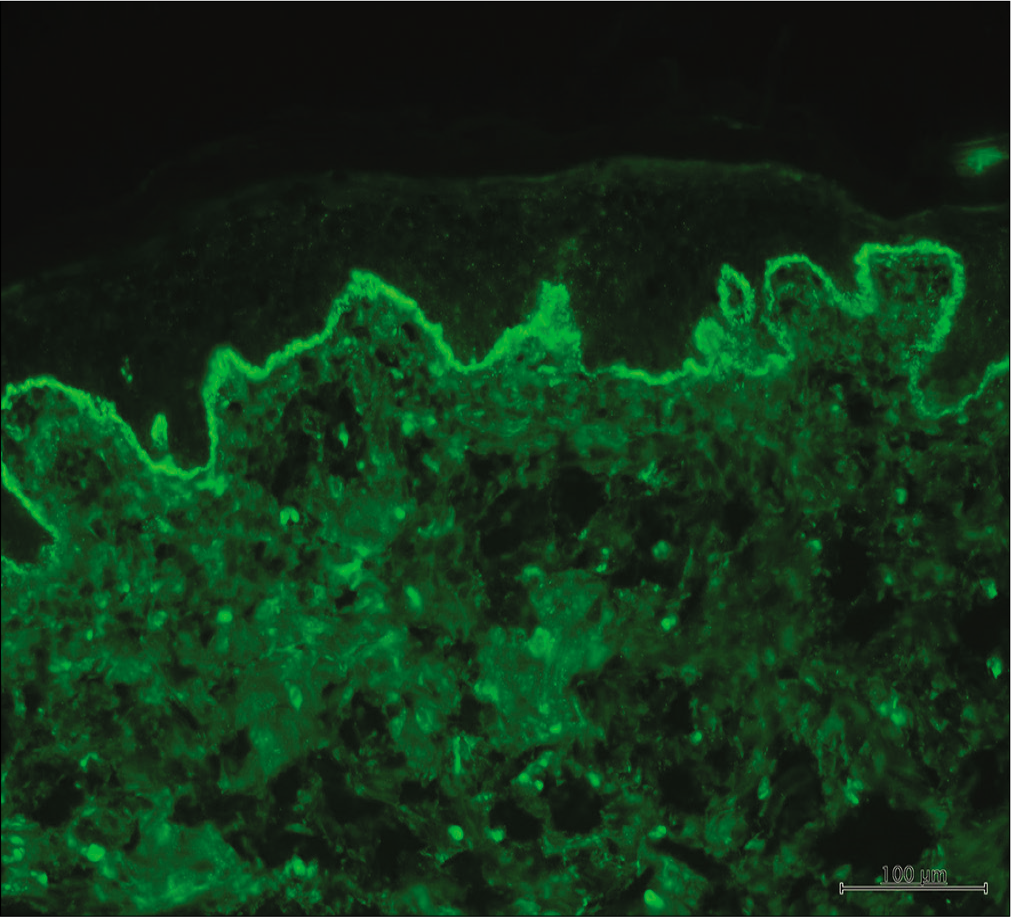
- Lupus band test showing granular deposition of IgG at the dermo-epidermal junction
Laboratory investigation showed leukocytopenia (2.7X103/uL), low complement levels (C3 and C4 25.2 and 2.62 mg/dL, respectively) as well as positive Anti Nuclear Antibody (speckled type: 1:1280), anti-sm (298AU/mL) and anti-dsDNA (203.3 WHO units). The diagnosis of systemic lupus erythematosus was made according to the 2012 systemic lupus international collaborating clinics SLE Criteria. The patient then received oral prednisolone 60 mg and hydroxychloroquine 400 mg daily along with application of clobetasol ointment twice a day.
There was slight improvement in erythema after treatment for 2 months [Figure 3a]. Ultrasonography of dermis and subcutis of the affected area showed a reduction in their thickness and the echogenicity of subcutis was also decreased [Figure 3b]. No increase in blood flow was detected [Figure 3c]. The ultrasonographic picture of the contralateral non-lesional area is demonstrated in Figure 3d. After 6 months of treatment, the lesions improved leaving behind post-inflammatory hyperpigmentation [Figure 4a]. The body weight of the patient also increased from 65 to 70 kg. Follow up imaging revealed reduced thickness of dermis and subcutis (1.0 mm and 15.0 mm, respectively) and the echogenicity of the subcutis returned to clear septate fat lobules [Figure 4b]. Color Doppler examination showed no increase in the blood flow [Figure 4c]. The thickness of dermis and subcutis in the ultrasonography of the contralateral non-lesional area was 1.1 mm and 15.3 mm, respectively [Figure 4d] Application of clobetasol ointment was discontinued but oral prednisolone and hydroxychloroquine were continued for the systemic involvement.
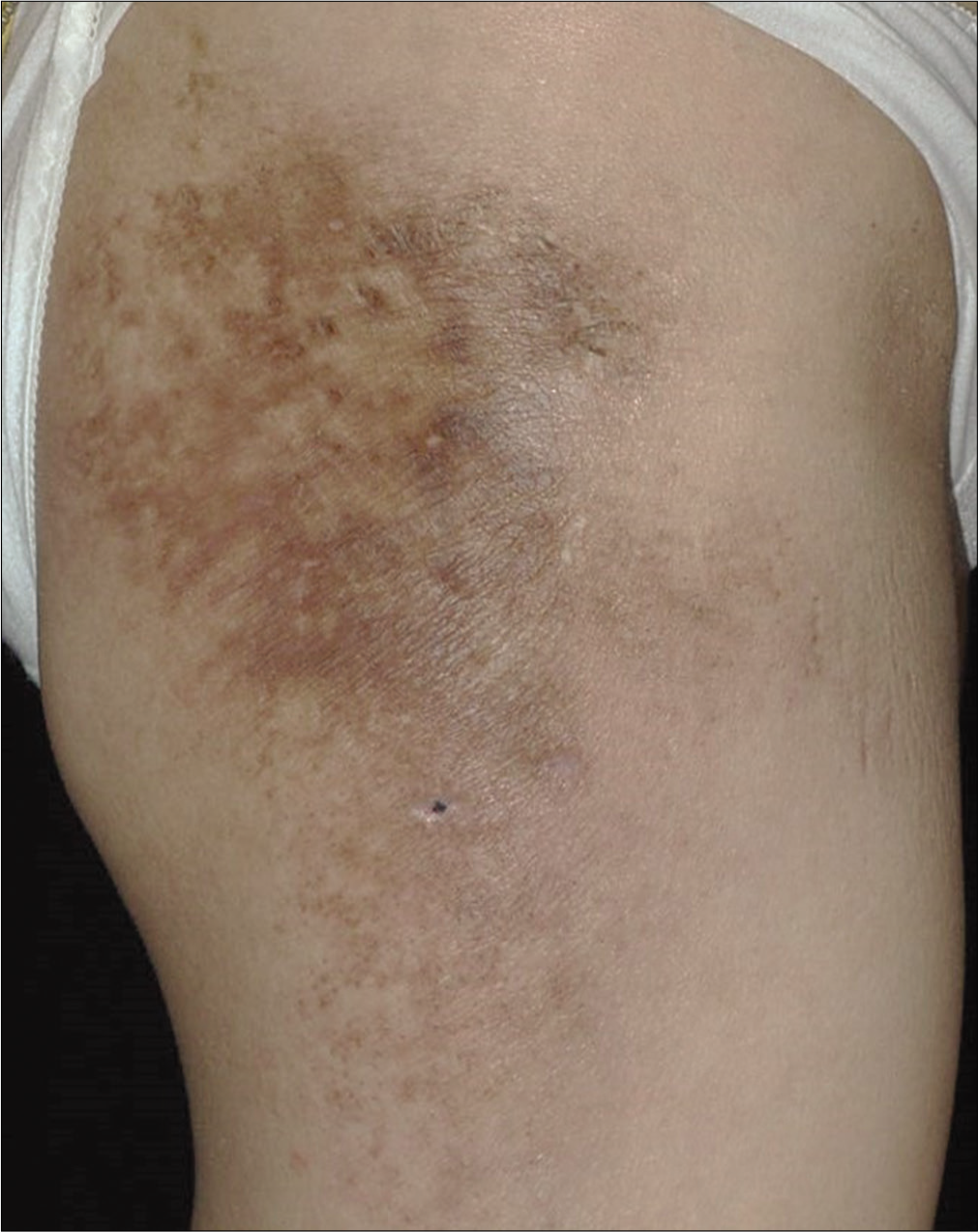
- The erythema of skin lesions improved slightly after treatment for 2 months
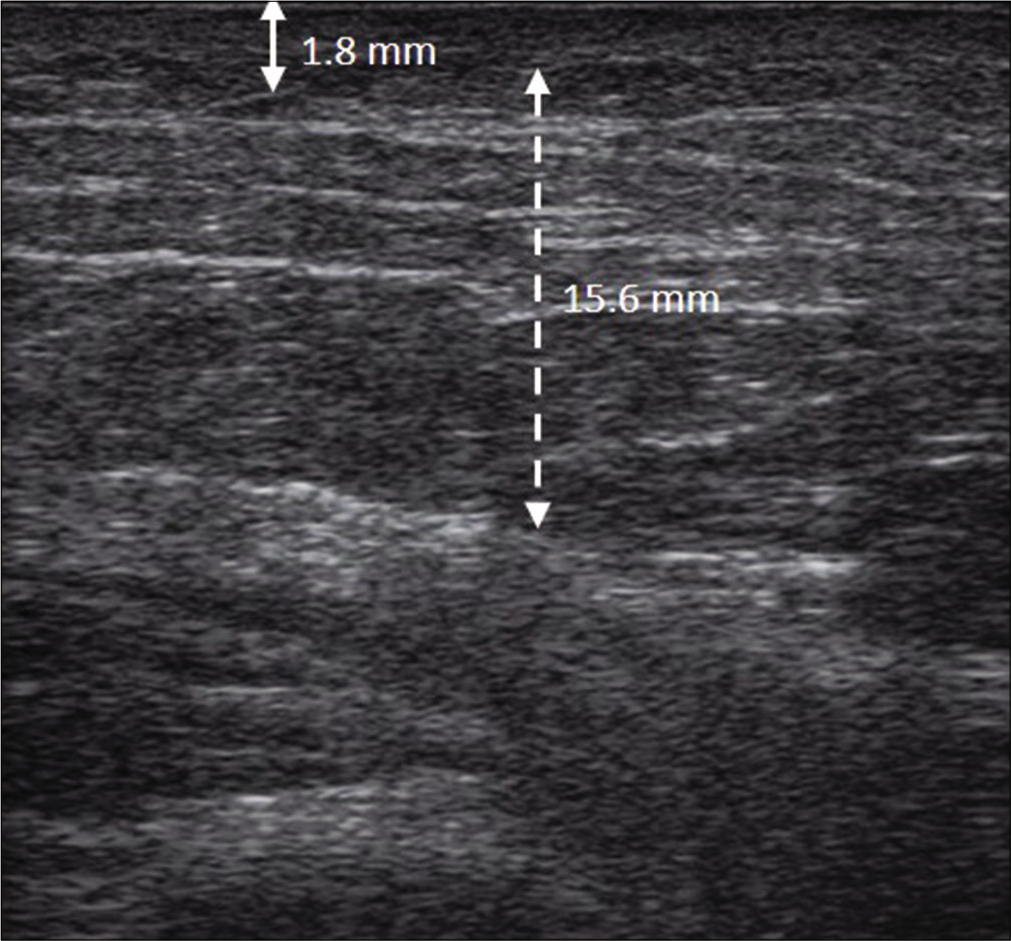
- The thickness of dermis and subcutis were reduced to 1.8 mm (solid double arrow line) and 15.6 mm (dotted double arrow line), respectively, and the echogenicity was also reduced
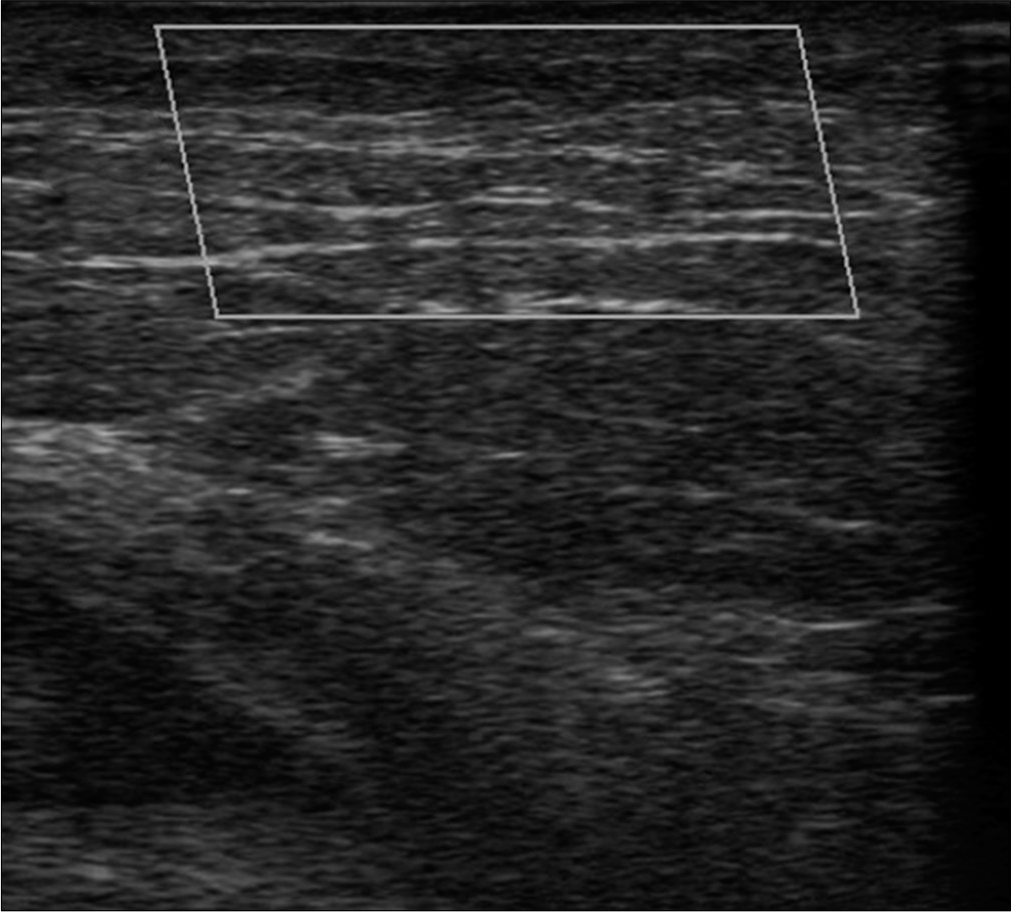
- No increased blood flow was detected in the color Doppler examination
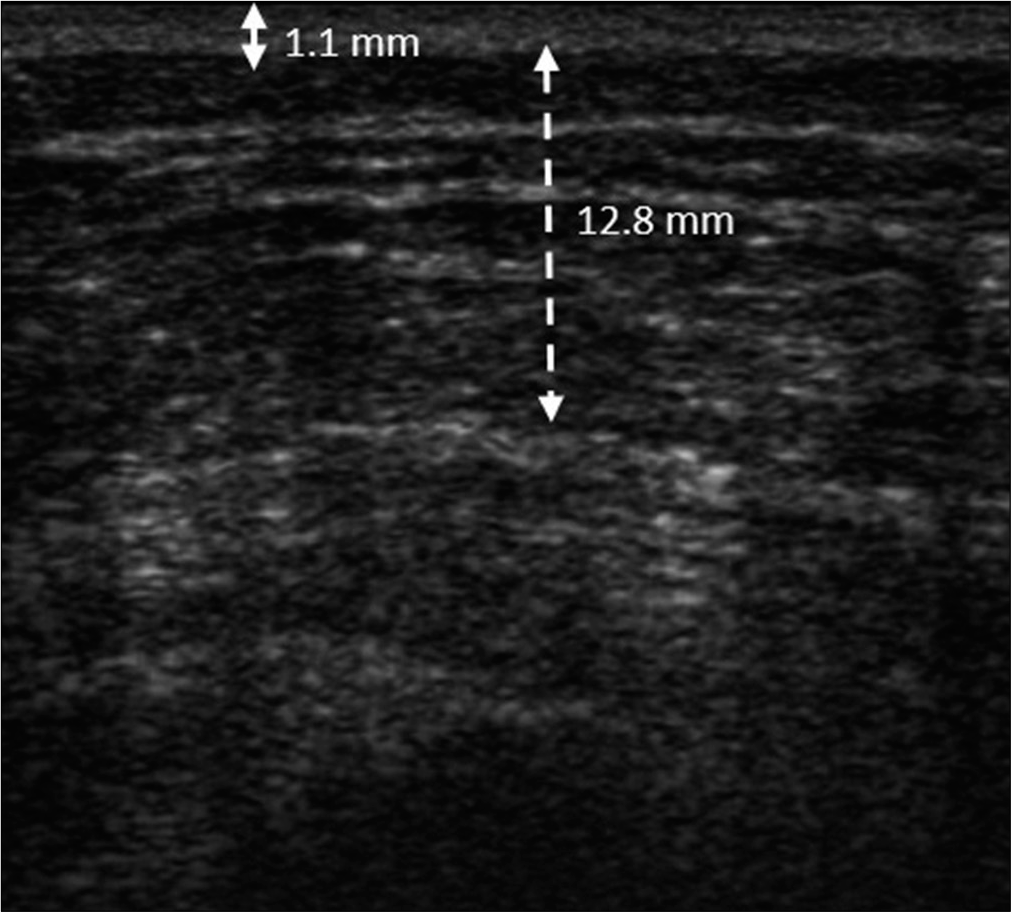
- The thickness of the dermis and subcutaneous layer of the contralateral non-lesional area was 1.1 mm (solid double arrow line) and 12.8 mm (dotted double arrow line), respectively
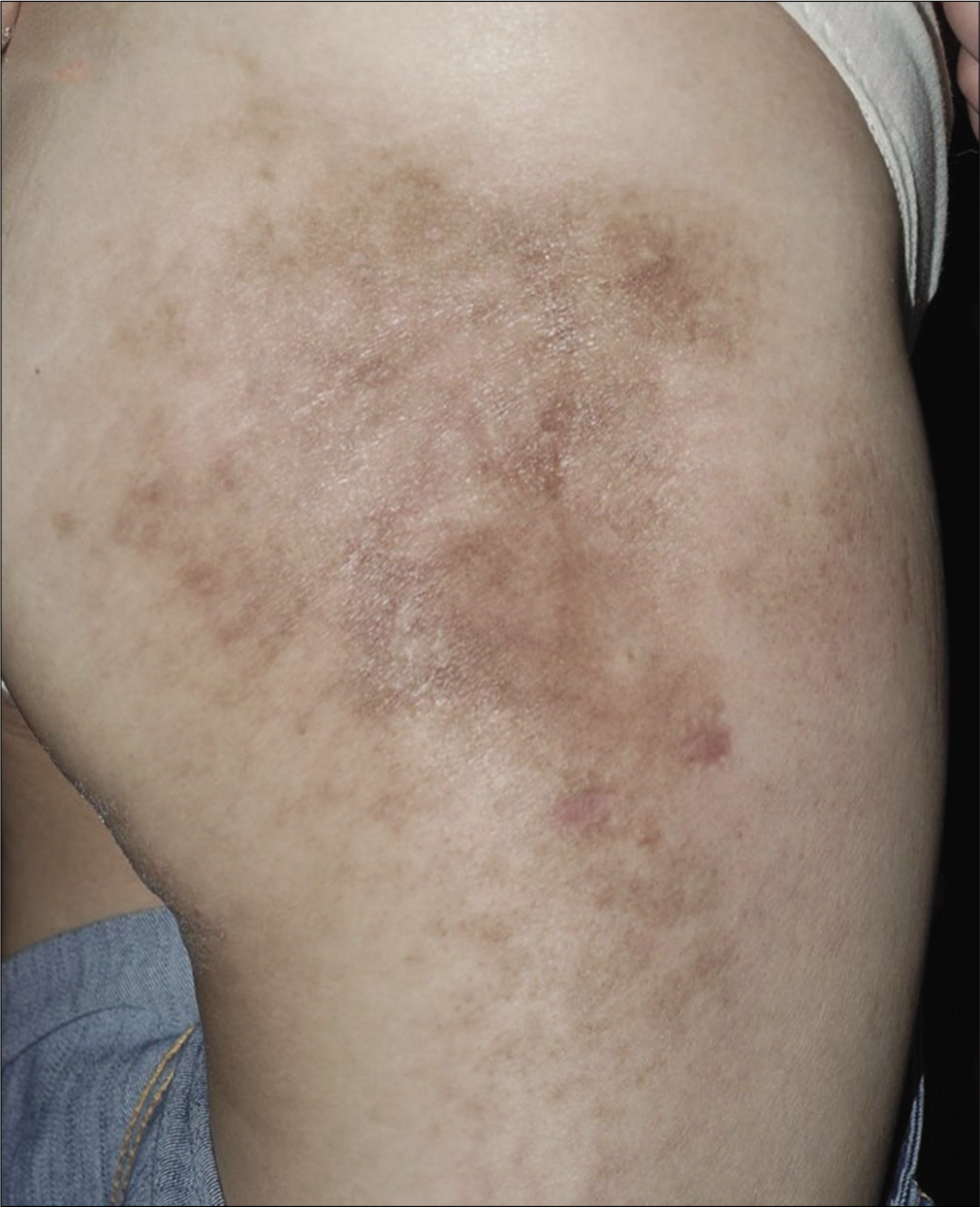
- After 6 months of treatment, the skin lesions improved with post- inflammatory hyperpigmentation name and initials will not be published and due efforts will be made to conceal the identity but anonymity cannot be guaranteed.
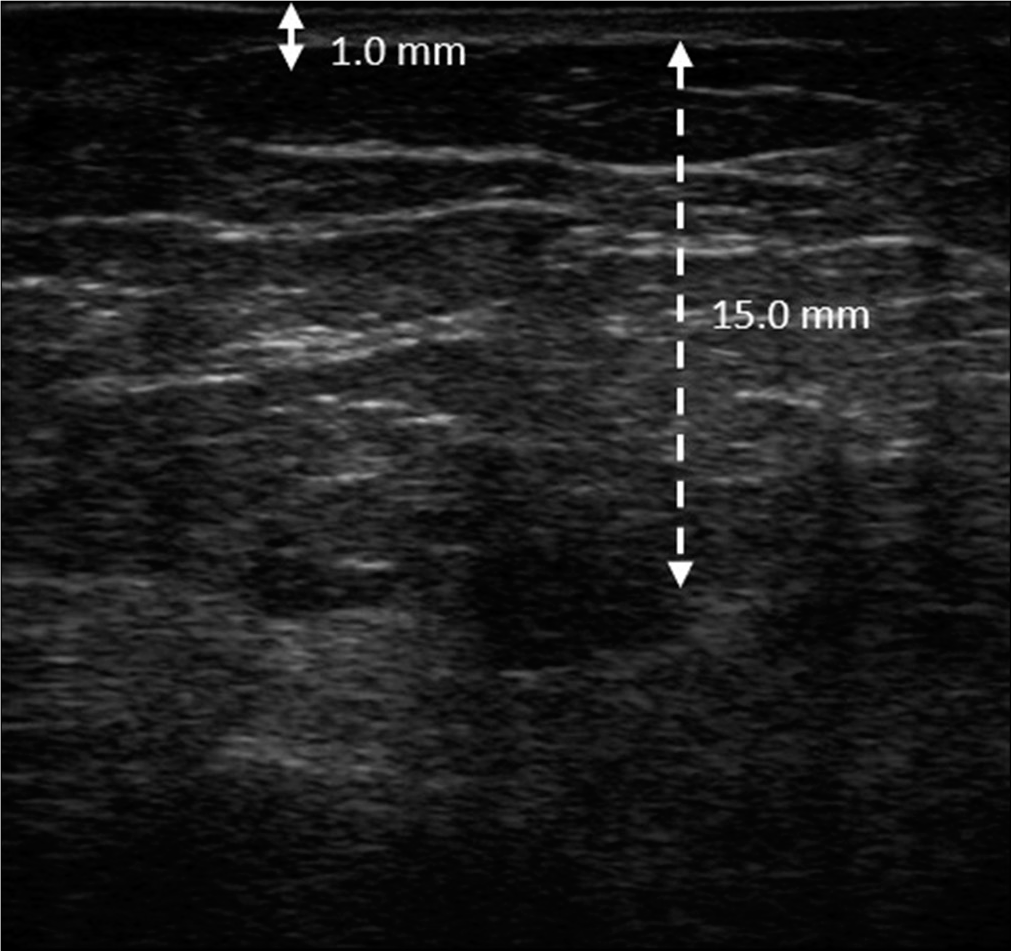
- The thickness of dermis and subcutis were reduced to 1.0 mm (solid double arrow line) and 15.0 mm (dotted double arrow line), respectively, and the echogenicity returned to clear septate fat lobules
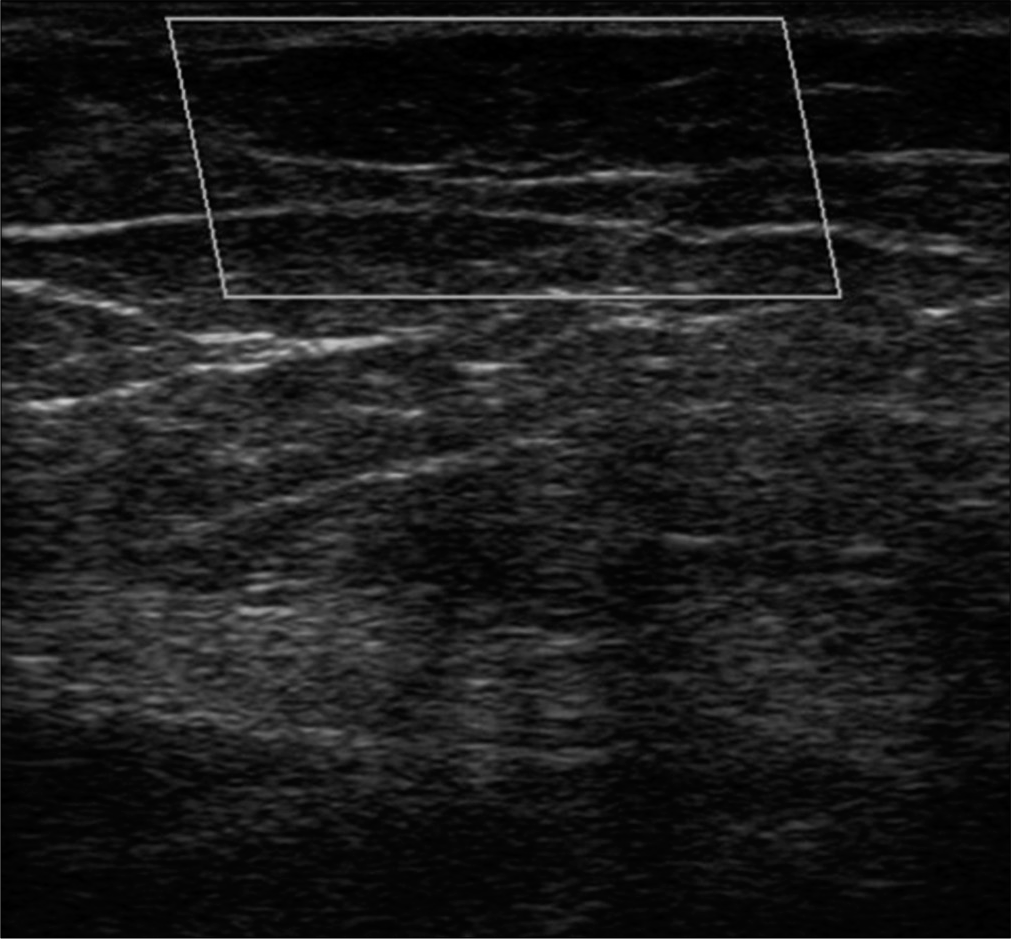
- No increase in blood flow after treatment in color Doppler examination
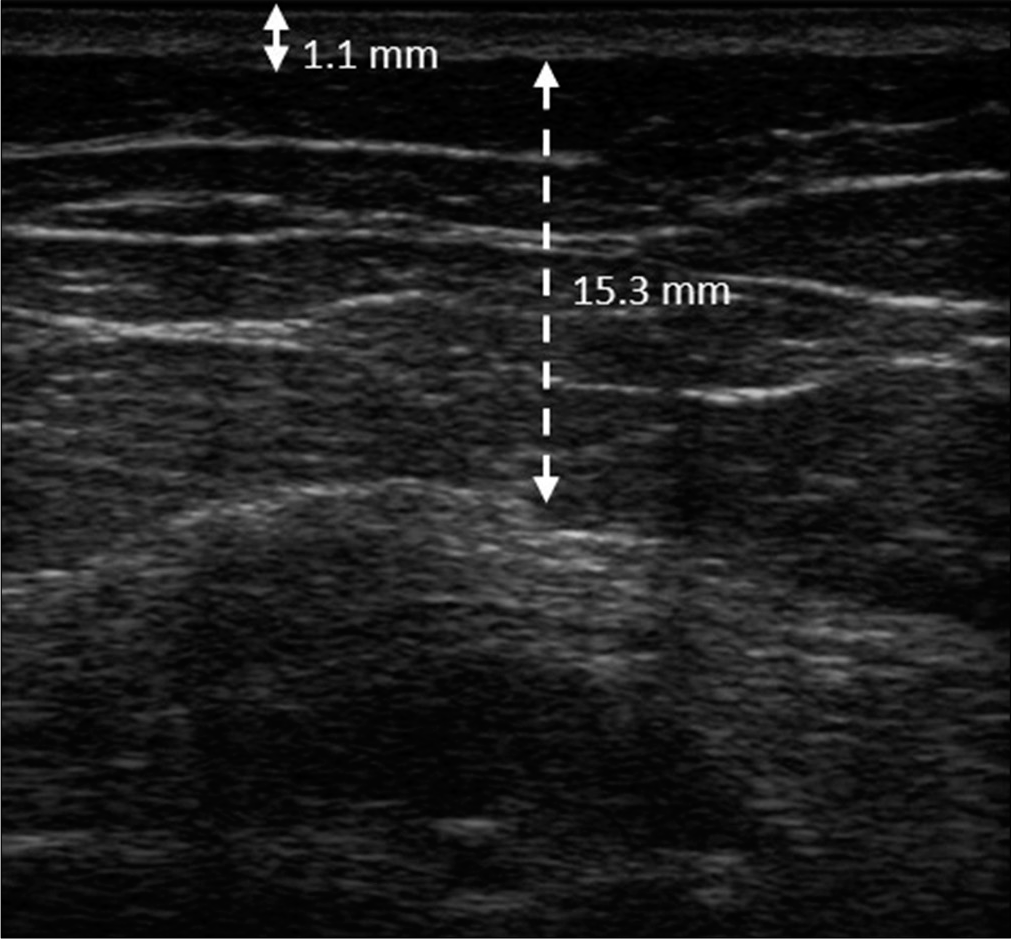
- The thickness of the dermis and subcutaneous layer of the contralateral non-lesional area was 1.1 mm (solid double arrow line) and 15.3 mm (dotted double arrow line), respectively
The clinical and histopathological coexistence of lupus erythematosus panniculitis and localized scleroderma was first described by Umbert and Winkelmann in 1978.1 According to previous studies, type I interferon and interferon-gamma inducible protein-10 play important roles in the inflammatory and fibrotic processes of both cutaneous lupus erythematosus and scleroderma.2 Therefore, the two diseases may have a common dysfunction of the immunological pathway. In addition, some authors also suggest that the lichenoid reaction of cutaneous lupus erythematosus may provoke the fibroblast-dependent reparative dysfunction and cause a sclerosing reaction.3
In a review patients with sclerodermoid lupus erythematosus panniculitis, the linear distribution of cutaneous lesions along the Blaschko’s line was found to be a common feature and this may be related to genetic mosaicism.3 Although the skin lesion in our patient is not in a typical linear pattern, the cutaneous distribution along Blaschko’s line may be regarded as a broadband variant mosaicism.
During recent years, ultrasonography has emerged as a new diagnostic imaging technique in dermatology and can be used to assess the activity of many someskin diseases. According to the previous literature, the ultrasonography of active morphea lesions revealed increase in the thickness of dermis and cutaneous blood flow.4 On the other hand, lobular panniculitis in ultrasonography revealed a diffuse increase in the echogenicity of the fatty lobules with increased hypodermal vascularity.5 In our patient we could find a correlation between the histopathological and ultrasonographic findings. The improvement noted in the ultrasonographic finding after treatment correlated with the clinical improvement in induration and tenderness. The clinical improvement in erythema was associated with the decreased vascularity noted in color Doppler. Besides, we also found that before treatment, the thickness of subcutis in the lesional area was more than the contralateral non-lesional area but there was no significant difference after treatment. However, the value of subcutaneous thickness in monitoring the activity of disease may be limited due to the effect of change in the body weight.
In summary, we report a rare case of sclerodermoid lupus erythematosus panniculitis and its ultrasonographic findings before and after treatment. Ultrasonography may be a useful noninvasive tool for monitoring therapeutic response and for modifying treatment in such patients.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patient has given her consent for her images and other clinical information to be reported in the journal. The patient understands that
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Concurrent localized scleroderma and discoid lupus erythematosusCutaneous 'mixed' or 'overlap' syndrome. Arch Dermatol. 1978;114:1473-8.
- [CrossRef] [PubMed] [Google Scholar]
- Type I interferon-mediated autoimmune diseases: Pathogenesis, diagnosis and targeted therapy. Rheumatology (Oxford). 2017;56:1662-75.
- [CrossRef] [PubMed] [Google Scholar]
- Sclerodermiform linear lupus erythematosus: A distinct entity or coexistence of two autoimmune diseases? J Am Acad Dermatol. 2008;58:665-7.
- [CrossRef] [PubMed] [Google Scholar]
- Activity assessment in morphea using color Doppler ultrasound. J Am Acad Dermatol. 2011;65:942-8.
- [CrossRef] [PubMed] [Google Scholar]
- Sonography of dermatologic emergencies. J Ultrasound Med. 2017;36:1905-14.
- [CrossRef] [PubMed] [Google Scholar]





