Translate this page into:
Ultraviolet-A1 phototherapy in Asian skin: A review of 159 cases in Singapore
2 Department of Occupational and Environmental Medicine, Singapore General Hospital, Singapore
Correspondence Address:
Brian Keng Yong Chia
National Skin Centre, 1 Mandalay Road, Singapore 308205
Singapore
| How to cite this article: Chia BK, Chia GS, Tan ES, Tan VW, Chong WS. Ultraviolet-A1 phototherapy in Asian skin: A review of 159 cases in Singapore. Indian J Dermatol Venereol Leprol 2020;86:162-168 |
Abstract
Background: Ultraviolet-A1 phototherapy has been used to treat many inflammatory dermatoses.
Aims: To determine the efficacy and safety of ultraviolet-A1 phototherapy in Asian skin.
Materials and Methods: We performed a review of records of patients undergoing ultraviolet-A1 phototherapy at our dermatology unit in Singapore from January 2007 to January 2011. Their electronic medical records were reviewed and a standardized questionnaire was filled up for data collection and tabulation. Chi-square or Fisher's exact tests were used to compare the difference in response between various groups for each characteristic. P value of < 0.05 was considered statistically significant.
Results: Our study comprised of 159 patients, of which 103 were patients with hand and foot eczema, 21 with atopic dermatitis, 17 with scleroderma and the remaining with miscellaneous dermatoses. Of these patients, 47.6% of patients with hand and feet eczema had good response after 10 sessions, which increased to 75% after 20 sessions and to 84.6% after 30 sessions. After 10 sessions, 47.6% of patients with atopic dermatitis had good response, which increased to 66.7% after 20 sessions. After 30 sessions, all the three remaining patients with atopic dermatitis experienced good response. For patients with scleroderma, only 11.8 and 10% had good response after 10 and 20 sessions, respectively, which increased to 40% after 30 sessions.
Limitations: Limitations of our study include its retrospective design and, consequently, the lack of standardized treatment protocol, as well as subjective assessment in terms of clinical improvement.
Conclusions: Ultraviolet-A1 phototherapy appears to be efficacious for the treatment of hand and foot eczema as well as atopic dermatitis. However, in patients with scleroderma, the response was partial and needed a longer duration of treatment.
Introduction
The development of lamp-emitting radiation predominantly in the ultraviolet-A1 spectrum was first described in 1981.[1] Efficacy of ultraviolet-A1 phototherapy for the treatment of atopic dermatitis was reported in 1992 and that for localized scleroderma in 1995.[2],[3],[4] Since then, ultraviolet-A1 phototherapy has also been shown to be effective in many inflammatory dermatoses and sclerotic skin diseases such as atopic dermatitis, morphea, systemic sclerosis, extragenital lichen sclerosus, chronic sclerodermic graft versus host disease, polyneuropathy, organomegaly, endocrinopathy, monoclonal gammopathy, skin changes syndrome (POEMS), systemic lupus erythematosus, cutaneous mastocytosis, hypereosinophilic syndrome, granuloma annulare, sarcoidosis, keloids, idiopathic follicular mucinosis, pityriasis lichenoides and cutaneous T-cell lymphoma.[5],[6],[7],[8],[9],[10],[11],[12],[13],[14],[15],[16],[17],[18],[19],[20],[21],[22],[23],[24],[25]
Action of ultraviolet-A1 phototherapy is thought to be mediated through activation of various cellular pathways, including induction of apoptosis of B and T lymphocytes, suppression of proinflammatory cytokines such as interleukin 12 and tumor necrosis factor-alpha, increased levels of collagenase expression in fibroblasts, activation of light-dependent mechanism of DNA repair, as well as modification of endothelial regulation and transformation resulting in neovascularization.[26],[27],[28],[29],[30]
Reports regarding efficacy and safety of ultraviolet A1 phototherapy have largely originated from Europe and the United States in predominantly Caucasian populations.[31],[32] There are only a few published studies regarding ultraviolet A1 phototherapy in Asian populations. The purpose of this study was to evaluate the experience of ultraviolet A1 phototherapy treatment in a predominantly Asian cohort at our dermatology unit in Singapore.
Materials and Methods
Study design
We performed a review of all patients who received ultraviolet A1 phototherapy at our center over a 4-year period from January 2007 to January 2011. Their electronic medical records were reviewed and a standardized questionnaire was filled up for data collection and tabulation. The following information was collected and analyzed: patient demographics including age, sex, race and Fitzpatrick skin type; disease characteristics including diagnosis, duration of the disease and prior treatment received; treatment factors including duration of ultraviolet A1 phototherapy, initial frequency of treatment, starting and final doses of ultraviolet A1, number of sessions before improvement seen, cumulative dose, total number of sessions received, any concomitant therapy received as well as adverse effects experienced. Clinical photographs were reviewed if available [Figure - 1] and [Figure - 2]. This study was approved by the Institutional Review Board.
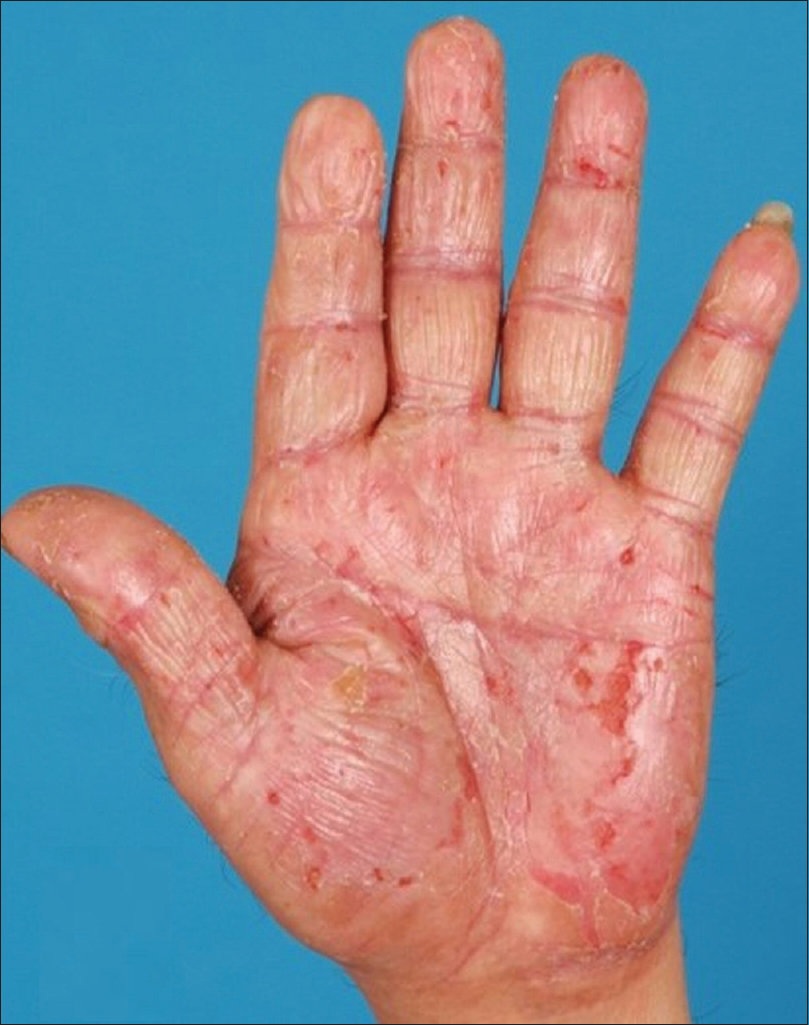 |
| Figure 1: Patient with hand eczema before initiation of ultraviolet-A1 phototherapy |
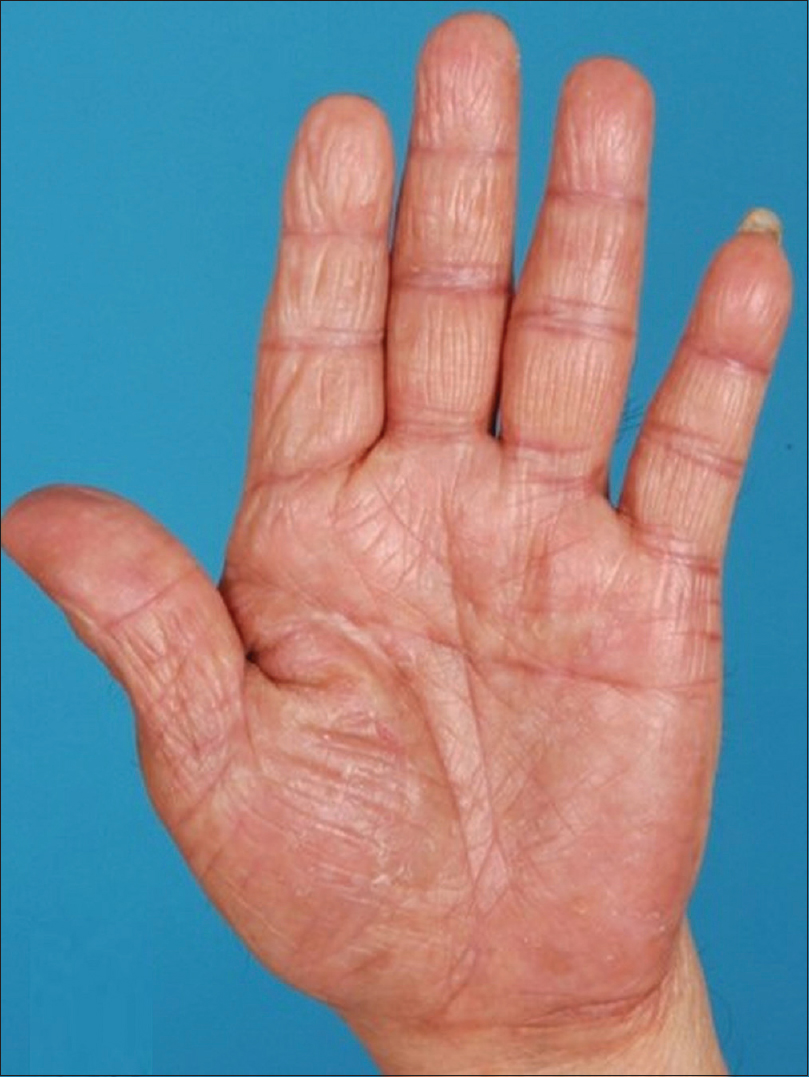 |
| Figure 2: Same patient with hand eczema after 12 weeks of phototherapy, showing moderate to good improvement |
Phototherapy
Patients were treated with low dose (<50 J/cm2), medium dose (50–90 J/cm2) or high dose (100–130 J/cm2) ultraviolet A1 phototherapy; frequency of treatment varying from once to thrice weekly. Frequency, duration and cumulative dose of ultraviolet A1 treatment were determined by the skin condition based on a standardized dosimetry protocol. Irradiation source used was Daavlin SL3000 (Daavlin, Bryan, OH, USA). Ultraviolet radiation spectrum was between 340 and 440 nm with a peak at 375 nm.
Statistical methods
Clinical response after 10, 20 and 30 sessions of ultraviolet A phototherapy was categorized into two groups for analysis: poor to fair (0–50% improvement) and moderate to good (50–100%). Bivariate analyses using Chi-square or Fisher's exact tests (for cell count <5) were used to compare the difference in response between various groups for each characteristic. P value of < 0.05 was considered statistically significant. Statistical analysis was performed using STATA® version 14.
Results
A total of 199 patients received ultraviolet A1 phototherapy during this period for various skin diseases. Of these, 40 patients (20.1%) were lost to follow-up or discontinued treatment after completing less than 10 treatment sessions for reasons other than worsening of their skin disease and were excluded from the study. The reasons for defaulting were lack of time (n = 8), distance constraints (n = 3), poor response (n = 4), switching to a different treatment modality (n = 4), itching experienced during phototherapy (n = 2) and side effect of skin-tanning (n = 1). Eighteen patients defaulted due to unknown reasons. Patients who stopped after <10 treatment sessions owing to worsening of their skin disease were included. The patient cohort comprised the remaining 159 (79.9%) patients.
Patient demographics
A total of 159 (91 female, 68 male) patients were included. Their age ranged from 4 to 84 years (mean 44.7 years old). The ethnic composition of patients was similar to the general population of Singapore[33], with 128 (80.5%) Chinese, 9 (5.7%) Malay, 15 (9.4%) Indian and seven (4.4%) patients belonging to other ethnic groups. Most patients (98.7%, n = 157) had Fitzpatrick skin type IV and he remaining two (1.3%) had type V.
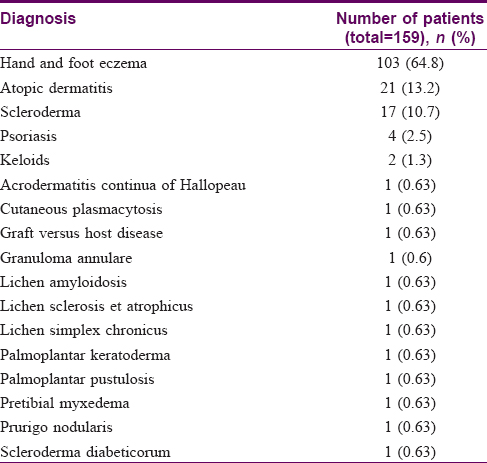
Most patients (n = 103, 64.8%) received ultraviolet-A1 phototherapy for hand and foot eczema and atopic dermatitis (n = 21, 13.2%). A smaller number (n = 17, 10.7%) received treatment for scleroderma and psoriasis (n = 4, 2.5%). The remaining patients received phototherapy for other heterogeneous dermatoses [Table - 1].
The majority of patients (n = 153, 96.2%) received prior treatment, of whom 150 (94.3%) had received previous topical therapy (topical steroids and topical calcineurin inhibitors), fifty six (35.2%) had received previous systemic therapy (immunosuppressant drugs or antibiotics) and 43 (27.0%) had received previous phototherapy such as ultraviolet-B phototherapy or psoralen combined with ultraviolet-A therapy.
Treatment regimens
The median duration of treatment was 15 weeks (range 3–188 weeks) with a median cumulative ultraviolet-A1 dosage of 1345 J/cm2 (range 210–18300 J/cm2). Based on our center's standardized dosimetry protocol, almost all patients (n = 156, 98.1%) were started at low dose of ultraviolet-A1 phototherapy (<50 J/cm2). The initial frequency of treatment was twice-weekly for the majority of patients (n = 121, 76.1%), once per week for four patients (2.5%), three times per week for 31 patients (19.5%), four times per week for one patient (0.63%) and five times per week for two patients (1.26%). We titrated the ultraviolet-A dose received based on response and adverse effects from therapy. Assessments of response and adverse effects from therapy were made before each treatment session by a trained nurse at the phototherapy unit and at three-monthly intervals by a trained dermatologist. Adjustments to the ultraviolet-A dose received were performed. Twenty four patients (15.1%) had low (<50 J/cm2), eighty seven patients (54.7%) had medium (50–90 J/cm2) and 49 patients (30.2%) had a high (100–130 J/cm2) final dose. The ultraviolet-A1 dosimetry used is summarized in [Table - 2].

Clinical response
The clinical response to ultraviolet-A1 phototherapy is summarized in [Table - 3].

Comparison of clinical response of hand and feet eczema with different ultraviolet-A1 treatment regimens
As patients with hand and foot eczema was the group most commonly treated with ultraviolet-A1 in our study, further analysis was carried out to determine their response to different dosing regimens.
Of the patients with hand and foot eczema, 103 patients received at least 10 sessions, sixty four patients received at least 20 sessions and 46 patients received at least 30 sessions of ultraviolet-A1 phototherapy. Of the patients who dropped out after completion of at least 10 sessions of phototherapy, a moderate to good response was seen in 59% of those who dropped out between 10 and 20 sessions and 61.1% of those who dropped out between 20 and 30 sessions respectively. Despite a higher composition of the remaining patients being those with initial poor to fair response, the percentage of patients with moderate to good improvement increased from 47.6% after 10 sessions of phototherapy to 75% after 20 sessions and to 84.8% after 30 sessions of phototherapy. This difference was statistically significant (P< 0.01).
With regard to the dosing regimen, 45 patients (97.8%) were started at an initial low dose regime of <50 J/cm2 based on our center's ultraviolet-A1 phototherapy protocol. 1 patient (2.2%) was started on medium dose regime of 50–90 J/cm2. Of these patients, 6 patients (13%) were maintained on low dose, 23 patients (50%) were switched to medium dose and 17 patients (37%) were switched to a high-dose regime. Frequency of phototherapy was twice-weekly in 39 patients (84.8%), thrice-weekly in 6 patients (13%) and once per week in one patient (2.2%). There was no statistical difference in the clinical response between the patients who received low, medium or high final-dose phototherapy (P = 1.000) with moderate to good response in 5 patients (83.3%) on low final dose, 20 patients (87%) with moderate final dose and 14 patients (82.4%) with high final dose.
Results of ultraviolet-A1 treatment in other skin conditions
The response of other cutaneous disorders to ultraviolet-A1 treatment was more variable. As we had only two patients receiving ultraviolet-A1 treatment for keloids and one patient in each of the other disease categories, we are unable to make definitive conclusions regarding the efficacy of ultraviolet-A1 phototherapy in these diseases. Of the two patients with keloids, only one had fair response after 18 sessions whereas the other had poor response after 32 sessions. Poor response was also seen in patients with lichen amyloidosis, lichen sclerosus et atrophicus, prurigo nodularis, graft versus host disease, pretibial myxedema and cutaneous plasmacytosis. The patient with acrodermatitis of Hallopeau showed fair response. A moderate response was seen in the patient with palmoplantar pustulosis, whereas one patient with palmoplantar keratoderma and another with lichen simplex chronicus showed a good response.
Adverse effects
In general, ultraviolet-A1 phototherapy was well tolerated. The most common adverse effects experienced in our study were itching and erythema, which were experienced by 106 patients (66.7%) and 34 patients (21.4%) respectively. Other adverse effects include tanning in 10 patients (6.3%) as well as pain in 13 patients (8.2%). Two patients had to discontinue treatment owing to itching, whereas one patient discontinued because of tanning. During the course of the study, no patients developed burns, blisters or skin malignancies. The adverse effects experienced by patients are summarized in [Figure - 3].
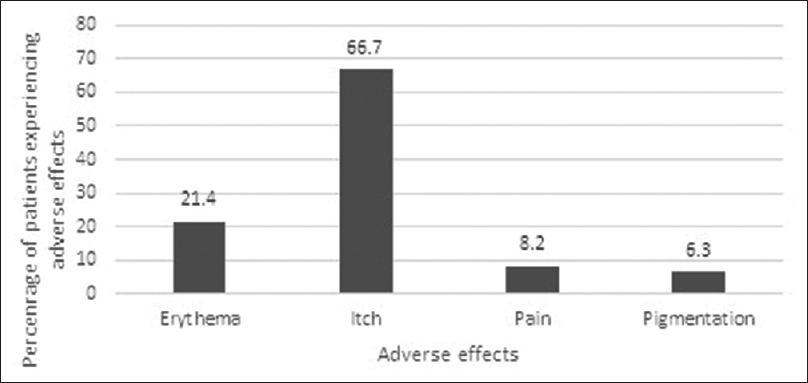 |
| Figure 3: Adverse effects experienced by patients treated with ultraviolet-A1 phototherapy |
Discussion
The most common indications for treatment with ultraviolet-A1 phototherapy in our study were hand and foot eczema, atopic dermatitis and scleroderma. In terms of patient composition, the majority of our patients (64.8%) received ultraviolet-A1 phototherapy for hand and foot eczema. This was in contrast to earlier studies published from the United States and Europe, where the majority of patients had morphea and atopic dermatitis, respectively.[31],[32]
Ultraviolet-A1 phototherapy has been shown to be effective for the treatment of chronic dyshidrotic eczema.[34],[35] In an earlier study published in the United States, the authors reported a moderate to good response rate in 50% of patients with hand and foot eczema.[32] Our study supports these findings with 47.6% of patients hand and foot eczema experiencing moderate to good response to ultraviolet-A1 phototherapy after 10 sessions of treatment. In our study, the treatment efficacy also correlated positively with the duration of phototherapy. Given that a greater proportion of the patients who dropped out were patients with initial moderate to good response, this would suggest that the observation was not a result of patients with poor response being excluded from the analysis. However, the final dose of phototherapy did not have any statistically significant impact on treatment efficacy in patients with hand and feet eczema. These findings are supported by another study by Schmidt et al.,[36] where 10 out of 12 patients experienced good improvement with low-dose ultraviolet-A1 phototherapy of 40 J/cm2. Findings of our study suggest that low-dose ultraviolet-A1 phototherapy over a longer duration is an effective treatment option for patients with hand and foot eczema.
Atopic dermatitis was the second most frequent indication for treatment in our study. Ultraviolet-A1 phototherapy was efficacious for the treatment of atopic dermatitis with 47.6% of patients experiencing moderate to good response after 10 sessions of treatment. This correlated well with the results reported by Tuchinda et al.,[32] who reported moderate to good response in 50% of the patients with atopic dermatitis.[32] The efficacy of ultraviolet-A1 phototherapy for atopic dermatitis has been demonstrated in earlier studies and a previous randomized controlled trial had shown that medium-dose ultraviolet-A1 had greater efficacy than low-dose ultraviolet-A1.[2],[37] In our study, after 10 sessions of phototherapy, two patients had low, fifteen patients had medium and four patients had high final-dose of ultraviolet-A1. Both patients with low final-dose ultraviolet-A1 had only poor to fair response, whereas 53.3% of patients with medium and 50% of patients with high doses had moderate to good response. But probably due to the small numbers, the difference between these groups was not statistically significant. Compared with patients with hand and foot eczema, it would suggest that patients with atopic dermatitis may require a higher dose of ultraviolet-A1 phototherapy for good response.
Our patients with scleroderma had an initially poor response to ultraviolet-A1 phototherapy- moderate to good response seen in 11.8% and 10% of patients after 10 and 20 sessions of, treatment respectively, which increased to 40% after 30 sessions. Tuchinda et al. reported better results (37.8% of patients showing moderate to good response) with medium- to high-dose ultraviolet-A1 phototherapy compared to low-dose phototherapy in morphoea.[32] Owing to the small number of patients, we were unable to assess if the response to ultraviolet-A1 was dose-dependent. However, the results of our study do suggest that for patients with scleroderma receiving ultraviolet-A1 phototherapy, a longer duration of treatment may be required compared to patients with other dermatological conditions.
Several other dermatoses including keloids, lichen sclerosus et atrophicus, graft versus host disease and granuloma annulare have been reported to improve with ultraviolet-A1 phototherapy.[11],[13],[14],[19],[21],[38] Our study included small numbers of patients with heterogenous skin disorders who underwent treatment with ultraviolet-A1 phototherapy and this is summarized in [Table - 1]. With the exception of patients with lichen simplex chronicus, palmoplantar keratoderma and palmoplantar pustulosis, other patients experienced only poor to fair response. A recent paper reported that ultraviolet-A1 phototherapy could be an effective treatment for palmoplantar pustulosis.[39] Mechanism of action of ultraviolet-A1 in the treatment of lichen simplex chronicus and palmoplantar keratoderma is unknown. Improvement of pruritus by inducing a decrease in dermal mast cells is a possible explanation.[6]
Ultraviolet-A1 phototherapy was generally well tolerated in our study. The most common adverse effect experienced in our study cohort was itching, which occurred in about 66.7% of patients and led to discontinuation of the study in two patients. The proportion of patients experiencing pruritus was more in our study, compared with the study by Tuchinda et al.(15%).,[32] We postulate that one possible reason could be the hotter and more humid environment in Singapore. Other adverse effects experienced by patients in our study included erythema (21.4%), tanning (6.3%), as well as pain (8.2%). During the course of the study, no serious adverse side effects such as skin malignancies were observed, a finding that is consistent with previous studies.[5],[32],[40],[41]
To the authors' knowledge, this study is the largest retrospective study that used ultraviolet-A1 phototherapy in an Asian cohort. Our study limitations include its retrospective design and consequently the lack of standardized treatment protocol, as well as subjective assessment in terms of clinical improvement. There was also a substantial number of patients who were lost to follow-up, leading to difficulties in analyzing the impact of treatment duration on clinical improvement. We attempted to mitigate this impact by analyzing the clinical response of these patients before discontinuation of therapy. Besides hand and foot eczema, atopic dermatitis, scleroderma and psoriasis, our study included very few patients with other dermatoses to draw meaningful conclusions. Studies involving larger number of patients will be required to ascertain the effectiveness of ultraviolet-A1 phototherapy in those conditions.
Conclusions
Our study demonstrated the efficacy of ultraviolet-A1 phototherapy in hand and foot eczema, atopic dermatitis and scleroderma. In hand and foot eczema, clinical response seems to be relatively independent of dose, though a longer duration of treatment correlates with increased treatment efficacy. In atopic dermatitis, a higher dosage of phototherapy may produce a better clinical response. For scleroderma, a longer duration of treatment appears to be required to achieve treatment efficacy.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patients have given their consent for their images and other clinical information to be reported in the journal. The patient understands that name and initials will not be published and due efforts will be made to conceal identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. |
Mutzhas MF, Hölzle E, Hofmann C, Plewig G. A new apparatus with high radiation energy between 320-460 nm: Physical description and dermatological applications. J Invest Dermatol 1981;76:42-7.
[Google Scholar]
|
| 2. |
Krutmann J, Czech W, Diepgen T, Niedner R, Kapp A, Schöpf E. High-dose UVA1 therapy in the treatment of patients with atopic dermatitis. J Am Acad Dermatol 1992;26:225-30.
[Google Scholar]
|
| 3. |
Krutmann J, Schöpf E. High-dose-UVA1 phototherapy: A novel and highly effective approach for the treatment of acute exacerbation of atopic dermatitis. Acta Derm Venereol Suppl (Stockh) 1992;176:120-2.
[Google Scholar]
|
| 4. |
Kerscher M, Volkenandt M, Gruss C, Reuther T, von Kobyletzki G, Freitag M, et al. Low-dose UVA phototherapy for treatment of localized scleroderma. J Am Acad Dermatol 1998;38:21-6.
[Google Scholar]
|
| 5. |
Krutmann J, Diepgen TL, Luger TA, Grabbe S, Meffert H, Sönnichsen N, et al. High-dose UVA1 therapy for atopic dermatitis: Results of a multicenter trial. J Am Acad Dermatol 1998;38:589-93.
[Google Scholar]
|
| 6. |
Grabbe J, Welker P, Humke S, Grewe M, Schöpf E, Henz BM, et al. High-dose ultraviolet A1 (UVA1), but not UVA/UVB therapy, decreases IgE-binding cells in lesional skin of patients with atopic eczema. J Invest Dermatol 1996;107:419-22.
[Google Scholar]
|
| 7. |
Camacho NR, Sánchez JE, Martin RF, González JR, Sánchez JL. Medium-dose UVA1 phototherapy in localized scleroderma and its effect in CD34-positive dendritic cells. J Am Acad Dermatol 2001;45:697-9.
[Google Scholar]
|
| 8. |
Gruss C, Stücker M, Kobyletzki G, Schreiber D, Altmeyer P, Kerscher M, et al. Low dose UVA1 phototherapy in disabling pansclerotic morphoea of childhood. Br J Dermatol 1997;136:293-4.
[Google Scholar]
|
| 9. |
de Rie MA, Enomoto DN, de Vries HJ, Bos JD. Evaluation of medium-dose UVA1 phototherapy in localized scleroderma with the cutometer and fast Fourier transform method. Dermatology 2003;207:298-301.
[Google Scholar]
|
| 10. |
Kreuter A, Breuckmann F, Uhle A, Brockmeyer N, Von Kobyletzki G, Freitag M, et al. Low-dose UVA1 phototherapy in systemic sclerosis: Effects on acrosclerosis. J Am Acad Dermatol 2004;50:740-7.
[Google Scholar]
|
| 11. |
Kreuter A, Jansen T, Stücker M, Herde M, Hoffmann K, Altmeyer P, et al. Low-dose ultraviolet-A1 phototherapy for lichen sclerosus et atrophicus. Clin Exp Dermatol 2001;26:30-2.
[Google Scholar]
|
| 12. |
Kreuter A, Gambichler T, Avermaete A, Happe M, Bacharach-Buhles M, Hoffmann K, et al. Low-dose ultraviolet A1 phototherapy for extragenital lichen sclerosus: Results of a preliminary study. J Am Acad Dermatol 2002;46:251-5.
[Google Scholar]
|
| 13. |
Grundmann-Kollmann M, Behrens S, Gruss C, Gottlöber P, Peter RU, Kerscher M. Chronic sclerodermic graft-versus-host disease refractory to immunosuppressive treatment responds to UVA1 phototherapy. J Am Acad Dermatol 2000;42:134-6.
[Google Scholar]
|
| 14. |
Ständer H, Schiller M, Schwarz T. UVA1 therapy for sclerodermic graft-versus-host disease of the skin. J Am Acad Dermatol 2002;46:799-800.
[Google Scholar]
|
| 15. |
Schaller M, Romiti R, Wollenberg A, Prinz B, Woerle B. Improvement of cutaneous manifestations in POEMS syndrome after UVA1 phototherapy. J Am Acad Dermatol 2001;45:969-70.
[Google Scholar]
|
| 16. |
Molina JF, McGrath H Jr. Longterm ultraviolet-A1 irradiation therapy in systemic lupus erythematosus. J Rheumatol 1997;24:1072-4.
[Google Scholar]
|
| 17. |
Stege H, Schöpf E, Ruzicka T, Krutmann J. High-dose UVA1 for urticaria pigmentosa. Lancet 1996;347:64.
[Google Scholar]
|
| 18. |
Plötz SG, Abeck D, Seitzer U, Hein R, Ring J. UVA1 for hypereosinophilic syndrome. Acta Derm Venereol 2000;80:221.
[Google Scholar]
|
| 19. |
Muchenberger S, Schöpf E, Simon JC. Phototherapy with UV-A-I for generalized granuloma annulare. Arch Dermatol 1997;133:1605.
[Google Scholar]
|
| 20. |
Mahnke N, Medve-Koenigs K, Berneburg M, Ruzicka T, Neumann NJ. Cutaneous sarcoidosis treated with medium-dose UVA1. J Am Acad Dermatol 2004;50:978-9.
[Google Scholar]
|
| 21. |
Asawanonda P, Khoo LS, Fitzpatrick TB, Taylor CR. UV-A1 for keloid. Arch Dermatol 1999;135:348-9.
[Google Scholar]
|
| 22. |
von Kobyletzki G, Kreuter JA, Nordmeier R, Stücker M, Altmeyer P. Treatment of idiopathic mucinosis follicularis with UVA1 cold light phototherapy. Dermatology 2000;201:76-7.
[Google Scholar]
|
| 23. |
Pinton PC, Capezzera R, Zane C, De Panfilis G. Medium-dose ultraviolet A1 therapy for pityriasis lichenoides et varioliformis acuta and pityriasis lichenoides chronica. J Am Acad Dermatol 2002;47:410-4.
[Google Scholar]
|
| 24. |
Zane C, Leali C, Airò P, De Panfilis G, Pinton PC. “High-dose” UVA1 therapy of widespread plaque-type, nodular, and erythrodermic mycosis fungoides. J Am Acad Dermatol 2001;44:629-33.
[Google Scholar]
|
| 25. |
Dawe RS. Ultraviolet A1 phototherapy. Br J Dermatol 2003;148:626-37.
[Google Scholar]
|
| 26. |
Godar DE. UVA1 radiation triggers two different final apoptotic pathways. J Invest Dermatol 1999;112:3-12.
[Google Scholar]
|
| 27. |
Skov L, Hansen H, Allen M, Villadsen L, Norval M, Barker JN, et al. Contrasting effects of ultraviolet A1 and ultraviolet B exposure on the induction of tumour necrosis factor-alpha in human skin. Br J Dermatol 1998;138:216-20.
[Google Scholar]
|
| 28. |
Gruss C, Reed JA, Altmeyer P, McNutt NS, Kerscher M. Induction of interstitial collagenase (MMP-1) by UVA-1 phototherapy in morphea fibroblasts. Lancet 1997;350:1295-6.
[Google Scholar]
|
| 29. |
Stege H, Roza L, Vink AA, Grewe M, Ruzicka T, Grether-Beck S, et al. Enzyme plus light therapy to repair DNA damage in ultraviolet-B-irradiated human skin. Proc Natl Acad Sci U S A 2000;97:1790-5.
[Google Scholar]
|
| 30. |
Breuckmann F, Stuecker M, Altmeyer P, Kreuter A. Modulation of endothelial dysfunction and apoptosis: UVA1-mediated skin improvement in systemic sclerosis. Arch Dermatol Res 2004;296:235-9.
[Google Scholar]
|
| 31. |
Rombold S, Lobisch K, Katzer K, Grazziotin TC, Ring J, Eberlein B. Efficacy of UVA1 phototherapy in 230 patients with various skin diseases. Photodermatol Photoimmunol Photomed 2008;24:19-23.
[Google Scholar]
|
| 32. |
Tuchinda C, Kerr HA, Taylor CR, Jacobe H, Bergamo BM, Elmets C, et al. UVA1 phototherapy for cutaneous diseases: An experience of 92 cases in the United States. Photodermatol Photoimmunol Photomed 2006;22:247-53.
[Google Scholar]
|
| 33. |
Kim WW. Population trends. In: Department of Statistics Singapore MoTI, Republic of Singapore, editor. Singapore 2017.
[Google Scholar]
|
| 34. |
Petering H, Breuer C, Herbst R, Kapp A, Werfel T. Comparison of localized high-dose UVA1 irradiation versus topical cream psoralen-UVA for treatment of chronic vesicular dyshidrotic eczema. J Am Acad Dermatol 2004;50:68-72.
[Google Scholar]
|
| 35. |
Polderman MC, Govaert JC, Le Cessie S, Pavel S. A double-blind placebo-controlled trial of UVA-1 in the treatment of dyshidrotic eczema. Clin Exp Dermatol 2003;28:584-7.
[Google Scholar]
|
| 36. |
Schmidt T, Abeck D, Boeck K, Mempel M, Ring J. UVA1 irradiation is effective in treatment of chronic vesicular dyshidrotic hand eczema. Acta Derm Venereol 1998;78:318-9.
[Google Scholar]
|
| 37. |
Kowalzick L. UVA1 for atopic dermatitis: Medium dose superior to low dose. J Am Acad Dermatol 2001;44:548.
[Google Scholar]
|
| 38. |
Kreuter A, von Kobyletzki G, Happe M, Herde M, Breuckmann F, Stücker M, et al. Ultraviolet-A1 (UVA1) phototherapy in lichen sclerosus et atrophicus. Hautarzt 2001;52:878-81.
[Google Scholar]
|
| 39. |
Su LN, Xu X, Tang L, Yu N, Ding YF. UVA1 phototherapy in the treatment of palmoplantar pustulosis: A pilot prospective study. Lasers Med Sci 2016;31:1641-3.
[Google Scholar]
|
| 40. |
von Kobyletzki G, Pieck C, Hoffmann K, Freitag M, Altmeyer P. Medium-dose UVA1 cold-light phototherapy in the treatment of severe atopic dermatitis. J Am Acad Dermatol 1999;41:931-7.
[Google Scholar]
|
| 41. |
Tzaneva S, Seeber A, Schwaiger M, Hönigsmann H, Tanew A. High-dose versus medium-dose UVA1 phototherapy for patients with severe generalized atopic dermatitis. J Am Acad Dermatol 2001;45:503-7.
[Google Scholar]
|
Fulltext Views
3,238
PDF downloads
2,349





