Translate this page into:
Viva questions from the IJDVL
2 Department of Dermatology, K J Somaiya Medical College and Research Centre, Mumbai, Maharashtra, India
Correspondence Address:
Vishalakshi Viswanath
Department of Dermatology, Rajiv Gandhi Medical College, Thane, Maharashtra
India
| How to cite this article: Viswanath V, Vasani R. Viva questions from the IJDVL. Indian J Dermatol Venereol Leprol 2015;81:550-555 |
Spotter - Infantile Hemangiomas [Figure - 1]A-C
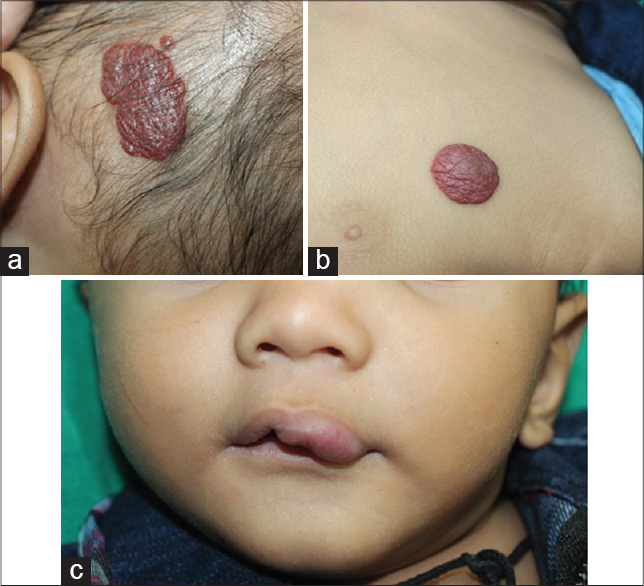 |
| Figure 1: (a-c) Multiple hemangiomas on the scalp, chest and lip in an infant (Courtesy: Department of Dermatology, Rajiv Gandhi Medical College, Thane, Maharashtra, India) |
How are infantile hemangiomas classified?
Infantile hemangiomas can be classified based on their location and extent of involvement [Table - 1] and [Table - 2].


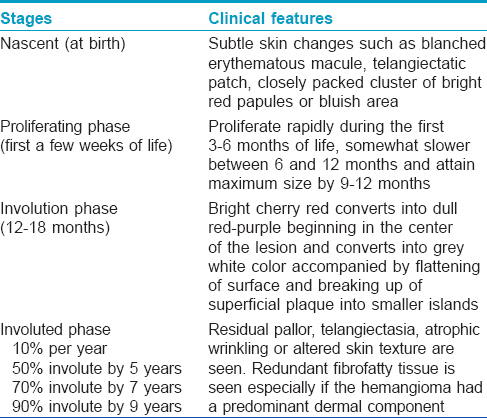
What are stages of an infantile hemangioma?
The stages of an infantile hemangioma include the nascent phase, proliferating phase, involution phase and the involuted phase [Table - 3].
What are the complications associated with infantile hemangiomas?
The complications associated with infantile hemangioma include:
- Bleeding
- Infection
- Ulceration
- Impingement of vital structures that interfere with vision, eating or hearing. For example:
- Hemangioma on the tip of the nose - Cyrano nose deformity
- Pinna or helix involvement - It can ulcerate and can cause structural deformity
- Periorbital or lid hemangioma - Astigmatism, deformation of cornea or refractory errors
- External auditory canal - Conductive hearing loss
- Lip involvement - Difficulty in suckling
- Kasabach-Meritt syndrome (hemangioma with severe thrombocytopenia)
- Consumption coagulopathy
- With disseminated disease, the possible complications are high output cardiac failure, visceral hemorrhage, hydrocephalus, jaundice, anemia, thrombocytopenia, thyroid and ocular anomalies
- Segmental hemangiomas over the mandible, chin and upper part of the neck have a high risk of concomitant upper airway and subglottic area involvement.
What are the indications for early active treatment for hemangiomas?
The indications for early and active treatment for hemangiomas include:
- Life-threatening complications such as airway obstruction, congestive cardiac failure, gastrointestinal bleeding
- Hemangiomas that are likely to cause difficulty in feeding, urination, defecation or gross impairment of hearing or vision
- Hemangiomas over nose, lip, glabellar area, ear are likely to result in disfigurement
- Ulcerated hemangiomas
- Large facial hemangiomas
- Hemangiomas with a large dermal component
- Exophytic hemangiomas are likely to leave a significant fibrofatty residue.
What are the treatment options for hemangioma?
Various systemic, topical and intralesional treatment modalities are now available for treating infantile hemangiomas. Systemic treatments chiefly include corticosteroids and propranolol. [Table - 4] and [Table - 5]. Topical treatments have been summarized in [Table - 6]. Interferon α, vincristine and cyclophosphamide are used for life-threatening hemangiomas unresponsive to conventional therapy. Other agents that have been used include intralesional corticosteroids, topical becaplermin, local biphosphonates, intralesional bleomycin, injectable pingyanmycin, vascular endothelial growth factors.



Surgical excision can be employed in the case of involuted lesions to remove the fibrofatty tissue and redundant skin and in cases of sharply demarcated, localized, exophytic and pedunculated lesions with a prominent dermal component. Chronically ulcerated hemangiomas can also be subjected to surgical excision.
Lasers which are used for hemangiomas include pulsed dye laser; frequency doubled neodymium-doped yttrium aluminum garnet laser, potassium titanyl phosphate laser and the argon laser.
How do you manage an ulcerated hemangioma?
An occlusive environment is created with petrolatum. Polyurethane or hydrocolloid dressing and application of topical antibiotic such as mupirocin, or metronidazole and clindamycin especially in the periorificial areas is beneficial. Oral analgesics, oral antibiotics and topical lignocaine can be used to alleviate the pain.
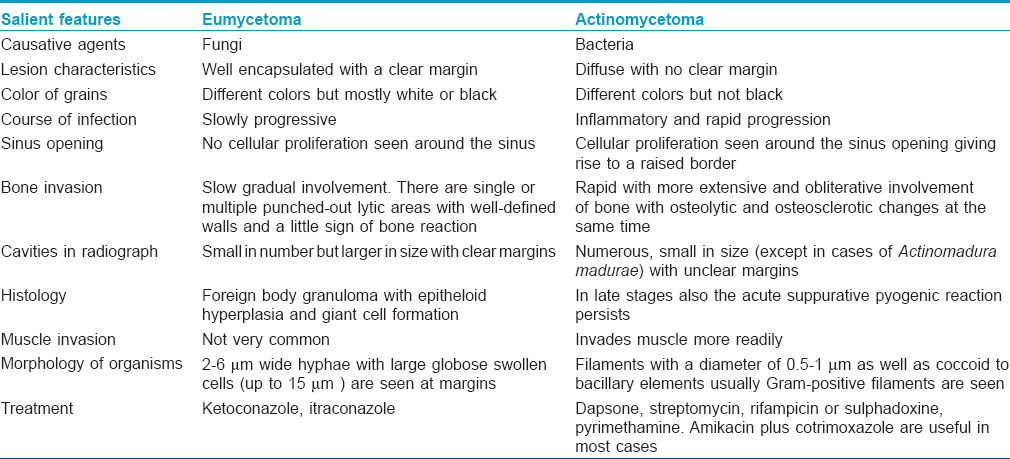
Enumerate the Differences Between Eumycetoma and Actinomycetoma
[Table - 7] summarizes the differences between eumycetoma and actinomycetoma.
Rosacea Subtypes
What are the various types of rosacea?
The various types of rosacea include:
- Erythematotelangiectatic variant
- Papulopustular - Small papules, pustules and even nodules resembling acne
- Phymatous rosacea - Overgrowth and hyperplasia of sebaceous glands in certain facial areas causing disfigurement. The phymas associated with rosacea are rhinophyma, otophyma, gnathophyma, metaphyma and blepharophyma
- Ocular rosacea - Can cause blepharitis and conjunctivitis
- Other variants of rosacea include rosacea fulminans and granulomatous rosacea
- Rosacea fulminans - Sudden eruption of cystic nodules, papules, pustules over the chin, cheeks and forehead
- Granulomatous rosacea - Erythematous and monomorphic papules and nodules in a periorificial location.
SYRINGOMAS
What are the presentations of syringomas?
Classically, syringomas are multiple and most frequently located on the eyelids and upper cheeks. Other presentations of syringomas include:
- A localized form
- A form associated with Down syndrome
- A generalized form that encompasses multiple and eruptive syringomas
- A familial form.
Localized lesions can be present:
- Over the axillae, abdomen, forehead
- Localized to the penis or vulva
- Unilateral or linear syringomas
- Over the distal extremities in a symmetric pattern.
What is the histopathology of syringoma?
Syringoma is located in the superficial dermis and is composed of numerous small ducts located in the sclerotic stroma. The dilated cystic spaces within the dermis are lined by two layers of cuboidal to flattened epithelial cells and have a lumen containing periodic acid-Schiff-positive, eosinophilic and amorphous debris. Some of the ducts have elongated tails of epithelial cells, producing a characteristic comma-shaped or tadpole or paisley tie appearance.
Pyoderma Gangrenosum
What are the types of pyoderma gangrenosum?
Pyoderma gangrenosum can manifest as the classic, ulcerative form or as various atypical forms:
- Classic/ulcerative pyoderma gangrenosum: This is the commonest and the best recognized variant of pyoderma gangrenosum, presenting with small tender, red blue papules, plaques or pustules that evolve into painful ulcers with characteristic violaceous undermined edges. Lesions heal with atrophic, cribriform scars
- Atypical forms:
- Pustular pyoderma gangrenosum - associated with inflammatory bowel disease
- Bullous pyoderma gangrenosum - associated with myeloproliferative disorders
- Vegetative type or superficial granulomatous pyoderma - usually not associated with an underlying systemic cause.
What are the disorders associated with pyoderma gangrenosum?
A few important associations of pyoderma gangrenosum are enumerated:
- Gastrointestinal - Ulcerative colitis, Crohn′s disease, chronic active hepatitis, primary biliary cirrhosis
- Rheumatological - Rheumatoid arthritis, enteropathic arthritis, osteoarthritis
- Hematological disorders - Myeloid leukemia, myeloma, polycythemia vera, myelofibrosis
- Connective tissue disorders - Systemic lupus erythematosus, Takayasu′s arteritis, Wegener′s granulomatosis, Bechet′s disease
- Infections - HIV, HCV, Chlamydia pneumoniae
- Drugs - Isotretinoin, Granulocyte-colony stimulating factor
- Other diseases - Solid tumors, monoclonal gammopathy, antiphospholipid antibody syndrome, diabetes mellitus.
Verrucous Hemangioma versus Angiokeratoma Circumscriptum
What are the salient features which differentiate a verrucous hemangioma from angiokeratoma circumscriptum?
Verrucous hemangioma and angiokeratoma circumscriptum are rare, localized, vascular malformations. Angiokeratoma circumscriptum is usually present at birth; verrucous hemangioma appears at birth or in early childhood. Histologically both these conditions are characterized by multiple dilated blood vessels in the dermis with overlying epidermal hyperplasia. Angiokeratoma circumscriptum involves only the papillary dermis while in verrucous hemangioma, the blood vessels extend into the deep dermis and subcutaneous fat and it may also show capillary endothelial proliferation.
Lipoid Proteinosis
What is the mode of inheritance and basic defect in lipoid proteinosis?
Lipoid proteinosis (Urbach and Wiethe disease or hyalinosis cutis et mucosae or lipoglycoproteinosis) is a rare genodermatosis inherited by autosomal recessive mode of inheritance and was first described in 1929.
It occurs due to mutations in the extracellular matrix gene 1 on chromosome 1q21. The basic defect is the deposition of hyaline material in the skin, oral cavity, larynx and internal organs. The periodic acid-Schiff-positive hyaline-like material is deposited in the dermis, around blood vessels and adnexae with resultant skin thickening, abnormal scarring, abnormal wound healing and premature aging.
Describe the clinical features of lipoid proteinosis
The onset of lipoid proteinosis is usually in early infancy, heralded by hoarseness of voice due to laryngeal infiltration. The vocal cords, lips, pharynx, soft palate, uvula and tonsils show deposition of yellowish white infiltrates. The tongue may be firm and enlarged with restrictive movements due to the short and thick frenulum.
The characteristic feature is the firm, knobby papules distributed on upper and lower eyelids (moniliform papules) and lid margins giving a beaded appearance. Other ocular manifestations include corneal opacities and secondary glaucoma. Diffuse skin infiltration may result in yellowish or waxy papules on knuckles, elbows and knees which are followed by nodules or plaques or pock-like scars. Blistering in early childhood may be followed by erosions, crusting and varioliform scar formation.
Neurological manifestations include epilepsy, schizophrenia and memory loss. Associated diabetes may be due to deposition of amorphous material in the capillary vessels or in pancreas, deposition of hyaline material in the intestine may result in intestinal bleeding.
Newer Carrier Agents
Enumerate the various carrier agents used in topical antifungal preparations
The various carrier agents include the following:
- Micelles: A group of surfactant molecules dispersed in a liquid
- Lipidic systems:
- Solid lipid nanoparticles: Carriers in which the drug is entrapped within a solid lipid core matrix
- Nanostructured lipid carriers: Second generation of lipid nanoparticles in which the matrix is composed of a mixture of solid and liquid lipids
- Microemulsions: Thermodynamically stable mixtures of oil and water stabilized by surfactants and co-surfactants with size in the nanometer range
- Vesicular delivery systems:
- Liposomes: Vesicles which consist of one or more concentric lipid bilayers separated by water or aqueous buffer compartments, ranging in size from 10 nanometers to 20 micrometers
- Niosomes: Similar to liposomes, they only differ in the replacement of phospholipids with non-ionic surfactants
- Transfersomes: Also termed as ultradeformable or flexible liposomes. These are formed of phospholipids and an edge activator; which is a surfactant with a high radius of curvature (that destabilizes the phospholipid lipid bilayers and increases the deformability of vesicles)
- Ethosomes: Another type of deformable vesicles which contain ethanol instead of edge activator
- Penetration enhancer vesicles: New elastic vesicular system prepared by penetration enhancers with or without soybean lecithin.
What are the advantages of using carrier agents in topical antifungal preparations?
Carrier systems have the ability to overcome the immediate drug release caused by conventional formulations. Hence they avoid the possibility of induction of allergic reactions. These agents improve topical drug delivery by either finding a way into a shunt such as hair follicle, accumulating between corneocytes, intermingling with skin lipids, or by disintegrating and merging with lipidic layers. They enhance the penetration of the antifungal agents, leading to more effective treatment.
Disorders Induced by Tattooing
Enumerate the disorders which can be induced by tattooing
Tattooing exposes an individual to various infective and non-infective dermatoses.
The infective disorders associated with tattooing include viral warts, syphilis, leprosy, hepatitis B, hepatitis C, HIV, molluscum contagiosum and those caused by methicillin-resistant Staphylococcus aureus. Inoculation tuberculosis and non-tuberculous mycobacterial infections caused by Mycobacterium haemophilum, Mycobacterium chelonae and Mycobacterium fortuitum at the tattoo site have also been described.
Allergic reactions to the tattoo pigment may manifest as sudden onset of irritation, swelling and redness in a part of the tattoo; these may develop within a few weeks or after many years. The allergic reaction may be localized or generalized; it may manifest as an eczematous eruption or erythroderma. Irritant reactions, lichenoid tattoo reactions or scarring can occur.
Certain skin diseases may be localized to areas of tattooing - lichen planus, psoriasis, sarcoidal granuloma, foreign body granulomas and rarely lupus erythematosus and melanoma.
Biologics versus Biosimilars
What are biosimilars?
Biosimilars are copies of the innovator biological agents and represent an important advance in the field of biological therapeutics. These are also known as "follow-on biologics" in the USA and "subsequent entry biologics" in Canada.
Essentially, a "biosimilar" is a biological agent similar to an already approved or licensed-for-use biological medicine, also known as the "reference product." By definition, a biosimilar will have a similar safety and efficacy profile as its reference product and is generally used to treat the same conditions. Although they are similar to the original biologic, there are certain differences in terms of structure, efficacy, safety and immunogenicity. Biosimilars, however, cannot be regarded as biogenerics or chemical generics.
What are the differences in the regulatory pathway for biosimilars and chemical generics?
The requirements for approval of biosimilars are more stringent than that for licensing of simpler generic drugs:
- Quality testing is performed on multiple batches for biosimilars (usually three or more), whereas it is performed on only one batch in a chemical generic
- Preclinical testing in biosimilars involves in-vitro characterization comparative to the reference product and animal model testing, whereas these are not needed for generics
- Phase III trials to compare with reference drug are essential for biosimilars and are not needed for generics.
However, the regulatory process involved for biosimilars is much shorter and less tedious compared to the original biologic.
Which are the biosimilars currently available in India?
Biosimilars to rituximab, etanercept and infliximab are currently available in India.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Fulltext Views
4,393
PDF downloads
2,399





