Translate this page into:
Viva questions from the IJDVL
2 Department of Dermatology, K J Somaiya Medical College and Research Centre, Mumbai, Maharashtra, India
Correspondence Address:
Vishalakshi Viswanath
Department of Dermatology, Rajiv Gandhi Medical College, Thane, Maharashtra
India
| How to cite this article: Viswanath V, Vasani R. Viva questions from the IJDVL. Indian J Dermatol Venereol Leprol 2017;83:137-143 |
Focus Article
Spot the diagnosis [Figure - 1]a and [Figure - 1]b and discuss the clinical types?
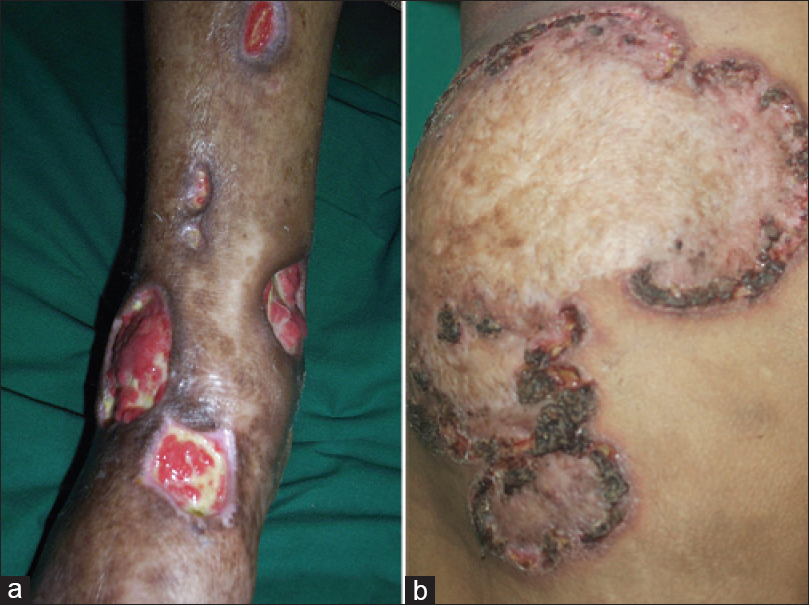 |
| Figure 1: (a and b) Spot the diagnosis. Courtesy- Department of Dermatology, Rajiv Gandhi Medical College, Thane, Maharashtra, India |
The diagnosis is pyoderma gangrenosum: (a) ulcerative and (b) vegetative or superficial granulomatous spreading type.
Pyoderma gangrenosum has varied clinical presentations. These include:
Ulcerative (classic) pyoderma gangrenosum
- Most common type
- Necrotic and mucopurulent tender ulcer with an edematous, violaceous, serpinginously expanding, undermined border
- Usual site is the lower limb
- Two patterns of clinical progression: explosive and rapidly progressive or indolent and gradually progressive.
Pustularpyoderma gangrenosum
- Pustules do not evolve into ulcers
- Painful lesions occur mainly on the extensor aspects of extremities and upper trunk
- Associated with inflammatory bowel disease.
Atypical or bullous pyoderma gangrenosum
- Rapidly evolving vesicles and bullae with central necrosis and erosion with an areola of erythema
- Usual sites are face and arms
- Associated with myeloproliferative diseases.
Vegetative pyoderma gangrenosum/superficial granulomatous spreading pyoderma gangrenosum
- Localized variant
- Non-aggressive form of pyoderma gangrenosum seen in the head and neck areas.
Peristomal pyoderma gangrenosum
- Seen around enterostomy/colostomy in patients with inflammatory bowel disease
- Possible pathergy phenomenon due to irritation to the peristomal skin caused by leakage of feces or adhesive stomal appliance.
Genital pyoderma gangrenosum
- Genital involvement can occur with lesions elsewhere on the body or can occur as an isolated phenomenon
- Genital pyoderma gangrenosum is seen more frequently in the infantile age group. Perineal involvement can be seen in human immunodeficiency virus infection
- Behcet's disease needs to be ruled out in addition to the other causes of genital ulcers.
Pyoderma gangrenosum in infants and children
- Pustules are seen as initial lesions in contrast to macules and papules seen as initial lesions in adults
- Generalized lesions are more common
- Infrequent association with underlying diseases as compared to adults
- Pathergy is negative
- Good prognosis.
Pyoderma gangrenosum associated with extracutaneous neutrophilic disease
This refers to sterile neutrophilic infiltrates occurring in various internal organs. Pulmonary neutrophilic infiltrates are most common.
Pyostomatitis vegetans
- Oral pustular pyoderma gangrenosum characterized by a pustular, vegetative process of mucous membranes
- Associated with inflammatory bowel disease.
What is pathergy phenomenon?
This is characterized by the localization of pyoderma gangrenosum to the sites of skin damaged by trauma, surgery or venipuncture. It possibly represents a localized, misdirected host-mediated effector cell response to cutaneous tissue which is antigenically changed by trauma in a patient with altered immune reactivity.
What are the types of pathergy test?
There are two types of pathergy test: oral and cutaneous.
Oral pathergy test
It can be performed by pricking the mucosa of the lower lip using a 20-gauge blunt disposable needle and taking readings after 48 h. A pustule or an ulcer at the site of trauma is considered to be positive. It is less sensitive as compared to the ordinary skin pathergy test but easier to assess.
Skin pathergy test
It is usually performed on the flexor aspect of the forearm with a 20-gauge needle inserted vertically or diagonally at an angle of 45° at a depth of 3–5 mm to reach the dermis. About 1–2 mm papule with an erythematous halo or its transformation into 1–5 mm pustule is considered to be positive.
As an alternative, injection of intradermal saline/monosodium urate crystals/streptococcal antigens has been used. Other routes of pathergy testing in addition to the intradermal are subcutaneous and intravenous routes.
What are the conditions that exhibit a positive pathergy test?
The conditions that exhibit a positive pathergy test include:
- Bechet's disease
- Pyoderma gangrenosum (25% of cases)
- Interferon α-treated chronic myeloid leukemia
- Sweet's syndrome
- Eosinophilic pustular folliculitis
- Inflammatory bowel disease
- Healthy individuals
- Rarely in spondyloarthropathies.
What are the associations of pyoderma gangrenosum?
The diseases which can be associated with pyoderma gangrenosum are as follows:
Inflammatory
- Inflammatory bowel disease
- Arthritis: rheumatoid arthritis, ankylosing spondylitis
- Collagen vascular disease
- Miscellaneous: human immunodeficiency infection, hidradenitis suppurativa.
Neoplastic
- Internal malignancy
- Carcinoid tumor.
Hematologic
- Leukemia
- Myeloproliferative diseases and myelodysplasia
- Polycythemia vera
- Gammopathies.
Drug induced
- Pefilgastrim
- Gefitinib
- Isotretinoin.
What are the criteria for the diagnosis of pyoderma gangrenosum?
The diagnostic criteria for pyoderma gangrenosum as suggested by W.P. Daniel Su et al. are as follows:
Major criteria
- Rapid progression of a painful, necrotic, cutaneous ulcer with an irregular, violaceous and undermined border
- Other causes of cutaneous ulceration have been excluded.
Minor criteria
- History suggestive of pathergy or clinical finding of cribriform scarring
- Systemic diseases associated with pyoderma gangrenosum
- Histopathological findings (sterile dermal neutrophilia ± mixed inflammation ± lymphocytic vasculitis)
- Treatment response (rapid response to systemic steroid therapy).
What are the treatment options for pyoderma gangrenosum?
Treatment guidelines include:
- Exclude other diagnoses
- Treatment of underlying disease
- No single agent is useful, various topical and systemic agents used in the treatment of pyoderma gangrenosum are enumerated in [Box 1].

Crossword Puzzle on Nail Tic Disorders
Fill in the crossword puzzle in [Figure - 2] using the clues provided in [Box 2]
 |
| Figure 2: Crossword on nail disorders (Answers in Figure 3) |

Answers to the crossword puzzle are provided in [Figure - 3].
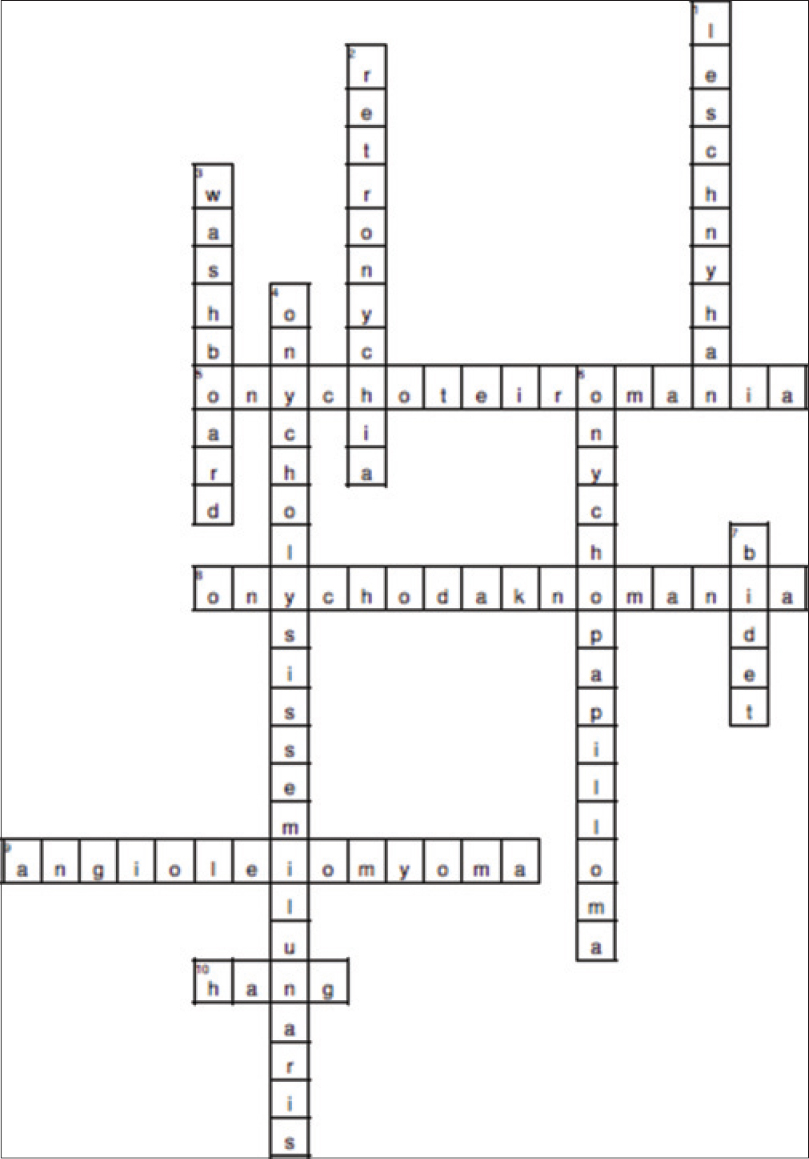 |
| Figure 3: Answers to the crossword puzzle |
Classification of Palmoplantar Keratodermas
How do you classify palmoplantar keratodermas?
Palmoplantar keratodermas can be classified on the basis of history or clinical pattern.
- Based on history: inherited and acquired.
- Clinical pattern:
- o Diffuse keratodermas: Whole of the palmar or plantar epidermis is abnormally and uniformly thickened
o Focal (or areate or nummular): Areas of palmoplantar skin under most pressure are disproportionately thickened. The lesions may be painful
o Striate: Overlaps clinically with focal keratoderma, but the lesions are mostly longitudinal, particularly on the fingers, where the lesions overlie flexor tendons
o Punctate (papular or disseminated): Consists of multiple, scattered, discrete round lesions.
- The keratodermas can additionally be classified into three subgroups:
- o Simple keratodermas with palmoplantar keratoderma as the sole manifestation
o Complex keratodermas associated with lesions of non-volar skin, hair, teeth, nails and/or sweat glands, including the ectodermal dysplasia
o Syndromic keratodermas associated with abnormalities of other organs, including deafness, cardiomyopathy and cancer.
What are the specific clinical features seen in keratodermas?
The keratodermas may show certain specific clinical features. These include:
- Transgradient keratoderma extends beyond palmoplantar skin, contiguously
- Confluent hyperkeratosis extends around the whole digit
- Cicatrizing keratodermas are those in which constricting bands appear around digits, usually distal to proximal interphalangeal joints.
Enumerate the various types of diffuse and focal keratodermas?
The various types of diffuse and focal keratodermas are outlined in [Box 3].

Which are the keratodermas with extracutaneous features?
The keratodermas with extracutaneous features include:
- Howel–Evans syndrome: Keratoderma with esophageal carcinoma (focal palmoplantar keratoderma with oral lesions)
- Naxos disease: Striate keratoderma, woolly hair and arrhythmogenic cardiomyopathy
- Carvajal syndrome: Striate keratoderma, woolly hair and dilated cardiomyopathy
- Vohwinkel syndrome: Cicatrizing keratoderma with hearing loss; papular lesions becoming confluent, starfish keratosis is the classical feature. Mild sensorineural hearing impairment present
- Papillon–Léfèvre syndrome: Keratoderma with severe periodontitis and other pyogenic infections
- Richner–Hanhart syndrome: Oculocutaneous tyrosinemia, corneal ulcers, painful keratoses and progressive mental impairment.
Which are the keratodermas associated with carcinoma?
The keratodermas associated with carcinomas include:
- Howel–Evans syndrome: It is a hereditary focal hyperkeratosis of palms and soles associated with esophageal carcinoma
- Papillon–Léfèvre syndrome (keratoderma with periodontitis): Malignant melanoma has been reported as a complication of this syndrome
- Huriez syndrome (keratoderma with scleroatrophy): It is associated with squamous and verrucous carcinomas
- Familial punctate keratoderma is associated with various internal organ malignancies
- Filiform palmoplantar keratoderma is associated with cancer of the breast, colon and kidney
- Acquired palmoplantar keratoderma can be paraneoplastic
- Tripe palms: It is a form of acquired palmar keratoderma with a thickened moss-like or velvety structure characterized by exaggerated dermatoglyphics of the palmar aspect of the hands. It is commonly associated with lung and stomach malignancies
- Bazex's acrokeratosis paraneoplastica: It is a form of acquired palmoplantar keratoderma seen in association with neoplasia of the upper aerodigestive tract
- Arsenical keratoderma is associated with internal malignancy commonly affecting the lung and bladder.
Which are the keratodermas associated with hearing impairment?
The keratodermas associated with hearing impairment include:
- Vohwinkel syndrome
- Bart–Pumphrey syndrome
- Mitochondrial palmoplantar keratoderma with hearing impairment.
Due to the defect in connexin function, hereditary keratodermas are associated with hearing loss.
Non-Pigmented Fixed Drug Eruption
What is non-pigmented fixed drug eruption?
The concept of non-pigmenting fixed drug eruption was popularized by Shelley and Shelley in 1987. Unlike fixed drug eruption in which residual pigmentation is a hallmark, this variant is rare and the drug-induced lesions resolve without leaving any residual pigmentation. It has been postulated that the likely site of the drug-induced hypersensitivity response is the dermis rather than the epidermis. Hence, there is absence of pigment incontinence and no pigmentary sequelae are seen after the initial lesions subside.
Which are the drugs causing fixed drug eruption?
Fixed drug eruption can be caused by various drugs including non-steroidal anti-inflammatory drugs, paracetamol, co-trimoxazole, tetracyclines, penicillins, metronidazole, rifampicin, angiotensin-converting enzyme inhibitors, pseudoephedrine, phenolphthalein, barbiturates, carbamazepine, calcium-channel blockers, omeprazole, contrast dyes, azoles, fluoroquinolones, complementary medicines and food items such as cashew nuts and asparagus.
Which are the drugs causing non-pigmented fixed drug eruption?
Non-pigmented fixed drug eruption has been reported in association with pseudoephedrine, tetrahydrozoline, diflunisal, thiopental, piroxicam, iothalamate, arsephenamine, paracetamol, intra-articular triamcinolone acetonide, eperisone hydrochloride, furazolidone and acetaminophen, sorafenib, tadalafil and esomeprazole and fluoroquinolones.
What is the cause of reactivation at the same site in fixed drug eruptions?
It is believed to be due to a delayed-type hypersensitivity reaction mediated by CD8+ T-cells. Epidermal CD8+ memory T-cells which are retained in the lesions get reactivated on rechallenge.
What are the histopathological findings in fixed drug eruption?
Histopathology shows interface dermatitis with vacuolar degeneration of basal keratinocytes, dermal edema and a perivascular lymphocytic infiltrate of the upper dermis along with eosinophils in early lesions whereas melanophages in the upper dermis are characteristic of resolving or healed lesions.
Enumerate various cutaneous adverse drug reactions due to fluoroquinolones
Fluoroquinolones have been implicated in various cutaneous adverse drug reactions such as exanthems, urticaria, fixed drug eruptions, angioedema, exfoliative dermatitis, photosensitivity, acute generalized exanthematous pustulosis, vasculitis, anaphylaxis and Stevens–Johnson syndrome or toxic epidermal necrolysis.
Pearly Penile Papules
What are pearly penile papules?
Pearly penile papules are common anatomical variations seen on the male genitalia. Clinically, these present as multiple, 1–2 mm, smooth, dome-shaped, skin-colored or pinkish papules in a single or double row along the corona of the glans penis. They may be rarely seen on the glans in rows or rings or ectopic lesions may be seen on the shaft of the penis.
What are the histological and dermatoscopic findings of pearly penile papules?
Histological findings of pearly penile papule are of an angiofibroma. Dermatoscopy demonstrates whitish pink tiny papules in rows with central dotted vessels.
How can the condition be treated?
Reassurance and counseling regarding the benign nature is sufficient. In anxious patients desiring treatment, cryotherapy or lasers can be effective.
Ultraviolet A-1 Therapy
What is ultraviolet A-1 phototherapy and enumerate the various dosing regimens?
Ultraviolet A-1 phototherapy uses long wavelength ultraviolet A radiation (340–400 nm), the erythemogenic ultraviolet A-2 and ultraviolet B wavelengths (290–340 nm) are filtered out.
Ultraviolet A-1 phototherapy is used at varying doses including high-dosage (>60 J/cm 2), medium-dosage (30–60 J/cm 2), low-dosage (10–20 J/cm 2) and very low-dosage (<10 J/cm 2) regimens. Therapy can be delivered either with low irradiance lamps (such as Philips TL-10) or from metal halide portable or whole body ultraviolet A-1 units.
What are the uses of ultraviolet A-1 phototherapy?
Ultraviolet A-1 phototherapy has been effective in a variety of inflammatory, sclerosing and neoplastic skin diseases that are characterized by dermal infiltrates rich in T-lymphocytes. The common cutaneous disorders in which ultraviolet A-1 has been tried include:
- Atopic dermatitis
- Scleroderma
- Mycosis fungoides
- Urticaria pigmentosa
- Lupus erythematosus
- Extragenital lichen sclerosus et atrophicus
- Granuloma annulare.
Other cutaneous conditions where it has been tried include keloids and hypertrophic scars, pompholyx, prurigo nodularis, chronic actinic dermatitis, discoid eczema, scleredema, perforating collagenosis and nephrogenic systemic fibrosis.
Fish Tank Granuloma
What is fish tank granuloma?
Fish tank granuloma is an atypical mycobacterial infection due to Mycobacterium marinum (formerly Mycobacterium balnei). The organism is an environmental, non-tuberculous mycobacterium that causes disease in fresh and salt water fish and rarely in humans. They are mainly aquarium related or the result of fish or shellfish injuries in swimming pools and other freshwater sources. Hence, infection with M. marinum has been named as “fish tank granuloma.” Recently, opportunistic M. marinum skin infections have also been reported in patients receiving anti-tumor necrosis factor agents.
Describe the clinical manifestations of fish tank granuloma
The disease begins as a violaceous papule or nodule and can also present as a psoriasiform or verrucous plaque, usually on the hands, feet, elbows or knees. Lesions develop at the site of trauma about 2 or 3 weeks after inoculation. These may be solitary but are often multiple and occasionally, sporotrichoid spread occurs. The lesions may ulcerate or frequently heal spontaneously within 1–2 years with residual scarring. Sometimes, penetration to the underlying structures may occur and complications include tenosynovitis, osteomyelitis, bursitis and septic arthritis.
Enumerate the diagnostic techniques and differential diagnosis for fish tank granuloma
History, high index of clinical suspicion, positive culture and use of polymerase chain reaction-reverse cross blot hybridization assay with species-specific gene probes are helpful in diagnosing fish tank granuloma.
Differential diagnosis includes leishmaniasis and sporotrichosis in endemic areas and other atypical mycobacterial infections due to Mycobacterium kansasii, Mycobacterium chelonae and Mycobacterium gordonae which may have a sporotrichoid pattern.
What are the treatment options for Mycobacterium marinum infections?
M. marinum is often resistant, both in vitro and in vivo, to several antibiotics and there are few documented therapeutic studies, hence there is no established therapy of choice.
In superficial skin infections, clarithromycin, minocycline, doxycycline and trimethoprim-sulfamethoxazole are used as monotherapy. Combined therapy with two or more drugs may be needed due to drug resistance. In severe infections, a combination of rifampicin and ethambutol has been recommended. Treatment should be given for at least 6 weeks and for up to 12 months, depending on the clinical evolution of the lesion. Debridement is generally not recommended and is only indicated in cases that are refractory to antibiotic therapy. Cryotherapy, laser and photodynamic therapy have been reported as effective choices but there are few studies evaluating the efficacy of these methods.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Fulltext Views
9,188
PDF downloads
3,194





