Translate this page into:
Accuracy of indirect immunofluorescence on sodium chloride-split skin in the differential diagnosis of bullous pemphigoid and epidermolysis bullosa acquisita
2 Shandong Provincial Hospital for Skin Diseases, Jinan; Shandong Provincial Medical Center for Dermatovenereology, Jinan, China
3 Shandong Provincial Institute of Dermatology and Venereology, Shandong Provincial Academy of Medical Science, Jinan; Shandong Provincial Hospital for Skin Diseases, Jinan; Shandong Provincial Key Laboratory for Dermatovenereology, Jinan; Shandong Provincial Medical Center for Dermatovenereology, Jinan, China
Correspondence Address:
Furen Zhang
Shandong Provincial Institute of Dermatology and Venereology, 27397 Jingshi Road, Jinan, Shandong Province, 250022
China
| How to cite this article: Yang B, Wang C, Chen S, Chen X, Zhou G, Tian H, Yu M, Zhang D, Shi Z, Zhang F. Accuracy of indirect immunofluorescence on sodium chloride-split skin in the differential diagnosis of bullous pemphigoid and epidermolysis bullosa acquisita. Indian J Dermatol Venereol Leprol 2011;77:677-682 |
Abstract
Background: Previous reports have shown that indirect immunofluorescence (IIF) performed on sodium chloride-split skin (SSS) is helpful to differentiate epidermolysis bullosa acquisita (EBA) from bullous pemphigoid (BP). Antibodies of BP may bind to the epidermal side of SSS, while antibodies of EBA bind to the dermal side. Aims: To determine the accuracy of IIF-SSS in the differential diagnosis of EBA and BP utilizing immunoblotting (IB) analysis. Methods: Sera from 78 patients, diagnosed with BP by clinical features, histopathology, and direct immunofluorescence (DIF), were assayed using IIF-SSS and IB. Results: Of the 43 serum samples with an epidermal reaction to IIF-SSS assay, 42 were recognized with BP antigens (180 kDa or 230 kDa). Of the 11 serum samples with a dermal reaction pattern, 7 were recognized with the 290 kDa antigen of EBA and 3 with sera bound BP antigens. Seven serum samples with epidermal and dermal combined staining, of which 5 of them reacted with BP antigens, 1 reacted with both BP and EBA antigens. One serum sample from each group showed a negative result by IB. Approximately 9.0% (7/78) of patients diagnosed with BP using regular methods were actually EBA. Conclusions: Epidermal reaction using the IIF-SSS assay highly correlated with the diagnosis of BP. However, dermal reactions correlated poorly with EBA, with some serum samples from BP patients binding to dermal-side antigens. In both epidermal and dermal stained sera using IIF-SSS, there was a possibility of BP and EBA. Differential diagnosis should be confirmed using IB, especially in cases of dermal and double staining patterns assayed using IIF-SSS.Introduction
Bullous pemphigoid (BP) is an acquired subepidermal autoimmune bullous dermatosis characterized by linear deposition of IgG and C3 at the cutaneous basement membrane zone (BMZ). [1] Collagen XVII (180 kDa, also termed BP180, BPAG2) and BP 230 (230 kDa, also termed BPAG1), located in the hemidesmosome, have been identified as target antigens in BP patients. [2] A passive transfer study utilizing mouse model has shown that antibodies to BP180 play a key role in the formation of subepidermal blisters in BP patients. [3] Epidermolysis bullosa acquisita (EBA) is a rare, acquired, chronic autoimmune disease characterized by subepidermal blisters and circulating IgGs bound to the BMZ. [4] The circulating IgG autoantibodies for EBA react with a 290 kDa dermal protein, type VII collagen, detected using immunoblotting (IB) analysis from dermal extracts. [5]
Previous studies have reported that at least 50% of patients with EBA show a BP-like clinical presentation, particularly with the inflammatory variant of EBA and 10% of patients with a clinical presentation of BP may have a diagnosis of EBA. [6] It is important to distinguish between EBA and BP. EBA can be associated with chronic systemic diseases, such as inflammatory bowel diseases; however, EBA is often unresponsive to high doses of prednisone.
It is difficult to differentiate EBA from BP using only histological and direct immunofluorescence (DIF) features. Although an enzyme-linked immunosorbent assay (ELISA) was developed to diagnose BP and EBA, [7],[8] indirect immunofluorescence (IIF) is still widely used to diagnose autoimmune blister skin diseases. Some studies have reported that IIF on sodium chloride-split skin (SSS) is a simple and accurate laboratory method to differentiate EBA from BP. [9] It is clear that IgG autoantibodies from patients with BP bind the epidermal or both sides of SSS, whereas those from patients with EBA bind only the dermal side of SSS. [9] Antibodies of a novel subepidermal bullous disease, anti-laminin gamma-1 pemphigoid, previously called anti-p200 pemphigoid, also bind the dermal side of SSS. [10],[11] IB is an effective method used to confirm the diagnosis of autoimmune blistering skin diseases according to the molecular weight of antigens; however, this technique is complicated and time consuming. [12]
In the present study, we assessed the accuracy of IIF-SSS with the application of an IB assay in the differential diagnosis of BP and EBA.
Methods
Patients and serum samples
Seventy-eight patients (47 males, 31 females) with BP, ages ranging from 5 to 83 years (mean age, 63 years), were enrolled in the study. The diagnosis of BP was based on the three following clinical and laboratory criteria: (i) clinical presence of cutaneous tense blisters or erosions; (ii) histological presence of a subepidermal blister; (iii) DIF findings of linear deposition of IgG and/or C3 at the BMZ. Sera were collected prior to systemic immunosuppressive therapy.
Serum samples from 20 normal healthy individuals and 10 patients with psoriasis were used as negative controls. A mouse monoclonal anti-collagen VII antibody, LH7.2 (Sigma-Aldrich, St. Louis, USA) was used as an internal standard.
This study was approved by the Medical Ethics Committee of Shandong Provincial Hospital for Skin Diseases.
Salt-split normal human skin
Adult fresh normal skin, obtained from patients undergoing surgical correction of breast cancer, was cut into fragments 1.0 cm×1.0 cm and washed with PBS. The fragments were then incubated in 1.0 mol/L sodium chloride solution for 48-72 h at 4 o C. The SSS was then imbedded in optimal cutting temperature (OCT) compound for frozen-section IIF diagnosis.
Indirect immunofluorescence on sodium chloride-split skin
IIF was performed on 4-μm cryostat sections of salt-split normal human skin, as described previously. [13] The serum samples were serially diluted from 1:10 and added onto the slides with SSS. The sections were incubated for 30 min at room temperature in a moist chamber. After washing three times with PBS, the sections were incubated with FITC-conjugated goat anti-human IgG (Dako, Denmark) for 30 min at room temperature. The sections were viewed using a fluorescence microscope.
Immunoblotting
The epidermis and dermis of adult fresh normal skin were separated using a 1.0 mol/L sodium chloride solution. Epidermal extracts were prepared as previously described by Mueller et al, [14] with some modifications. After dermal-epidermal separation, the epidermis was placed in extraction buffer (containing: 65 mmol/L Tris-HCl, 2 mmol/L PMSF, 10 mmol/L EDTA, 2% sodium dodecyl sulfate (SDS), pH 6.8, containing 0.01 mg/ml of each of protease inhibitors antipain, pepstatin, leupeptin and chymostatin). The homogenate was freeze-thawed repeatedly three times in liquid nitrogen, and then centrifuged at 13,000g for 30 min at 4 o C. The supernatant was collected and stored at -80 o C until the time of study.
Dermal extracts were prepared by methods described by Woodly et al., [15] with some modifications. After dermal-epidermal separation, the dermal portion was immersed in 31.2 mmol/L of Tris-HCl buffer (pH 6.8) containing 8 mol/L urea, 5% 2-ME, 2% SDS, 10 mmol/L EDTA, and 10 mmol/L PMSF, and then vortexed for 1 h at room temperature, and centrifuged at 13,000g for 30 min at 4 o C. The supernatant was collected and stored at -80 o C until further use. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and IB assays were performed. Epidermal and dermal proteins were separated using a 6% SDS-PAGE. The proteins were transferred electrophoretically onto a nitrocellulose member. The member was then cut into strips and sequentially incubated with (i) 5% dried milk in 10 mmol/L Tris-HCL 140 mmol/L NaCl, pH 7.4 (TBS/milk) for 3 h at room temperature; (ii) 1:10 dilution of each serum sample in TBS/milk for 3 h at 37 o C; (iii) 1:500 dilution peroxidase-conjugate rabbit anti-human IgG (Dako) for 1 h at 37 o C. Intervening washes were performed with TBS containing 0.1% Tween-20. The strips were developed with 3,3-diaminobenzidine (DAB)-4 HCl (0.5 mg/ml) in 10 mmol/L Tris-HCL (pH 7.6) containing 0.03% H 2 O 2 .
Statistical analyses
A 2×2 contingency table Chi-squared test was used to evaluate whether there was any significant difference between IIF-SSS and IB samples. Statistical significance was defined as P<0.05.
Results
Indirect immunofluorescence on sodium chloride-split skin and Immunoblotting
Three types of staining were observed by IIF-SSS: epidermal side, dermal side, and both of the two sides [Figure - 1]. If the antibodies of the sera bind the 180 kDa and/or 230 kDa proteins of epidermal side by IB, the diagnosis was BP [Figure - 2](a). If the antibodies bind the 290 kDa protein of dermal side, the diagnosis was EBA [Figure - 2](b). If the antibodies bind the 200 kDa protein of dermal side, the diagnosis should be anti-laminin gamma-1 pemphigoid.
 |
| Figure 1: Indirect immunofluorescence on salt-split skin: (a) epidermal staining; (b) dermal staining, (c) combined epidermal and dermal staining |
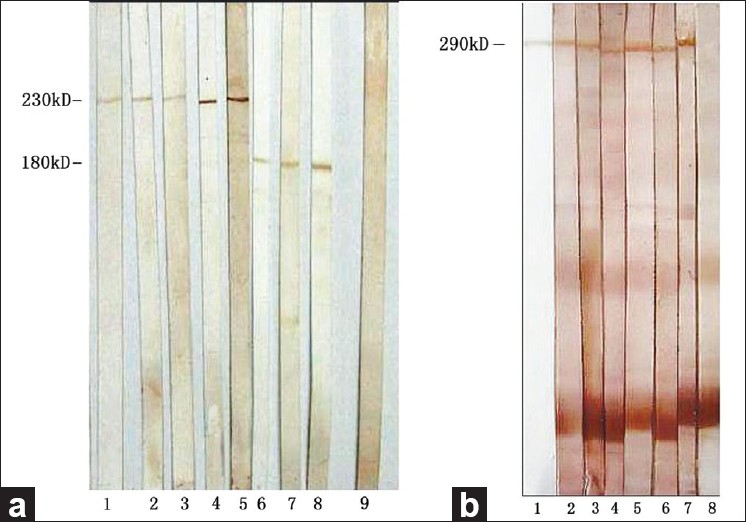 |
| Figure 2: Results of immunoblotting. (a) Sera from BP patients reacted with epidermal extracts. Lanes 1-5: 230 kDa positive; lanes 6-8: 180 kDa positive; lane 9: normal human control serum: negative. (b) Sera from EBA patients reacted with dermal extracts. Lane 1: LH7.2; Lanes 2-7 290 kDa positive; Lane 8: normal human control serum negative |
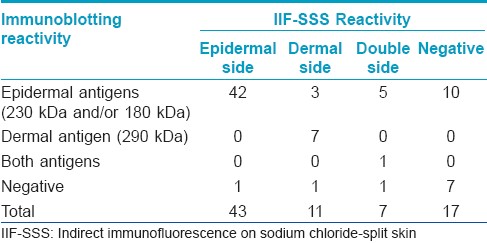
Among the 78 sera from BP patients, the positivity of IIF-SSS and IB was 78.2% (61/78) and 87.2% (68/78), respectively. There was no significant difference between these groups using a Chi-squared test [Table - 1]. There were no positive results in the control group. Seventeen serum samples were negative using the IIF-SSS assay, 10 were positive and 7 were negative using IB. The results and details are shown in [Table - 2]. By the combination of IIF-SSS and IB, 62 patients with BP were confirmed as well as 7 patients with EBA and 1 patient with both BP and EBA. Approximately 9.0% (7/78) of patients diagnosed with BP using regular methods were actually EBA. None was diagnosed as anti-laminin gamma-1 pemphigoid in this study.

Discussion
The present results of epidermal staining showed that the sensitivity between IIF-SSS and IB assays was similar. Of the 43 sera from BP patients with epidermal staining patterns of IIF-SSS, 42 samples were detected using BP antigens (180 kDa and/or 230 kDa) by IB, with one exception. These results suggest a good correlation of epidermal staining patterns in IIF-SSS and the diagnosis of BP and consistent with IIF-SSS sensitivity in detecting circulating antibodies of BP. [16] Therefore, the value of IIF-SSS and IB were demonstrated in the differential diagnosis of BP and EBA.
The autoantibody against BP180 is the most suspected pathogenic factor for BP. Approximately 90% of BP patients have antibodies to the synthetic noncollagenous domain 16a of BP180. [17] Autoantibodies to the transmembrane antigen BP180, but not to the intracellularly located BP230, are thought to be pathogenically relevant. [3],[18] The pathologic role of anti-BP230 antibodies remains unclear. Kiss et al., [19] demonstrated that antibodies against BP230 can elicit the clinical and immunopathological features of BP in neonatal mice. Using three bacterial recombinant proteins covering the entire human BP230 molecule, Hamada et al. [20] found that anti-BP230 autoantibodies were present in 80% of BP sera, which were selected using an IIF titer of 1:40 or more. Recently, Tampoia et al. [21] evaluated the sensitivity and specificity of two new ELISA kits for the detection of BP180 and BP230 autoantibodies. They found that the specificity for both tests was over 98% and the sensitivity was 90% and 60% for anti-BP180 and anti-BP230, respectively. These results indicate that both IB and ELISA for BP180 and BP230 are useful tools for the diagnosis of BP.
In the present study of dermal staining, 7 of 11 serum samples were reactive to type VII collagen (290 kDa), while 3 serum samples were detected as having an autoantibody to BP180, with 1 being negative. These results agree with other reports that the sera with dermal reactivity were not specific to EBA. [22] These three BP sera positive for BP180 could be explained by the fact that the extracellular domain (ECD) of BP180 contains a long 15-repeated collagenous domain which spans the lamina lucida and extends to the lamina densa. Therefore, some antigen epitopes of BP180 located in the area of lamina densa could be separated to the dermal side after salt split process. [23] Interestingly, 2 of these samples also reacted with BP230, however the reasons remain unclear. It is speculated that the titer of antibodies to BP230 was too low to stain the epidermal side using IIF-SSS. Anti-p200 pemphigoid may closely resemble BP in clinical and DIF findings. But antibodies of patients with anti-p200 pemphigoid bind the dermal side of SSS. [10] Recently, laminin g l, localized to the lower lamina lucida, was recognized by anti-p200 pemphigoid autoantibody and the disease was renamed as anti-laminin gamma-1 pemphigoid. [11] However, no patients were diagnosed as anti-laminin gamma-1 pemphigoid in this study.
In 7 serum samples with double staining, 5 reacted with BP antigens. Among these 5 sera, all reacted with BP180, meanwhile, 3 sera were also positive with BP230. These results suggest that there is a relationship between the presence of anti-BP180 antibody and combined patterns of IIF-SSS. The combined pattern may be due to the presence of antibodies in the patients′ sera against the multiple antigenic epitopes along the BP180. [9] Remarkably, we found a rare serum which reacted to both BP180 and 290 kDa dermal antigens. To our knowledge, two similar cases have been reported. [24],[25] These special cases could be attributed to the phenomena of intermolecular epitope spreading. The remaining one serum sample was negative using IB.
Among IIF-SSS negative sera, 10 of 17 were found to be positive using IB. Seven sera recognized BP180, 2 recognized BP230, and one serum sample recognized both BP antigens. These results were consistent with a previous study [26] and suggest that sera containing an anti-BP180 antibody are more likely to be negative using IIF-SSS than those containing anti-BP230 antibody. Two possible reasons could account for this special condition. The titer of the anti-BP180 antibody is too low to be detected by IIF-SSS. Also, the main epitopes of BP180 are located in the excellular domain, such as BP180 NC16A, and this conformation could easily be destroyed during the salt split process and lose its immunoreactivity.
Our present results suggest that IIF-SSS is a useful tool for the primary diagnosis of subepidermal blistering skin diseases; however, some patients need additional tests, including IB or ELISA, to confirm the diagnosis. Recently, Müller et al. [27] reported that type VII collagen-NC1 ELISA is a powerful tool for the diagnosis of EBA. When an ELISA combined NC1 and NC2 domains of type VII collagen was used to detect anti-type VII collagen antibodies of EBA, its sensitivity and specificity could be improved to 93.8% and 98.1%, respectively. [28] In addition, an ELISA for the detection of autoantibodies in anti-p200 pemphigoid is also available recently. [29] The limitation of this study is that ELISA was not used for the differential diagnosis of EBA and BP. Although ELISA is a simple, cheaper, fast, and accurate method for evaluation of the disease activity and assessment of the treatment effect of bullous skin diseases, it can only be performed on serum samples. Alternatively, IB is a useful tool to find new subepidermal bullous dermatosis, such as anti-p200 pemphigoid. Therefore, IB has the advantage over ELISA that different subepidermal bullous dermatosis can be diagnosed in one system.
In conclusion, epidermal reactions of the IIF-SSS assay are highly correlated with BP, but the dermal reaction correlates poorly with EBA. A few sera of BP samples may also bind to the dermal side. In cases of double stained sera using IIF-SSS, there is a possibility of a coincidence of BP and EBA and should be confirmed using IB or ELISA.
| 1. |
Zillikens D. Acquired skin disease of hemidesmosomes. J Dermatol Sci 1999;20:134-54.
[Google Scholar]
|
| 2. |
Stanley JR. Autoantibodies against adhesion molecules and structures in blistering skin diseases. J Exp Med 1995;181:1-4.
[Google Scholar]
|
| 3. |
Liu Z, Diaz LA, Troy JL, Taylor AF, Emery DJ, Fairley JA, et al. A passive transfer model of the organ-specific autoimmune disease, bullous pemphigoid, using antibodies generated against the hemidesmosomal antigen, BP180. J Clin Invest 1993;92:2480-8.
[Google Scholar]
|
| 4. |
Hallel-Halevy D, Nadelman C, Chen M, Woodley DT. Epidermolysis bullosa acquisita: Update and review. Clin Dermatol 2001;19:712-8.
[Google Scholar]
|
| 5. |
Woodley DT, Briggaman RA, Gammon WR, Falk RJ, Reese MJ, Tomsick RS, et al. Epidermolysis bullosa acquisita antigen, a major cutaneous basement membrane component, is synthesized by human dermal fibroblasts and other cutaneous tissues. J Invest Dermatol 1986;87:227-31.
[Google Scholar]
|
| 6. |
Gammon WR, Briggaman RA, Woodley DT, Heald PW, Wheeler CE Jr. Epidermolysis bullosa acquisita--a pemphigoid-like disease. J Am Acad Dermatol 1984;11:820-32.
[Google Scholar]
|
| 7. |
Barnadas MA, Rubiales MV, González MJ, Puig L, García P, Baselga E, et al. Enzyme-linked immunosorbent assay (ELISA) and indirect immunofluorescence testing in a bullous pemphigoid and pemphigoid gestationis. Int J Dermatol 2008;47:1245-9.
[Google Scholar]
|
| 8. |
Chen M, Chan LS, Cai X, O'Toole EA, Sample JC, Woodley DT. Development of an ELISA for rapid detection of anti-type VII collagen autoantibodies in epidermolysis bullosa acquisita. J Invest Dermatol 1997;108:68-72.
[Google Scholar]
|
| 9. |
Lazarova Z, Yancey KB. Reactivity of autoantibodies from patients with defined subepidermal bullous diseases against 1 mol/L salt-split skin. Specificity, sensitivity, and practical considerations. J Am Acad Dermatol 1996;35:398-403.
[Google Scholar]
|
| 10. |
Zillikens D, Kawahara Y, Ishiko A, Shimizu H, Mayer J, Rank CV, et al. A novel subepidermal blistering disease with autoantibodies to a 200-kDa antigen of the basement membrane zone. J Invest Dermatol 1996;106:1333-8.
[Google Scholar]
|
| 11. |
Dainichi T, Kurono S, Ohyama B, Ishii N, Sanzen N, Hayashi M, et al. Anti-laminin gamma-1 pemphigoid. Proc Natl Acad Sci U S A 2009;106:2800-5.
[Google Scholar]
|
| 12. |
Pas HH. Immunoblot assay in differential diagnosis of autoimmune blistering skin diseases. Clin Dermatol 2001;19:622-30.
[Google Scholar]
|
| 13. |
Gammon WR, Briggaman RA, Inman AO 3rd, Queen LL, Wheeler CE. Differentiating anti-lamina lucida and anti-sublamina densa anti-BMZ antibodies by indirect immunofluorescence on 1.0 M sodium chloride-separated skin. J Invest Dermatol 1984;82:139-44.
[Google Scholar]
|
| 14. |
Mueller S, Klaus-Kovtun V, Stanley JR. A 230-kD basic protein is the major bullous pemphigoid antigen. J Invest Dermatol 1989;92:33-8.
[Google Scholar]
|
| 15. |
Woodley DT, Briggaman RA, O'Keefe EJ, Inman AO, Queen LL, Gammon WR. Identification of the skin basement-membrane autoantigen in epidermolysis bullosa acquisita. N Engl J Med 1984;310:1007-13.
[Google Scholar]
|
| 16. |
Satyapal S, Amladi S, Jerajani HR. Evaluation of salt split technique of immunofluorescence in bullous pemphigoid. Indian J Dermatol Venereol Leprol 2002;68:330-3.
[Google Scholar]
|
| 17. |
Matsumura K, Amagai M, Nishikawa T, Hashimoto T. Majority of bullous pemphigoid and herpes gestationis sera react with NC16a domain of the 180 kD bullous pemphigoid antigen. Arch Dermatol Res 1996;288:507-9.
[Google Scholar]
|
| 18. |
Liu Z, Diaz LA. Bullous pemphigoid: End of the century overview. J Dermatol 2001;28:647-50.
[Google Scholar]
|
| 19. |
Kiss M, Husz S, Jánossy T, Marczinovits I, Molnár J, Korom I, et al. Experimental bullous pemphigoid generated in mice with an antigenic epitope of the human hemidesmosomal protein BP230. J Autoimmun 2005;24:1-10.
[Google Scholar]
|
| 20. |
Hamada T, Nagata Y, Tomita M, Salmhofer W, Hashimoto T. Bullous pemphigoid sera react specifically with various domains of BP230, most frequently with C-terminal domain, by immunoblot analyses using bacterial recombinant proteins covering the entire molecule. Exp Dermatol 2001;10:256-63.
[Google Scholar]
|
| 21. |
Tampoia M, Lattanzi V, Zucano A, Villalta D, Filotico R, Fontana A, et al. Evaluation of a new ELISA assay for detection of BP230 autoantibodies in bullous pemphigoid. Ann N Y Acad Sci 2009;1173:15-20.
[Google Scholar]
|
| 22. |
Ghohestani RF, Nicolas JF, Rousselle P, Claudy AL. Diagnostic value of indirect immunofluorescence on sodium chloride-split skin in differential diagnosis of subepidermal autoimmune bullous dermatoses. Arch Dermatol 1997;133:1102-7.
[Google Scholar]
|
| 23. |
Di Zenzo G, Grosso F, Terracina M, Mariotti F, De Pità O, Owaribe K, et al. Characterization of the anti-BP180 autoantibody reactivity profile and epitope mapping in bullous pemphigoid patients. J Invest Dermatol 2004;122:103-10.
[Google Scholar]
|
| 24. |
Kawachi Y, Ikegami M, Hashimoto T, Matsumura K, Tanaka T, Otsuka F. Autoantibodies to bullous pemphigoid and epidermolysis bullosa acquisita antigens in an infant. Br J Dermatol 1996;135:443-7.
[Google Scholar]
|
| 25. |
Fairley JA, Woodley DT, Chen M, Giudice GJ, Lin MS. A patient with both bullous pemphigoid and epidermolysis bullosa acquisita: An example of intermolecular epitope spreading. J Am Acad Dermatol 2004;51:118-22.
[Google Scholar]
|
| 26. |
Ghohestani R, Kanitakis J, Nicolas JF, Cozzani E, Claudy A. Comparative sensitivity of indirect immunofluorescence to immunoblot assay for the detection of circulating antibodies to bullous pemphigoid antigens 1 and 2. Br J Dermatol 1996;135:74-9.
[Google Scholar]
|
| 27. |
Müller R, Dahler C, Möbs C, Wenzel E, Eming R, Messer G, et al. T and B cells target identical regions of the non-collagenous domain 1 of type VII collagen in epidermolysis bullosa acquisita. Clin Immunol 2010;135:99-107.
[Google Scholar]
|
| 28. |
Saleh MA, Ishii K, Kim YJ, Murakami A, Ishii N, Hashimoto T, et al. Development of NC1 and NC2 domains of Type VII collagen ELISA for the diagnosis and analysis of the time course of epidermolysis bullosa acquisita patients. J Dermatol Sci 2011;62:169-75.
[Google Scholar]
|
| 29. |
Groth S, Recke A, Vafia K, Ludwig RJ, Hashimoto T, Zillikens D, et al. Development of a simple enzyme-linked immunosorbent assay for the detection of autoantibodies in anti-p200 pemphigoid. Br J Dermatol 2011;164:76-82.
[Google Scholar]
|
Fulltext Views
3,922
PDF downloads
1,640





