Translate this page into:
Therapeutic agents for Hailey-Hailey disease: A narrative review
Corresponding author: Dr. Kabir Sardana, Department of Dermatology, Venereology and Leprosy, Atal Bihari Vajpayee Institute of Medical Sciences & Dr Ram Manohar Lohia Hospital, New Delhi, India. email id: kabirijdvl@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Sardana K, Bansal A, Muddebihal A, Khurana A. Therapeutic agents for Hailey-Hailey disease: A narrative review. Indian J Dermatol Venereol Leprol. doi: 10.25259/IJDVL_906_2024
Abstract
Hailey–Hailey disease (HHD) is an autosomal dominant genetic disorder of keratinocyte adhesion. It occurs due to mutations in ATP2C1, a gene on chromosome 3q21-24 which encodes human secretory pathway Ca2+/Mn2+ ATPase isoform 1, a calcium pump on the Golgi apparatus membrane. Recently, there has been a focus on certain pro-inflammatory cytokines such as IL-6 and IL-8 which play a role in HHD. Various triggers include excessive heat, menstruation, pregnancy, sweating, friction, exposure to sunlight and superficial infections. The therapy of the disorder is better understood by the varied pathogenetic steps and we aimed to comprehensively search for the various medical therapies used in HHD and align them with the existing knowledge on the pathogenesis and delineate them according to their major mode of action.
Keywords
Hailey–Hailey disease
therapy
cytokines
pathogenesis
JAK inhibitors
Introduction
While HHD has been managed by varied topical, systemic and procedural methods, the drugs administered either in sequence or isolation largely disregard the pathogenesis. The drugs seem to be directed at either the trigger factors or inflammatory mediators. In the absence of good-quality evidence, the clinician is at a loss to arrive at the ideal drug regimen for HHD.
We comprehensively searched the literature for the varied medical therapies used in HHD, align them with the existing knowledge on the pathogenetic aspects of the disease and encapsulate the existing drugs which can be used by the clinicians for the treatment of HHD.
Methods
This narrative review was conducted by searching database using PubMed, Embase and the Cochrane Central Register of Controlled Trials (CENTRAL), using the keywords, ‘Hailey–Hailey Disease AND treatment, pathogenesis, trigger factors’. Grey literature was avoided due to the lack of veracity of the published data.
Using the keywords ‘Hailey–Hailey Disease and ‘therapy’, sorted by Most Recent,’ from July 1984 to May 2024, 364 articles were found. Search was focussed on the active therapy arm as a monotherapy and included papers which were studies, Double blinded (DB) randomised control trials (RCT), DB placebo-controlled studies, case series and also case reports if a novel drug had been administered. Papers that looked at the molecular aspects of HHD and the rationale of the treatment were particularly looked into. This data was then used to divide various therapies into broad mechanistic categories and the drugs were categorised depending on the level of evidence as first-line, second-line and third-line . First-line drugs were chosen from studies, DB RCT and placebo-controlled studies. Second-line drugs were added from case series and third-line drugs were from case reports.
Overview & Pathogenesis
Hailey–Hailey disease (HHD) is a genetic disorder of keratinocyte adhesion with autosomal dominant inheritance and incomplete penetrance. The diagnosis may therefore be delayed in mild cases.
The defect is due to mutations in ATP2C1, a gene at chromosome 3q21-24 which encodes for the human secretory pathway Ca2+/Mn2+ ATPase isoform 1 (SPCA1), a calcium pump of the Golgi apparatus membrane. SPCA1 and sarcoendoplasmic reticulum Ca2+ ATPase2 (SERCA2), encoded by ATP2C1 and ATP2A2 genes, respectively, are two essential calcium pumps necessary for Ca2+ homeostasis in keratinocytes. The specific defect in HHD is linked to hSPCA1, the function of which is to transport Ca2+ and Mn2+ into the Golgi lumen.1 ATP2C1 is highly expressed in the basal layer of the epidermis. In normal epidermis, there is a calcium gradient with the highest levels in the basal layer and granular layer, both intra- and extracellularly. This gradient appears to be a key element for the differentiation process in the epidermis and helps in counter-talk between keratinocytes and the extracellular space which has a calcium-sensing receptor (CaR). In the case of external stressors, this gradient is disturbed but reverses within 24 hours and the normally functioning hSPCA1 compensates with increased Ca2+ levels and prevents acantholysis. In HHD, there is an impaired Ca2+ gradient and sequestration which causes a depletion of Ca2+ within the Golgi lumen and this leads to a dysfunction in the processing and translocation of junctional proteins that function in cell–cell adhesion leading to suprabasal acantholysis [Figure 1].
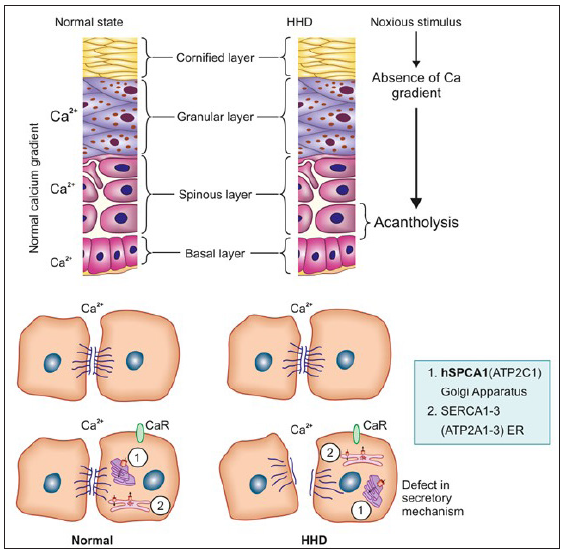
- In the normal state, there is a gradient of calcium which increases from the basal to the granular layer. This helps to sustain the cell-to-cell adhesion in the lower layers of the epidermis by a direct, post-translational effect of extracellular Ca2+ elevation that stabilises desmosomal integrity. The elevated level of extracellular calcium via calcium receptors likely affects the ER/Golgi Ca2+ homeostasis. In the case of any noxious stimuli, the gradient is disturbed but is restored in normal skin unlike in HHD skin leading to suprabasal acantholysis as the increase of extracellular Ca2+ in the suprabasal layer (SBL) of the epidermis does not occur. Other factors that are implicated include decrease in CaR activation and alteration of the ER/Golgi Ca2+ homeostasis due to lower amounts of hSPCA1 and consequently decreased levels of Ca2+ and Mn2+ in these compartments. (HHD, Hailey–Hailey disease; SBL, Suprabasal layer; CaR, Calcium-sensing receptor.)
Recent studies have reported that apart from the low levels of hSPCA1 in HHD patients, there is an increase of miR-203 level and a decrease of p63 and HKII levels.2 A novel pathogenic mechanism involving SPCA1, p63 and IRF6 may play a role in the skin lesions occurring in HHD.3
Various triggers have been implicated including excessive heat, menstruation, pregnancy, sweating, friction, exposure to sunlight and superficial infections with bacteria, viruses, candida, and scabies.4–6 Contact allergy to topical agents (e.g. crotamiton)7 or application of occlusive dressings8 can also flare the condition. The series of events resulting in acantholysis is depicted in Figure 2.
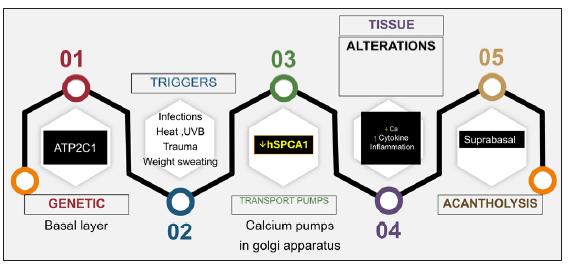
- A linear depiction of the salient sequence of events that determines and dictates the acantholysis in HHD. (UVB: Ultraviolet B therapy)
There has been a renewed focus on certain pro-inflammatory cytokines such as IL-6 and IL-8 which play a role in HHD and Ultraviolet B (UVB) radiation has been implicated in reducing SPCA1 to critical levels.6 The addition of IL-6 antibody prevents the UVB-induced suppression of ATP2A2 and ATP2C1 mRNA; in contrast, the addition of an anti-IL-8 antibody accelerates the suppression. The reduction of ATP2C1 mRNA expression by UVB irradiation in keratinocytes is reversed by retinoids, corticosteroids, ciclosporin, tacrolimus, and vitamin D3.9 The role of newer drugs like dupilumab and JAK inhibitors (JAKibs) revolve around their inhibitory effect on the Th2 cytokines.
Clinical Features
The onset of the disease is in young adult life and involves the sites of maceration and friction such as the axillae, groins, perianal region, and lateral aspect of the neck. Classic morphology is of ‘a dirt road drying out after a rainstorm.’
The initial lesion is a broad flaccid blister which ruptures to form plaques with crusts and fissures. The lesions are usually symmetric, round, oval, or circinate and can ooze, become foul-smelling, and develop papillomatous morphology. Unlike Darier’s disease, pruritus is not common. In less occluded areas, like the trunk and neck, the lesions are vesico-pustular with flaccid bullae or may be crusted [Figures 3a–3d]. Occasionally, palmar pits as well as longitudinal nail stripes can be seen.
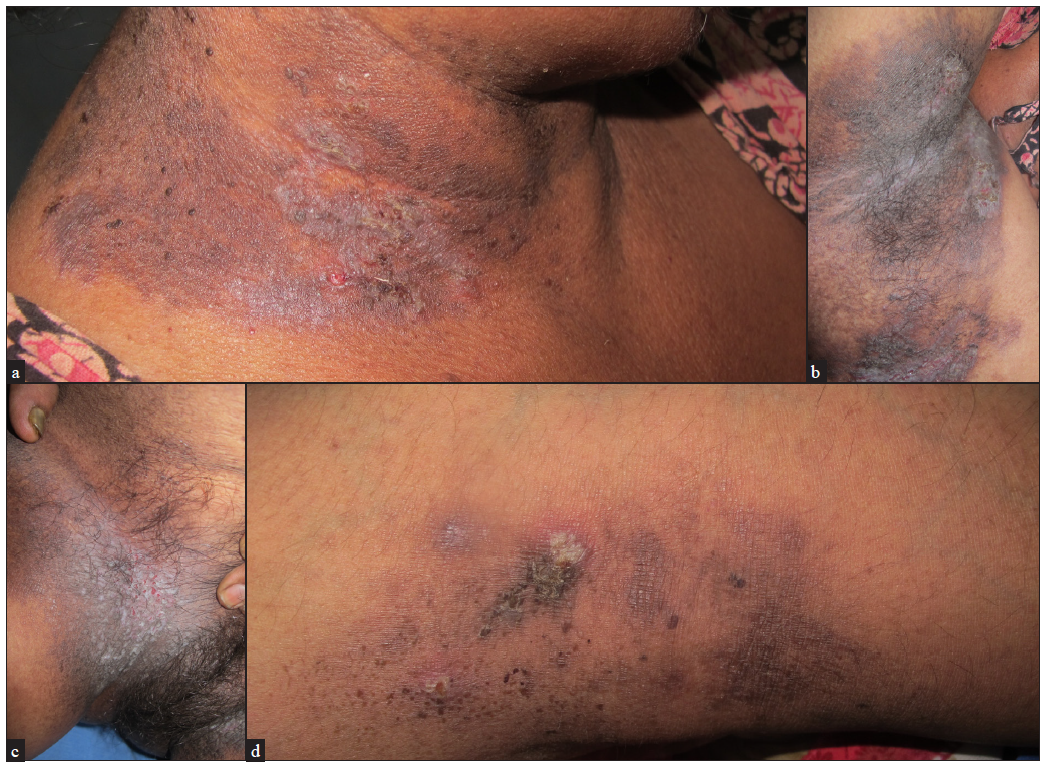
- Presence of flaccid vesicles, erosions and macerated crusted plaques (a) over the neck, (b) axilla, (c) groin and (d) popliteal fossa.
Differentials mainly include other conditions which can lead to erosive and oozing lesions in the intertriginous areas, such as Darier disease, pemphigus vegetans, inverse psoriasis, and intertriginous candidiasis.
The histology is characterised by prominent acantholysis, reminiscent of a ‘dilapidated brick wall’, affecting whole rete pegs and continues over the papilla tips. Dyskeratoses is only occasionally seen and the dermis has relatively dense lymphocyte-dominated inflammatory infiltrate.10
Treatment
The disorder is an inherited acantholytic disorder and hence has a chronic course with recurrent crops of lesions appearing intermittently. One of the basic pillars of therapy is to avoid triggers.
General Measures
Patients should avoid heat or friction, lose weight and wear loose-fitting cotton clothes, and avoid tight-fitting jeans. Adhesives and occlusive dressings should be avoided. There is a high chance of secondary infections. Hence, occasional topical and/or systemic antimicrobial and antifungal agents are indicated and long-term antimicrobial cleansers can be used as a preventive measure.11 Another useful option is bleach baths that can be used twice a week to reduce superficial infection.
Topical Agents
Topical agents are best described as adjunctive agents and include topical steroids, tacrolimus 0.03–0.1% or pimecrolimus 1% cream, and topical vitamin D analogues like calcitriol and talcalcitol.11 The various topical antimicrobial agents used include aminoglycosides (gentamicin 0.1% gel), fusidic acid, Castellani paint, and imidazoles.12 Tacrolimus has been used on a long-term basis to prevent flares.13 Here, it must be noted that the use of tacrolimus can itself lead to infections, including herpes with a report of SCC following prolonged use in vulvar HHD.14 A recent report used topical 15% aluminium chloride twice a week in an aqueous solution which is based on the perceived role of hyperhidrosis in aggravating HHD.15 Gentamicin in vitro can induce translational read-through of nonsense mutations in ATP2C1,16 though this might not be consistently seen in all genotypes of HHD. Steroids are advised only during acute flares and for a short course, as excessive use can lead to side effects including steroid atrophy, secondary infections, and contact dermatitis.
There are single case reports of topical fluorouracil 5% cream (thrice weekly for 3 months, then once weekly for 3 months) in groin involvement.17 This is based on the previous successful use of topical fluorouracil 5% cream in Darier’s disease with the hypothesis that 5-FU restores normal intracytoplasmic calcium concentrations.18 Topical cadexomer iodine, a sterile formulation of 0.9% elemental iodine bound to hydrophilic matrix cadexomer starch molecules with anti-microbial, anti-inflammatory, absorbent, and wound-healing properties has also been successfully tried in a single case.19
There are isolated case reports of the beneficial effect of topical timolol 0.5% and topical cinacalcet 3% ointment, the former being a beta adrenergic blocker restoring calcium homeostasis, the latter being a CaR agonist.20,21 The purported action would be to increase the intracellular calcium levels.
Systemic Agents
The systemic agents include oral corticosteroids, retinoids (acitretin 10–25 mg daily, etretinate 25–60 mg daily, alitretinoin 20–30 mg daily or Isotretinoin)22–24 and immunomodulatory drugs such as cyclosporine (2.8 to 3.4 mg/kg per day),25 methotrexate (7.5 mg weekly),26,27 azathioprine28 and dapsone (50–200 mg per day).29 All retinoids except isotretinoin have demonstrated resolution of patients’ symptoms, although failure with retinoids does occur.24
Broad-spectrum antibiotics such as oral flucloxacillin, erythromycin, or tetracyclines may be helpful, but the ideal antimicrobial agent to be used should be guided by the skin swabs for bacterial, fungal, and yeast culture and viral polymerase chain reaction (PCR).11,9 Painful, recalcitrant disease is suggestive of superinfection by herpes simplex and systemic antiviral treatment should be considered. Long-term antibiotics have also been tried including oral erythromycin (500 mg twice daily, 600 mg thrice daily {case reports}) or doxycycline (100 mg per day for 3 months) while a recent report has used the antibiotic and anti-inflammatory properties of minocycline (100 mg/day).30,31
Other drugs tried include low-dose naltrexone with/without oral magnesium,32–36 oral magnesium chloride,35–40 vitamin D,39,40 afamelanotide,41 oral glycopyrrolate42 and liraglutide43 [Table 1]. Biologicals like adalimumab,44 etanercept45 and dupilumab,46–49 have also been reported to be useful. Apremilast,50–52 and oral thalidomide53 have been used in isolated cases. Lately, JAK inhibitors (upadacitinib, abrocitinib) have been reported to be useful in HHD.54,55 Dupilumab and JAKibs possibly work via their inhibitory effect on IL-4 and IL-13 which have been shown to lead to a significant decrease in the peak amplitude of Ca2+ influx.56
| Systemic agents | Mechanism of action in HHD | Comments/Dose | Level of evidence |
|---|---|---|---|
| Doxycycline 30 | Antimicrobial, anti-inflammatory property, anticollagenase activity via inhibition of matrix metalloproteinases | Recurrence is seen after stoppage of therapy | Case series |
| Oral corticosteroids |
Anti-inflammatory action Reduces protease activity Reduces expression of ATP2C1 |
Recurrence after stoppage of the drug is seen and hence good as a ‘rescue therapy’ | Retrospective study and large case series |
| Oral retinoids 22–24 |
Normalise the epidermal turnover, reduce the inflammation Reduces expression of ATP2C1 |
Acitretin 25 mg/day (10–50 mg/day) Used alone, in adjunct with cyclosporine, NB UVB Etretinate 25–60 mg daily, Alitretinoin 20–30 mg daily or isotretinoin |
Case series |
| Cyclosporine 25 |
Anti-inflammatory Reduces expression of ATP2C1 |
Rapid control of flare, 2.8–3.4 mg/kg/day Cannot be used on a long-term basis |
Case reports |
| Methotrexate 26,27 | Anti-proliferative and anti-inflammatory (especially at low doses) | 7.5–15 mg weekly | Case reports |
| Thalidomide 53 | Thalidomide inhibits TNF alpha that causes downregulation of NOTCH 1 signalling (upregulated in HHD), thereby reducing ROS formation and leading to proper terminal differentiation of keratinocytes |
Refractory HHD Started on oral thalidomide 50 mg once daily (OD) for 24 weeks |
Case report |
| Low-dose naltrexone 32–36 | Modulates calcium transportation dysfunction by inhibiting toll like receptor (TLR) 4, leading to further reduction in Interleukin (IL) 6, TNF alpha and nitric oxide levels | The dose varies from 9 mg to 50 mg. A better option seems to be to use 0.1 mg/kg/day | Case reports and small case series |
| Magnesium chloride 35–40 | Mg2+ inhibits the extrusion of Ca2+ ion via inhibiting the plasma membrane Ca2+ ATPase pump on the human RBC, thereby increasing the intracellular Ca2+ stores and preventing acantholysis | Has been used alone and in combination with high-dose oral vitamin D (8,000 IU) & low-dose oral naltrexone | Case reports |
| Vitamin D 39–40 | Increases intracellular calcium levels, thus reducing acantholysis | High-dose oral vitamin D (up to 800–8,000 IU) has been tried | Case reports |
| Afamelanotide 41 |
Anti-oxidative effects Restores defective keratinocyte proliferative activity |
Injection, 16 mg sustained release implant, 30 days apart | Case report |
| Glycopyrrolate 42 | Reduces hyperhidrosis by blocking M3 muscarinic receptors on eccrine sweat glands and thereby reducing Ach release from the presynaptic terminal | Combination of topical mid-potent steroid, oral minocycline 50 mg BD and oral glycopyrrolate 1 mg OD (combination for 1 month) à Thereafter glycopyrrolate monotherapy | Case report |
| Liraglutide 43 | GLP 1 agonist, used as an adjunct for HHD for weight reduction and decreasing friction |
Useful in the case of concomitant obesity and diabetes (Formulation–subcutaneous injection 1.8 mg) |
Single case report |
| Apremilast 50–52 | Specific inhibition of PDE-4, leading to Th1 and Th17 inhibition and a decrease in CXCL10 release by the keratinocytes which mediates inflammations and flares in HHD | Has been used in dose of 30 mg BD either alone or in combination with dermabrasion and BTX-A | Case reports |
| Dupilumab 46–49 | Fully humanised monoclonal Ab against IL-4a receptor subunit that interferes with signalling of IL-4 and IL-13. IL-4 acts via CCR1 and CCR5 (which inhibit increase in intracellular calcium and actin depolarisation) | Given as subcutaneous injection 600 mg loading dose, thereafter 300 mg every other week |
Case series Case reports |
|
Adalimumab 44 Etanercept 45 |
TNF alpha modifies intracellular calcium signalling, induces inositol phosphate turnover and also has a role in secondary infection in HHD |
Adalimumab: 40 mg every other week Etanercept: 25 mg/week for 1 month, then 50 mg/week for 6 months, then 75 mg/week |
Case report |
| Oral JAKibs 54–55 | Skin barrier defect in HHD leads to Th2-mediated inflammation via IL-4 and IL-13. These cytokines mediate their action via JAK1 and STAT6, hence JAK1 inhibitors play a role in reducing inflammation |
Upadacitinib (15 mg daily) Abrocitinib (100 mg/day) |
Case report |
| Azathioprine 28 | Dose not mentioned | Case reports |
BTX-A: Botulinum toxin A, CCR:Chemokine receptor, CXCL: chemokine (C-X-C motif) ligand, IU: International Unit, GLP: Glucagon like peptide, NOTCH 1: Neurogenic locus notch homolog protein 1, PDE- Phosphodiesterase, ROS: Reactive oxygen species, HHD: Hailey Hailey disease, TNF: Tumpur Necrosis Factor, JAKib: Janus Kinase inhibitor, ATP2C1: ATPase secretory pathway Ca2+ transporting 1.
Procedural Therapy and Surgical Intervention
Some authors report positive results with intralesional botulinum toxin type A, as a result of reduced lesional sweating, reducing chances of maceration and secondary infections.57,58 The modality has been shown to have good tolerance and minimal adverse effects. Combination treatment using Narrow Band (NB) UVB along with prednisolone59/acitretin60/alitretinoin61 has been shown to achieve resolution in refractory cases. Surgical modalities used for limited extent disease include dermabrasion62 and ablative lasers (CO2 & erbium: YAG).58,63 Surgical excision should ideally be done with split skin grafts.64 If so performed, a mid-dermis depth should be achieved. Other locally directed interventions include electron beam therapy and 5-aminolevulinic acid (ALA)-Photodynamic therapy (PDT).65–67
Mechanistic Targets of Existent Therapeutic Agents
The varied therapeutic agents in HHD aim to target various proven and possible pathogenetic mechanisms. An overview of the sequential steps that lead to HHD is given in Figure 2. Some of the agents address the trigger factors and thus combinations or sequential oral antimicrobial agents have been tried as an initial step in the management of HHD. Of these, doxycycline seems to have the maximum evidence.30 This is followed by agents that reduce sweating including botulinum toxin injections,47,48 followed by measures to reduce weight and friction. The diverse triggers result in a shift in the tissue cytokine environment, which appears to be limited to Th2 cytokines, along with impaired calcium metabolism caused by defects in the Golgi apparatus’s secretory pathway Ca2+/Mn2+ ATPase.
Thus, after addressing the triggers, drugs are needed to target the local cytokine-mediated inflammation and secretory defects. Of the drugs listed in Table 2, there is a need to explore drugs that can address both the cytokine dysfunction and the secretory defect. This includes retinoids, corticosteroids, ciclosporin, tacrolimus, and vitamin D3 analogues which have been shown to address the activity of the ATP2C1 gene and counteract the reduced calcium levels.9 Naltrexone has been a novel addition though the ideal dose needs to be rationalised.33–35 Lately, drugs that target the cytokines have been used in HHD and include dupilumab46–49 and JAK inhibitors56 both of which target the Th2 cytokines5,6,9,56 which are implicated in the process of calcium imbalance in HHD. While many other drugs have been tried, they are usually effective in combination which itself is a reflection on their relative inferiority in monotherapy of HHD. The various agents are listed according to their level of evidence in Table 3.
| Infectious triggers | Topical antibiotics, systemic antibiotics (tetracycline, minocycline, doxycycline, dapsone, erythromycin, flucloxacillin), topical antifungals (miconazole) |
| Inflammation | Oral corticosteroids, dapsone, immunosuppressive agents (cyclosporine, methotrexate) |
| Sweating | Topical aluminium chloride, oral glycopyrrolate, oral oxybutynin, botulinum toxin A |
| Weight | Liraglutide |
|
Keratinocyte Differentiation |
Afamelanotide, oral retinoids, thalidomide |
| Cytokines | Dupilumab, TNF alpha inhibitors (adalimumab, etanercept), JAK inhibitors |
|
Calcium-dependent secretory Defect |
Low-dose naltrexone, vitamin D analogues, oral magnesium chloride |
| Tissue removal techniques | NB-UVB, PDT, EBRT, dermabrasion, ablative lasers (CO2 & Er: YAG), split skin grafts |
NBUVB-Narrow band UVB, PDT- Photodynamic therapy, ERBT- External beam radiotherapy, Er: YAG: Erbium YAG. CO2: Carbon dioxide, HHD: Hailey Hailey Disease, TNF: Tumour necrosis factor, JAK: Janus kinase.
| Topical | Systemic | Surgical | |
|---|---|---|---|
| First line | Topical corticosteroids and antimicrobial agents | Oral doxycycline 100 mg/day × 3 months | |
| Second line |
Calcitriol 3 mcg/g ointment Tacrolimus, 0.1% ointment Topical aluminium chloride |
Dapsone: 100 to 200 mg daily to as low as 50 mg every other day | |
| Cyclosporine: 2.5 mg/kg/day | |||
| Oral glycopyrrolate: 1 mg daily | |||
| Third line | Methotrexate: 15 mg/week | Excision followed by split-thickness skin grafting | |
| Acitretin: 0.25–0.5 mg/kg/day mg | EBRT, PDT, LASER | ||
| Dupilumab: 600 mg loading dose F/B 300 mg every 2 weeks | |||
| Liraglutide: 0.6–1.2 mg daily | |||
| Upadacitinib: 15 mg daily | |||
| Magnesium Chloride: 300 mg | |||
| Naltrexone: 3– 4.5 mg |
EBRT- External beam radiotherapy, PDT- Photodynamic therapy
First line: DBRCT, placebo-controlled studies, retrospective studies; second line: Large case series, retrospective studies; third line: case reports
Surgical modalities, including lasers, remove the involved tissue62–64 which is an inelegant method of treating HHD. In most cases, surgery should be the last resort because ir is fraught with the possibility of local recurrence and sequelae.
Conclusion
While the basic defect in HHD is known and the resultant pathology is that of a decrease in intracellular calcium levels (decreased CaR response and/or defective protein processing in the ER/Golgi apparatus), drugs that address this pivotal defect have yet not been rigorously tested. As triggers can upset calcium homeostasis, they should be addressed in all cases. To date, the exact link between cytokines and the defect in the secretory mechanism is not known though possibly a cause-and-effect hypothesis is probable and accounts for the efficacy of JAKis and dupilumab. Certain drugs that act on multiple pathways would make them superior to the existing drugs. There is as yet no double-blinded randomised controlled trial or real-world comparison of drugs to arrive at an ideal drug regimen so that treatment can be guided based on the existing level of evidence. There is an urgent need for therapeutic agents that address the basic pathogenetic pathway in HHD and we hope that future studies will address this concern with pre- and post-treatment proteomic and mutational expression studies that can determine the ideal drug for HHD. But, it is important to reiterate that being a genetic disorder long-term remissions off therapy are difficult to achieve at the present time.
Acknowledgements
Figure 1 was created by a medical illustrator at CBS Publishers, New Delhi.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
References
- Calcium pumps and keratinocytes: lessons from Darier’s disease and Hailey-Hailey disease. Br J Dermatol. 2004;150:821-8.
- [CrossRef] [PubMed] [Google Scholar]
- Identification of ATP2C1 mutations in the patients of Hailey-Hailey disease. BMC Med Genet. 2020;21:120.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Hailey-Hailey disease: investigation of a possible compensatory SERCA2 up-regulation and analysis of SPCA1, p63, and IRF6 expression. Arch Dermatol Res. 2015;307:143-9.
- [CrossRef] [PubMed] [Google Scholar]
- Hailey-Hailey disease: A fold (intertriginous) dermatosis. Clin Dermatol. 2015;33:452-5.
- [CrossRef] [PubMed] [Google Scholar]
- Role of pro-inflammatory cytokines in the pathophysiology of herpes simplex virus superinfection in Darier’s disease. J Dermatol. 2021;48:1607-11.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of ultraviolet B irradiation, proinflammatory cytokines and raised extracellular calcium concentration on the expression of ATP2A2 and ATP2C1. Br J Dermatol. 2005;152:697-701.
- [CrossRef] [PubMed] [Google Scholar]
- Disrupted desmosomes in induced lesions of familial benign chronic pemphigus. J Cutan Pathol. 1979;6:418-24.
- [CrossRef] [PubMed] [Google Scholar]
- Hailey-Hailey disease diagnosed based on an exacerbation of contact dermatitis with topical crotamiton. Eur J Dermatol. 2014;24:263-4.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of drugs and anticytokine anti-bodies on expression of ATP2A2 and ATP2C1 in cultured normal human keratinocytes. Br J Dermatol. 2005;152:920-4.
- [CrossRef] [PubMed] [Google Scholar]
- Hailey-Hailey disease and review of management. J Drugs Dermatol. 2014;13:1254-7.
- [PubMed] [Google Scholar]
- Management of familial benign chronic pemphigus. Clin Cosmet Investig Dermatol. 2016;9:281-90.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Management of Hailey-Hailey disease with Castellani paint. JAAD Case Rep. 2019;5:855-6.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Treatment of Hailey-Hailey disease with tacrolimus ointment and clobetasol propionate foam. J Drugs Dermatol. 2004;3:200-3.
- [PubMed] [Google Scholar]
- Squamous cell carcinoma arising from a localized vulval lesion of Hailey-Hailey disease after tacrolimus therapy. Am J Obstet Gynecol. 2010;203:e5-7.
- [CrossRef] [PubMed] [Google Scholar]
- Topical aluminum chloride as a treatment option for Hailey-Hailey disease: a remarkable therapeutic outcome case report. An Bras Dermatol. 2024;99:321-3.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Topical gentamicin for the treatment of genetic skin diseases. J Invest Dermatol. 2018;138:731-4.
- [CrossRef] [PubMed] [Google Scholar]
- Successful treatment of Hailey–Hailey disease with topical 5-fuorouracil. Br J Dermatol. 2009;161:967-8.
- [CrossRef] [PubMed] [Google Scholar]
- Improvement of Darier’s disease on treatment with topical 5-fluorouracil. Ann Dermatol Venereol. 2006;133:366-8.
- [CrossRef] [PubMed] [Google Scholar]
- Hailey-Hailey disease: effective treatment with topical cadexomer iodine. J Dermatolog Treat. 2011;22:304-5.
- [CrossRef] [PubMed] [Google Scholar]
- Topical timolol 0.5% as a novel therapeutic approach for Hailey-Hailey disease. Clin Exp Dermatol. 2023;49:76-8.
- [CrossRef] [PubMed] [Google Scholar]
- Improvement of Hailey-Hailey Disease With Topical Cinacalcet, 3%, Ointment. JAMA Dermatol. 2023;159:669-71.
- [CrossRef] [PubMed] [Google Scholar]
- Successful treatment of Hailey-Hailey disease with acitretin. J Drugs Dermatol. 2007;6:734-6.
- [PubMed] [Google Scholar]
- Acitretin as a successful treatment for Hailey-Hailey disease. Clin Exp Dermatol. 2019;44:450-2.
- [CrossRef] [PubMed] [Google Scholar]
- Hailey-Hailey Disease: An Update Review with a Focus on Treatment Data. Am J Clin Dermatol. 2020;21:49-68.
- [CrossRef] [PubMed] [Google Scholar]
- Benign familial chronic pemphigus (Hailey-Hailey disease) responds to cyclosporin. Clin Exp Dermatol. 1995;20:70-2.
- [CrossRef] [PubMed] [Google Scholar]
- Methotrexate for refractory Hailey-Hailey disease. J Eur Acad Dermatol Venereol. 2010;24:106.
- [CrossRef] [PubMed] [Google Scholar]
- Oral methotrexate in the treatment of Hailey‒Hailey disease: a case report. Acta Dermatovenerol Alp Pannonica Adriat. 2024;33 actaapa.2024.11
- [Google Scholar]
- Hailey-Hailey disease: the role of azathioprine an immunomodulator. Pan Afr Med J. 2019;32:65.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Benign familial chronic pemphigus treated with dapsone. Arch Dermatol. 1971;103:262-5.
- [CrossRef] [PubMed] [Google Scholar]
- Familial benign chronic pemphigus and doxycycline: a review of 6 cases. J Eur Acad Dermatol Venereol. 2014;28:370-3.
- [CrossRef] [PubMed] [Google Scholar]
- generalised Hailey-Hailey disease with flexural keratotic papules: An interesting presentation and remarkable response with minocycline. Dermatol Ther. 2019;32:e12945.
- [CrossRef] [PubMed] [Google Scholar]
- Generalized familial benign chronic pemphigus (Hailey-Hailey disease) treated successfully with low-dose naltrexone. JAAD Case Rep. 2018;4:725-7.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Variable response to naltrexone in patients with Hailey-Hailey disease. JAMA Dermatol. 2018;154:362-3.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The importance of dosage for naltrexone treatment in Hailey-Hailey disease. JAAD Case Rep. 2022;23:155-7.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Hailey-Hailey disease treated successfully with naltrexone and magnesium. JAAD Case Rep. 2019;5:760-2.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Improvement in Hailey-Hailey disease with a combination of low-dose naltrexone and oral magnesium chloride: A case report. SAGE Open Med Case Rep. 2020;8:2050313X20984121.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Oral magnesium chloride: A novel approach in the management of Hailey–Hailey disease. Indian J Dermatol Venereol Leprol. 2017;83:259-62.
- [CrossRef] [PubMed] [Google Scholar]
- Efficacy of magnesium chloride in the treatment of Hailey-Hailey disease: from serendipity to evidence of its effect on intracellular Ca(2+) homeostasis. Int J Dermatol. 2015;54:543-8.
- [CrossRef] [PubMed] [Google Scholar]
- A Case of Hailey-Hailey Disease Managed With Oral Magnesium Citrate and High-Dose Vitamin D3. J Cutan Med Surg. 2018;22:362-4.
- [CrossRef] [PubMed] [Google Scholar]
- Hailey-Hailey disease successfully treated with vitamin D oral supplementation. Dermatol Ther. 2019;32:e12767.
- [CrossRef] [PubMed] [Google Scholar]
- Efficacy of the melanocortin analogue Nle4-D-Phe7-α-melanocyte-stimulating hormone in the treatment of patients with Hailey-Hailey disease. Clin Exp Dermatol. 2014;39:168-75.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Oral glycopyrrolate for the treatment of Hailey-Hailey disease. JAMA Dermatol. 2015;151:328-9.
- [CrossRef] [PubMed] [Google Scholar]
- Liraglutide, a GLP-1 agonist, as a new adjunct treatment in Hailey-Hailey disease: a case report. Clin Exp Dermatol. 2024;49:409-11.
- [CrossRef] [PubMed] [Google Scholar]
- Adalimumab therapy used successfully for recalcitrant Hailey-Hailey disease. JAAD Case Rep. 2022;29:173-6.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Case reports of etanercept in infammatory dermatoses. J Am Acad Dermatol. 2006;54:139-42.
- [Google Scholar]
- Dupilumab in Hailey-Hailey disease: a case series. J Eur Acad Dermatol Venereol. 2022;36:e776-9.
- [CrossRef] [PubMed] [Google Scholar]
- Hailey-Hailey disease treated with dupilumab: a case series. Br J Dermatol. 2021;185:680-2.
- [CrossRef] [PubMed] [Google Scholar]
- Successful Treatment of Hailey-Hailey Disease With Dupilumab. Actas Dermosifiliogr. 2023;114:914-5.
- [CrossRef] [PubMed] [Google Scholar]
- A case of Hailey-Hailey disease successfully treated with dupilumab. Int J Dermatol. 2022;61:1427-8.
- [CrossRef] [PubMed] [Google Scholar]
- Treatment of Severe Hailey-Hailey Disease With Apremilast. JAMA Dermatol. 2018;154:1453-6.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Combination of dermabrasion and Apremilast for Hailey-Hailey disease. Ital J Dermatol Venerol. 2021;156:727-8.
- [CrossRef] [PubMed] [Google Scholar]
- Apremilast in combination with botulinum toxin-A injection for recalcitrant Hailey-Hailey disease. Int J Dermatol. 2022;61:600-2.
- [CrossRef] [PubMed] [Google Scholar]
- Old drug, new tricks - successful treatment of Hailey-Hailey disease with thalidomide. Australas J Dermatol. 2021;62:94-6.
- [CrossRef] [PubMed] [Google Scholar]
- Refractory Hailey-Hailey disease cleared with upadacitinib. JAAD Case Rep. 2023;41:64-7.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Improvement of Hailey-Hailey disease with abrocitinib. Clin Exp Dermatol. 2023;48:532-3.
- [CrossRef] [PubMed] [Google Scholar]
- SMOC1 and IL-4 and IL-13 Cytokines Interfere with Ca2+ Mobilization in Primary Human Keratinocytes. J Invest Dermatol. 2021;141:1792-1801.e5.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Botulinum toxin type A for the treatment of axillary Hailey-Hailey disease. Dermatol Surg. 2000;26:371-4.
- [CrossRef] [PubMed] [Google Scholar]
- Interventional treatments for Hailey-Hailey disease. J Am Acad Dermatol. 2017;76:551-8.e3.
- [CrossRef] [PubMed] [Google Scholar]
- Narrowband UV-B Phototherapy in the Treatment of Generalized Hailey-Hailey Disease. Actas Dermosifiliogr (Engl Ed). 2018;109:924-7.
- [CrossRef] [PubMed] [Google Scholar]
- Treatment of Hailey-Hailey disease with narrowband phototherapy and acitretin: A case report. SAGE Open Med Case Rep. 2019;7:2050313X19845221.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Combined therapeutic use of oral alitretinoin and Narrowband ultraviolet-B therapy in the treatment of Hailey-Hailey disease. Dermatol Rep. 2014;6:5604-23.
- [CrossRef] [Google Scholar]
- Refractory Hailey-Hailey disease successfully treated with sandpaper dermabrasion. Skinmed.. 2011;9:263-4.
- [PubMed] [Google Scholar]
- Carbon dioxide laserabrasion: a new approach to management of familial benign chronic pemphigus (Hailey-Hailey disease) J Dermatol Surg Oncol. 1987;13:1187-94.
- [CrossRef] [PubMed] [Google Scholar]
- Traitement chirurgical de la maladie de Hailey-Hailey ou pemphigus chronique bénin familial. A propos d’une localisation sous-mammaire [Surgical treatment of Hailey-Hailey disease or benign chronic familial pemphigus. Apropos of a case of submammary localization] Ann Chir Plast Esthet. 1993;38:568-71.
- [PubMed] [Google Scholar]
- Successful treatment of Hailey-Hailey disease with aminolevulinic acid photodynamic therapy. Ann Dermatol. 2015;27:222-3.
- [CrossRef] [PubMed] [Google Scholar]
- Photodynamic therapy with 5-aminolevulinic acid for recalcitrant familial benign pemphigus (Hailey-Hailey disease) J Am Acad Dermatol. 2002;47:740-2.
- [CrossRef] [PubMed] [Google Scholar]
- Experience with photodynamic therapy in Hailey-Hailey disease. J Dermatolog Treat. 2008;19:288-90.
- [CrossRef] [PubMed] [Google Scholar]







