Translate this page into:
A study of blood CD3+, CD4+, and CD8+ T cell levels and CD4+:CD8+ ratio in vitiligo patients
2 Department of Biochemistry, Pt. J.N.M. Medical College, Raipur, Chhattisgarh, India
Correspondence Address:
P K Nigam
D-30-A, Shailendra Nagar, Raipur - 492 001, Chhattisgarh
India
| How to cite this article: Nigam P K, Patra P K, Khodiar P K, Gual J. A study of blood CD3+, CD4+, and CD8+ T cell levels and CD4+:CD8+ ratio in vitiligo patients. Indian J Dermatol Venereol Leprol 2011;77:111 |
Sir,
Recent researches focus on a melanocyte-specific cytotoxic T-cell immune reaction in the melanocyte destruction, [1] and by immune infiltrates and skin-homing autoreactive T cells [2] that target melanocyte-specific Ags, such as melan-A/MART1, Gp100, tyrosinase, and tyrosinase-related proteins, TRP-1 and TRP-2, [3] in the pathogenesis of vitiligo. It has been shown that CD4-dependent destruction of melanocytes is partially inhibited by blocking Fas-Fas ligand interactions and also highlights the importance of local control of autoimmunity, as vitiligo remains patchy and never proceeds to confluence even when Ag and autoreactive CD4 + T cells are abundant. [1] In view of these proposed roles of T cell activity in vitiligo, a study of CD3, CD4 and CD8+ T cells was done in vitiligo patients from this part of the country.
Fifty patients of non-segmental vitiligo were taken for study. A detailed history was taken and recorded, and patients having any known associated immunomodulator disease such as diabetes, tuberculosis, thyroiditis, HIV or any other viral infection were excluded from the study. Five milliliters of venous blood sample was collected from the patient in a vial with ethylenediaminetetraacetic acid (EDTA) for CD count. One hundred microliters of this whole blood was taken in a test tube and 20 ΅l antibodies (CD3, CD4 and CD8) were added. The percentage of CD3+ CD4+ and CD3+ CD8+ lymphocytes within the total lymphocyte light-scatter gate were determined by flow cytometry, using the two color antibody combinations, and adjusted for purity by dividing the measured subset percentage by the percent purity. Absolute CD3+ CD4+ and CD3+ CD8+ lymphocyte counts (herein referred to as CD4 T-cell count and CD8 T-cell counts, respectively) were calculated by the absolute lymphocyte count. The results were compared with 25 age- and sex-matched controls which were taken from the family members of the patients.
Among the 50 consecutive patients studied, there were 21 males and 29 females. The age of the patients ranged from 7 to 51 years. Majority of the patients (38%) belonged to the age group of 20-30 years. The duration of vitiligo ranged from 1 month to 6 years. A definite family history of vitiligo was present in 4 (8%) cases. Thirty-four patients had active and 16 had stable vitiligo disease. Absolute and relative counts of lymphocyte subtypes were normal in all the patients. The mean absolute CD cell counts in vitiligo patients and controls are given in [Table - 1].
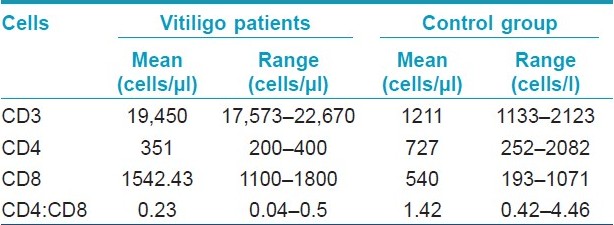
Pathogenetic role for melanocyte specific CD8+ T cells in vitiligo came from the direct correlation between their frequency within the total T-cell pool and disease activity. [4] The consistent presence of immune infiltrates, composed of T cells and macrophages, in skin of patients with the generalized type of vitiligo, suggests that an ongoing local immune response is associated with melanocyte destruction. [5] The immune infiltrates are mainly composed of T cells and macrophages. The number of infiltrating T cells, mostly consisting of CD8 + /CD45RO + cells, was highest in the peri-lesional area. It has also been shown that the presence of T cells and macrophages in inflammatory vitiligo skin parallels melanocyte disappearance. [2] High frequencies of melanocyte specific CD8+ T lymphocytes are found in vitiligo patients. [4] As seen in the present study, the mean count of CD4+ and CD8+ T-cell count was deviated from its normal value so CD4+:CD8+ ratio also deviated in them, i.e., its value was less than 1. The mean absolute CD4+ and CD8+ count was further lowered in vitiligo patients with active disease as compared to that of stable vitiligo cases. However, there was no statistical significant difference in CD4+:CD8+ ratio among these two groups. A decrease in the CD4+ T-cell population and increase in the CD8+ T-cell population have also been observed in vitiligo cases by Grimes et al. [6] and Halder et al. [7] who had documented a perceptible decrease in the ratio of CD4+/CD8+. Although recently Pichler et al. [8] reported an increase in CD4+/CD8+ ratio in vitiligo patients, 40% of their patients had associated autoimmune thyroiditis which might have influenced their results.
The study concludes that the estimation of cell surface markers in vitiligo patients has additional diagnostic value along with clinical data and routine investigations done for vitiligo. Estimation of CD molecules (CD3+, CD4+, and CD8+) count and CD4+:CD8+ ratio may be a useful tool for the diagnosis of vitiligo, especially in cases of early vitiligo having diagnostic difficulties or with lesions over the face where biopsy is not feasible due to cosmetic reasons. Further large sample studies are needed to substantiate our findings.
(Note: The instrument, BD FACS Calibur, used for this study for immunophenotyping application and the reagents were supplied by Becton Dickinson, Franklin Lakes, NJ USA 07417.)
| 1. |
Lambe T, Leung JC, Bouriez-Jones T, Silver K, Makinen K, Crockford TL, et al. CD4 T cell-dependent autoimmunity against a melanocyte neoantigen induces spontaneous vitiligo and depends upon Fas-Fas ligand interactions. J Immunol 2006;177:3055-62.
[Google Scholar]
|
| 2. |
Le Poole IC, van den Wijngaard RM, Westerhof W, Das PK. Presence of T cells and macrophages in inflammatory vitiligo skin parallels melanocyte disappearance. Am J Pathol 1996;148:1219-28.
[Google Scholar]
|
| 3. |
Wankowicz-Kalinska A, Le Poole C, van den Wijngaard R, Storkus WJ, Das PK. Melanocyte-specific immune response in melanoma and vitiligo: Two faces of the same coin? Pigment Cell Res 2003;16:254-60.
[Google Scholar]
|
| 4. |
Ogg GS, Rod Dunbar P, Romero P, Chen JL, Cerundolo V. High frequency of skin-homing melanocyte-specific cytotoxic T lymphocytes in autoimmune vitiligo. J Exp Med 1998;188:1203-8.
[Google Scholar]
|
| 5. |
van den Wijngaard R, Wankowicz-Kalinska A, Le Poole C, Tigges B, Westerhof W, Das P. Local immune response in skin of generalized vitiligo patients. Destruction of melanocytes is associated with the prominent presence of CLA+ T cells at the perilesional site. Lab Invest 2000;80:1299-309.
[Google Scholar]
|
| 6. |
Grimes PE, Ghoneum M, Stockton T, Payne C, Kelly AP, Alfred L. T cell profiles in vitiligo. J Am Acad Dermatol 1986;14:196-201.
[Google Scholar]
|
| 7. |
Halder RM, Walters CS, Johnson BA, Chakrabarti SG, Kenney JA Jr. Aberrations in T lymphocytes and natural killer cells in vitiligo: A flow cytometric study. J Am Acad Dermatol 1986;14:733-7.
[Google Scholar]
|
| 8. |
Pichler R, Sfetsos K, Badics B, Gutenbrunner S, Berg J, Aubφck J. Lymphocyte imbalance in vitiligo patients indicated by elevated CD4+/CD8+ T-cell ratio. Wien Med Wochenschr 2009;159:337-41.
[Google Scholar]
|
Fulltext Views
2,153
PDF downloads
2,301





