Translate this page into:
Carbamazepine-induced follicular mucinosis-like drug eruption
Corresponding author: Prof. Zhongxiang Shi, Department of Dermatology, Shandong Provincial Hospital for Skin Disease & Shandong Provincial Institute of Dermatology and Venereology, Shandong First Medical University & Shandong Academy of Medical Sciences, Shandong, China. szxsd2003@sina.com
-
Received: ,
Accepted: ,
How to cite this article: Ran D, Wang N, Bao F, Liu Y, Zhou G, Zhou S, et al. Carbamazepine-induced follicular mucinosis-like drug eruption. Indian J Dermatol Venereol Leprol 2022;88:826-8.
Sir,
Drug eruptions caused by carbamazepine have been widely reported and often present as drug reaction with eosinophilia and systemic symptoms (DRESS), Stevens-Johnson syndrome, toxic epidermal necrolysis and maculopapular rash.1-3 Here, we describe a case of drug eruption caused by carbamazepine that showed histopathological features of follicular mucinosis.
A 70-year-old Chinese man presented with pruritic follicular papules and erythema of the trunk and extremities along with oedema and erythema of the face, lips and ears of three days duration. Initially the lesions presented with oedema and erythema of the head and face, followed by follicular papules on the trunk and extremities. He had no other symptoms. The rash appeared five days after starting carbamazepine 100 mg twice a day orally for trigeminal neuralgia. Carbamazepine was discontinued after the onset of the rash. There was no history of recent infections, comorbidities or any other drug intake.
On clinical examination, he was afebrile. Vitals were stable with no significant lymphadenopathy. There was erythema and oedema of the face, along with erythematous follicular papules and plaques (some of them showing scaling) involving the entire body [Figures 1a to c]. Laboratory examination revealed leucocytosis (11000 cells/mm3), neutrophilia (9000 cells/mm3) and a normal eosinophil count (30 cells/mm). Peripheral blood smear, liver function tests, serology for human immunodeficiency virus, chest radiography, transabdominal colour doppler scanning, and electrocardiography showed no abnormalities. Histopathology of a follicular papule on the back showed focal spongiosis, obvious oedema of the folliculus pili and sebaceous gland epithelium [Figure 2a] along with lymphocytes and histiocytes infiltrating the hair follicles and sebaceous glands and its surrounding areas [Figure 2b]. Alcian blue staining showed mucin deposition in the hair follicles [Figure 2c]. Immunohistochemistry was not performed as atypical lymphocytes were not observed in the biopsy specimen.
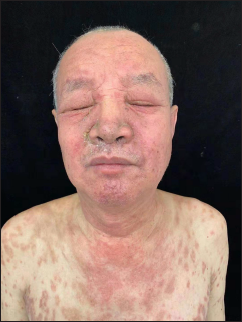
- Erythema, oedema, erythematous papules and scaling involving the face
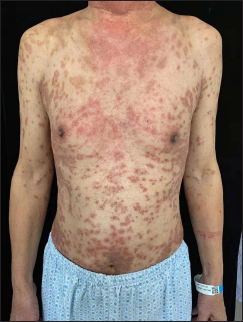
- Erythematous and hyperpigmented follicular papules and plaques on the trunk and upper limbs
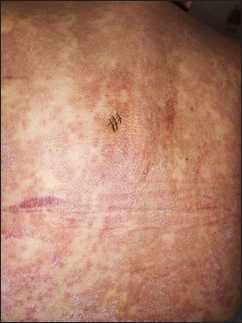
- A close-up view of the lesions on the back showing follicular papules and plaques with scaling
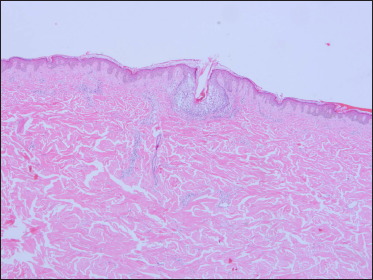
- Mild hyperkeratosis of the epidermis, focal spongiosis, obvious oedema of the folliculus pili and sebaceous gland epithelium (H and E, × 40)
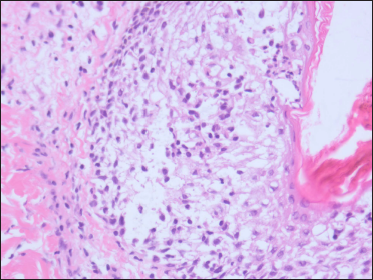
- Lymphocytes and histiocytes infiltrating within and around the hair follicles and sebaceous glands (H and E, × 400)
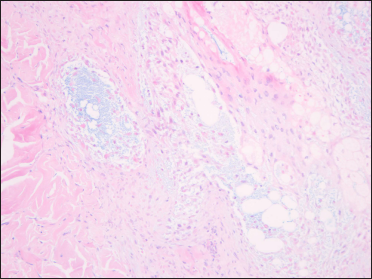
- Mucin deposition in the hair follicles (Alcian blue, × 200)
The histopathology findings were consistent with the diagnosis of follicular mucinosis. Review of his history did not reveal any similar rash in the past. Suspecting a carbamazepine-induced follicular mucinosis-like drug reaction, methylprednisolone 48 mg/day was administered intravenously with gradual tapering and stopped.
After 13 days of treatment, the lesions resolved with post-inflammatory pigmentation [Figures 3a and b], supporting the diagnosis of a drug reaction rather than primary follicular mucinosis. There was no recurrence during the nine months follow up period.
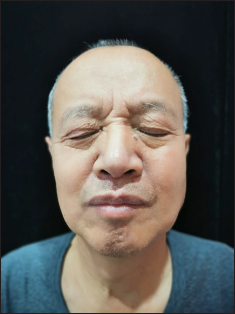
- Resolution of lesions on the face
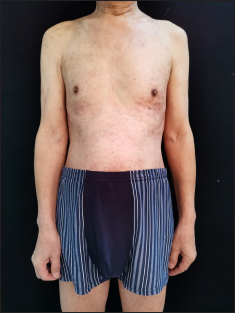
- Post-inflammatory pigmentation on the trunk after treatment
Follicular mucinosis is an epithelial reaction that follows many inflammatory, infectious, and neoplastic conditions and is characterised by the accumulation of mucin within hair follicles4. There are three forms of follicular mucinosis. Primary form with a short duration usually manifests in children and young adults, with one or two lesions seen on the head and neck regions. The lesions resolve spontaneously within two months to two years without recurrences. Another primary form with a prolonged course has been described, which appears in older patients with numerous, sometimes disseminated, lesions that mostly appear on the face, trunk and extremities. Despite its chronic course with multiple recurrences, it is a benign condition. The third form affects those aged 40–70 years and is characterised by generalised, multiple infiltrated plaques. This variant is associated with T-cell lymphomas, especially mycosis fungoides.4
In our case, the older age of onset, disseminated lesions and shorter duration excluded the primary form of follicular mucinosis. Although disseminated lesions were present, this was unlikely to be a case of the primary disseminated form with a prolonged course. Prompt resolution on withdrawal of carbamazepine and administration of systemic corticosteroid, a recurrence-free follow-up period of nine months and older age of disease onset were the differentiating features. Follicular mucinosis associated with lymphoma was unlikely in view of the histopathological findings. In addition, there was a reasonable temporal relationship between the drug exposure and the eruption, and also the lesions resolved with withdrawal of the drug. No alternative etiology, including any disease or other medicines, was likely to be responsible for this adverse event. According to the World Health Organisation–Uppsala Monitoring Centre (WHO-UMC) standardised case causality assessment criteria5, this event would be considered a “probable” adverse reaction to carbamazepine, so we eventually made a diagnosis of carbamazepine-induced follicular mucinosis-like drug eruption.
Carbamazepine inducing a rash that shows histopathology of follicular mucinosis is extremely rare. In the literature, we found two reported cases: one was a case of DRESS, in which the skin biopsy revealed a lymphocytic infiltrate with atypical lymphocytes, exocytosis and follicular mucinosis mimicking mycosis fungoides,6 and another case also resembling mycosis fungoides with histopathology showing atypical intraepidermal lymphocytes and follicular mucinosis.7 Our case showed only mucin deposition in the hair follicles and no atypical lymphocytes, which was an obvious differentiating feature from the previously published cases. He is under long-term follow-up, since literature suggests that histopathology, immunohistochemistry, and even T-cell gene rearrangement studies may not always be able to distinguish lymphoma-associated follicular mucinosis from benign variants.8
We report this case to draw the attention of the clinicians to this rare form of drug eruption (follicular mucinosis-like) induced by carbamazepine.
Ethical Approval: The study was approved by the institutional research ethics committee of Shandong Provincial Hospital for Skin Disease & Shandong Provincial Institute of Dermatology and Venereology, Shandong First Medical University & Shandong Academy of Medical Sciences.
Author contributions: Zhongxiang Shi designed the study, Shengji Zhou provided the case. Delin Ran, Na Wang and Fangfang Bao wrote the paper. Yongxia Liu and Guizhi Zhou did the histopathological examination. All authors have read and approved the final manuscript.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms.
Financial support and sponsorship
Nil.
Conflict of interest
There are no conflicts of interest.
References
- Anticonvulsant hypersensitivity syndrome: Clinic case and literature review. Rev Fac Cien Med Univ Nac Cordoba. 2020;77:211-13.
- [CrossRef] [PubMed] [Google Scholar]
- Recent advances in managing and understanding Stevens-Johnson syndrome and toxic epidermal necrolysis. F1000Res. 2020;9:612.
- [CrossRef] [PubMed] [Google Scholar]
- Genetic susceptibility to carbamazepine-induced cutaneous adverse drug reactions. Pharmacogenet Genomics. 2006;16:297-306.
- [CrossRef] [PubMed] [Google Scholar]
- Follicular mucinosis: Literature review and case report. An bras Dermatol. 2002;77:701-706.
- [Google Scholar]
- Uppsala: The Uppsala Monitoring Centre; 2005. The use of the WHO-UMC system for standardized case causality assessment [monograph on the Internet]
- Follicular mucinosis in a mycosis fungoides-like hypersensitivity syndrome induced by oxcarbamazepine. J Cutan Pathol. 2011;38:1009-1011.
- [CrossRef] [PubMed] [Google Scholar]
- Follicular mucinosis and mycosis-fungoides-like drug eruption due to leuprolide acetate: A case report and review. J Cutan Pathol. 2012;39:1022-1025.
- [CrossRef] [PubMed] [Google Scholar]
- Follicular mucinosis: A critical reappraisal of clinicopathologic features and association with mycosis fungoides and Sézary syndrome. Arch Dermatol. 2002;138:182-9.
- [CrossRef] [PubMed] [Google Scholar]





