Translate this page into:
Colocalization of mucosal vitiligo and oral pemphigus vulgaris
2 Department of Histopathology, Postgraduate Institute of Medical Education and Research, Chandigarh, India
Correspondence Address:
Amrinder J Kanwar
Department of Dermatology, Postgraduate Institute of Medical Education and Research, Chandigarh
India
| How to cite this article: De D, Kanwar AJ, Saikia UN, Jindal R. Colocalization of mucosal vitiligo and oral pemphigus vulgaris. Indian J Dermatol Venereol Leprol 2012;78:111-113 |
Sir ,
Vitiligo is an acquired depigmentation disorder of skin with occasional involvement of other melanized tissues. Though many hypotheses have been proposed regarding its pathogenesis, autoimmune mechanism has achieved maximum support. Pemphigus is an autoimmune disease with immunologic attack targeted against the desmogleins on the keratinocytes. We herein report a case where lesions of vitiligo and pemphigus vulgaris were colocalized. Though many autoimmune diseases have been associated with both the disorders, to the best of our knowledge, this is the third report of association of pemphigus and vitiligo. Interesting aspect in our patient is the colocalization of the lesions, which can shed some light on the pathogenesis of both the disorders.
A 52-year-old woman presented with painful oral blisters of 3-months duration. The lesions started appearing in the anterior aspect of buccal mucosa of both the sides. Individual lesions used to persist for 1 to 2 days followed by spontaneous rupture, leaving painful erosions. She had depigmentation of both the lips and buccal mucosae for 3 years before onset of the present symptoms. None in her family had either depigmented patches or blistering skin disease. She had no other significant medical illness . On dermatologic examination, she had irregular superficial erosions with overlying whitish slough localized to buccal mucosae. In addition, she had depigmented vitiliginous patches involving mucosal aspect of her lips and buccal mucosae [Figure - 1]. No other part of the body was affected by either of the diseases. Mucosal biopsy was taken from one of the small flaccid blisters, which on hematoxylin and eosin staining showed suprabasal cleft with acantholytic cells and keratinocytes in a "row of tombstones" at the base [Figure - 2]. Another field in the same sample showed total absence of melanocytes. Direct immunofluorescence revealed net-like deposition of IgG and C 3 on the surface of the keratinocytes, thus consistent with pemphigus vulgaris. She was treated with oral prednisolone 40 mg/day and azathioprine 50 mg given twice a day, following which her oral blisters and erosions improved, but the vitiliginous patches remained as such.
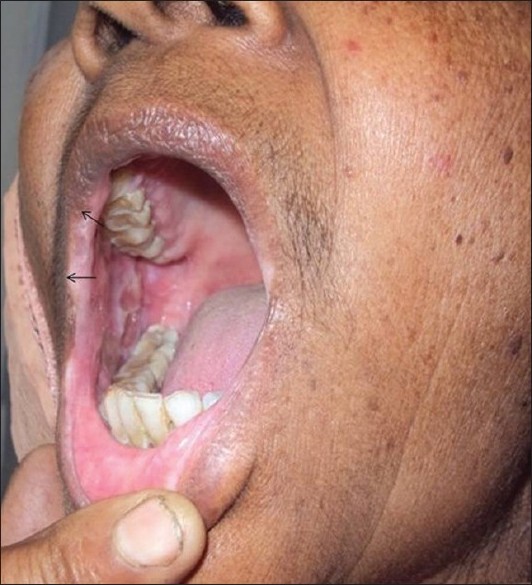 |
| Figure 1: Irregular superficial erosions with overlying whitish slough localized to buccal mucosa and depigmented patches involving mucosal aspect of lips. Irregular borders between normally pigmented and depigmented areas around right angle of mandible have been indicated with arrows |
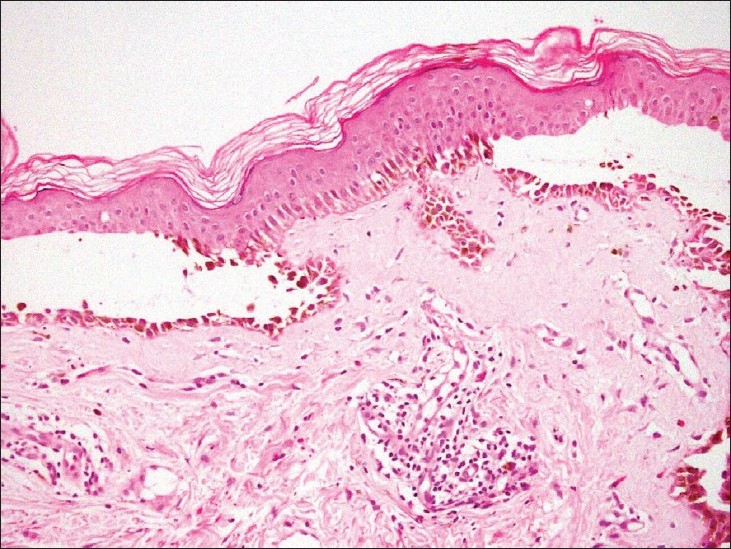 |
| Figure 2: Suprabasal cleft with acantholytic cells and keratinocytes in a "row of tombstones" at the base (H and E, ×140) |
Mucosal involvement of vitiligo is common in pigmented races with lip involvement occurring in up to 50% of vitiligo patients. The incidence in white population is probably underestimated apparently due to inability to detect hypopigmentation in the background of constitutionally pale mucosa. [1] Occasional hyperpigmentation at the periphery of the lesions may be a helpful clue to the diagnosis.
Coexistence of pemphigus and vitiligo has been reported twice previously. In the first report of its kind by Jain et al., [2] a 10-year-old boy was affected by extensive pemphigus and vitiligo with no obvious reported colocalization. In the second report by Yalcin et al., [3] a 38-year-old lady was affected by pemphigus, vitiligo, and Hashimoto′s thyroiditis. One interesting aspect of their case was the localization of pemphigus lesions on the vitiliginous side of the junction between vitiliginous and normal skin. Their patient had autoimmune polyglandular syndrome, characterized by three or more autoimmune diseases in a single patient. [3]
The exact mechanism for association of these two autoimmune dermatologic diseases, i.e., vitiligo and pemphigus, is not known. A common autoimmune process may be responsible. Vitiligo is predisposed by association of HLA DR 4 -DQ 1 haplotypes. [4] The same haplotype is also present in pemphigus patients. [5] Though genetic associations may explain coexistence of two autoimmune diseases, for colocalization, there should be some effective local factors which can explain expression of both the diseases simultaneously at the same site. The following hypotheses can explain the apparent enigma. First, in vitiliginous skin, there is decreased acetylcholinesterase level and thus increased acetylcholine activity. [6] Cholinergic receptors regulate desmosomal adhesion of keratinocytes. Persistent high level of acetylcholine in neuronal synapses may downregulate acetylcholine receptors, thus precipitating pemphigus. Incidentally, acantholysis in pemphigus can also occur in the presence of antibodies against 9-a nicotinic acetylcholine receptor. [7] Second, TNF-a level has been found to be elevated in perilesional skin of pemphigus. The same mediator has been implicated in the pathogenesis of vitiligo also. [8] Third, Schallreuter and Wood [9] have demonstrated low levels of the enzyme thioredoxin reductase in keratinocytes cultured from depigmented skin of vitiligo. This deficiency leads to increased susceptibility to complement-mediated injury. Complements, in addition to autoantibodies, have been implicated in the cascade of pathogenesis of pemphigus vulgaris as well. Lastly, there is intimate functional relationship between keratinocytes and melanocytes. Though in vitiligo melanocytes are destroyed primarily, loss of structural support from keratinocytes leads to melanocytorrhagy. Similarly, autoantibody-mediated destruction of melanocytes leads to damage of keratinocytes and anti-keratinocyte antibodies. This damage of keratinocytes may lead to immunogenicity of keratinocyte antigens and thus pemphigus. Above all, mere coincidence for colocalization of both the entities cannot be ruled out.
Though exact reason for such interesting colocalization remains speculative, further research in this area may throw more light in the pathogenesis of both the diseases.
| 1. |
Kanwar AJ, Parsad D, De D. Mucsoal involvement in vitiligo: A comprehensive review of 241 cases. J Eur Acad Dermatol Venereol 2011;25:1361-3.
[Google Scholar]
|
| 2. |
Jain R, Dogra S, Sandhu K, Handa S, Kumar B. Co- existence of vitiligo and pemphigus vulgaris in an Indian patient. Pediatr Dermatol 2003;20:369-70.
[Google Scholar]
|
| 3. |
Yalcin B, Tamer E, Toy GG, Oztas P, Alli N. Development of pemphigus vulgaris in a patient with vitiligo and Hashimoto's thyroiditis. J Endocrinol Invest 2005;28:558-60.
[Google Scholar]
|
| 4. |
Dunston GM, Halder RM, Ofosu MD. HLA-D region antigens in black patients with vitiligo. J Invest Dermatol 1988;90:555.
[Google Scholar]
|
| 5. |
Ahmed AR, Younis EJ, Khatri K. Major histocompatibility complex haplotype studies in Ashkenazi Jewish patients with pemphigus vulgaris. Proc Natl Acad Sci USA 1990;87:7658-62.
[Google Scholar]
|
| 6. |
Iyengar B. Modulation of melanocytic activity by acetylcholine. Acta Anat (Basel) 1989;136:139-41.
[Google Scholar]
|
| 7. |
Grando SA. Pemphigus in 21st century: New life to an old story. Autoimmunity 2006;39:521-30.
[Google Scholar]
|
| 8. |
Kovacs SO, Missouri SF. Vitiligo. J Am Acad Dermatol 1998;38:647-66.
[Google Scholar]
|
| 9. |
Schallreuter KU, Wood JM. The role of thioredoxin reducatse in the reduction of free radicals at the surface of epidermis. Biochem Biophys Res Commun 1986;136:630-7.
[Google Scholar]
|
Fulltext Views
2,615
PDF downloads
1,176





