Translate this page into:
Does using a high sun protection factor sunscreen on face, along with physical photoprotection advice, in patients with melasma, change serum vitamin D concentration in Indian conditions? A pragmatic pretest-posttest study
2 Department of Community Medicine, Government Medical College, Kannauj, Uttar Pradesh, India
3 Department of Endocrinology and Metabolism, Institute of Medical Sciences, Banaras Hindu University, Varanasi, Uttar Pradesh, India
Correspondence Address:
Sanjay Singh
Department of Dermatology and Venereology, Institute of Medical Sciences, Banaras Hindu University, Varanasi, Uttar Pradesh
India
| How to cite this article: Singh S, Jha B, Tiwary NK, Agrawal NK. Does using a high sun protection factor sunscreen on face, along with physical photoprotection advice, in patients with melasma, change serum vitamin D concentration in Indian conditions? A pragmatic pretest-posttest study. Indian J Dermatol Venereol Leprol 2019;85:282-286 |
Abstract
Background: Use of sunscreens on the face is becoming popular, and patients with melasma are prescribed sunscreen for use on the face. Results of a few Western studies on the effect of sunscreen use on serum vitamin D concentration are not applicable to Indian conditions.
Aims: To examine the effect of use of a high sun protection factor (SPF 50+, PA++++) sunscreen on face in patients with melasma on serum concentration of 25-hydroxyvitamin D.
Methods: Forty-five Indian patients (Fitzpatrick skin types III and IV) with melasma were advised to use a sunscreen with SPF 50 + for 3 months, 43 (33 female, 10 male; age 32.9 ± 8 years) completed the study. Patients staying outdoor for <4 hours applied sunscreen once daily after bath. Patients staying outdoors for >4 hours reapplied sunscreen 4 hours after first application. Patients were provided a container to measure the amount of sunscreen for use, which was approximately equal to recommended thickness. Compliance was tested by weighing the used tubes and tubes in use during monthly visits. Serum concentration of 25-hydroxyvitamin D was tested before and after the study period.
Results: Amount of sunscreen advised (100.5 ± 29.2 ml) and the actual amount used (96.6 ± 27.9 ml) were similar (P = 0.53, t-test). The difference between serum concentrations of 25-hydroxyvitamin D at the baseline (19.20 ± 9.06 ng/ml) and at 3 months (18.91 ± 8.39 ng/ml) was not significant (P = 0.87, paired t-test, 95% confidence interval of difference −3.33 to 3.92). No correlation was found between the amount of sunscreen used and the percentage change in serum 25-hydroxyvitamin D concentration at 3 months (rho = 0.099, P = 0.528, Spearman's rank correlation).
Limitations: Longer duration of application and a larger sample size may detect minor differences in vitamin D concentration.
Conclusion: Using a high SPF sunscreen on the face, along with physical photoprotection advice, in patients with melasma for 3 months does not influence serum 25-hydroxyvitamin D concentration in Indian conditions.
Introduction
Use of sunscreens on the face is becoming popular among the general population. Sunscreens are also prescribed for the treatment of skin diseases caused or aggravated by sun exposure. Over the years, sunscreens with higher sun protection factor (SPF) have become available. Higher sun protection factor means that the use of such sunscreens will more efficiently stop the very light (ultraviolet B, UVB) which causes synthesis of vitamin D in the skin. This issue is serious and needs careful attention as evidence indicates inverse associations of circulating 25-hydroxyvitamin D with risk of death due to cardiovascular disease, cancer and other causes.[1]
The results of a few Western studies on the effect of sunscreen use on serum vitamin D concentration cannot be applied to Indian conditions, which are different. Indian studies on this problem are lacking. In the present study, we examined the effect of use of a high sun protection factor (SPF 50+, PA++++) sunscreen on the face in patients with melasma on the serum concentration of 25-hydroxyvitamin D.
Methods
Forty-five patients (Fitzpatrick skin type III and IV) with melasma attending the dermatology out-patient department of Sir Sunderlal Hospital, Banaras Hindu University, Varanasi, were included in the study. Ethics Committee approval was obtained. Pregnant women and patients who expressed an inability to come for regular follow-ups were excluded. Patients were not receiving treatment, including sunscreen, at the time of enrolment. Only some patients had used self-purchased sunscreen made by cosmetic companies on and off. Patients were not taking calcium and/or vitamin D supplements or any drugs prior to enrolment, nor did they do so during the study period. They were given no instruction to change their diet and continued taking the same diet as earlier. Informed consent was taken. Pretreatment blood samples were obtained. The patients were instructed to use a sunscreen with sun protection factor 50+ and PA++++ for a total of three months. They were advised to apply 1 ml of sunscreen evenly over the entire face every morning after bath. Patients who stayed outdoors for less than four hours applied the sunscreen once daily. Those with outdoor activities of more than four hours reapplied the sunscreen four hours after the first application. Patients were provided a measuring device with a capacity of 1 ml to ensure the feasibility and accuracy of the application. They were instructed not to share the sunscreen with others and not to use any other sunscreen than what was prescribed. They were also advised to use umbrella, stole over face or cap whenever possible and appropriate. Patients were asked not to change their clothing behavior and they did not use sunscreen on any other body part(s). No instructions were given to cover other body parts normally exposed to sunlight. The patients were also prescribed a demelanizing agent (kojic acid 2% gel) to apply only on the lesions of melasma at night.
Patients were called for follow-up every month (i.e., 4, 8 and 12 weeks). At the end of every month, they were asked to bring the used sunscreen tubes and the tubes in use. The weight of the sunscreen tubes was measured to assess compliance. At the end of three months, a second blood sample was taken to measure serum vitamin D concentration.
Determination of the quantity of the sunscreen to be used
A recent study from India showed that the size of the face of men and women vary, which might lead to the need for lesser quantity of sunscreen in females.[2] We calculated the area of ten randomly selected male patients and ten randomly selected female patients attending the dermatology outpatient department. Considering the shape of the face to be elliptical, the average area of face of the male patients was calculated to be 434 cm[2] and for female patients it was 389cm[2]. Thus, the amount of sunscreen to be applied with optimum thickness (2 mg/cm[2]) by male patients was approximately 0.9 ml, and for female patients it was approximately 0.8 ml per application. For the sake of feasibility, we decided to use a standard of 1 ml of sunscreen to be applied over the entire face for all patients.
Estimation of 25-hydroxyvitamin D
The quantitative estimation of vitamin D was done by enzyme-linked immunosorbent assay (ELISA) technique by EDI Total 25-OH vitamin D Kit (Epitope Diagnostics, Inc. San Diego, CA92121, USA), which utilizes competitive immunoassay technique.[3],[4] The assay measures total 25-hydroxyvitamin D (i.e., vitamin D2 and D3). The analytical sensitivity of this test is approximately 0.9557 ng/ml.
Results
Forty-five patients were enrolled in the study, of whom 43 (33 female, 10 male; age 32.9 ± 8 years) completed the study [Table - 1]. The amount of sunscreen advised (100.5 ± 29.2 ml) and the actual amount used (96.6 ± 27.9 ml) were not significantly different (P = 0.53, t-test). Difference between the serum concentrations of 25-hydroxyvitamin D at the baseline (19.20 ± 9.06 ng/ml) and at 3 months (18.91 ± 8.39 ng/ml) was not significant (P = 0.87, paired t-test, 95% confidence interval of difference −3.33 to 3.92) [Table - 2]. No correlation was found between the amount of sunscreen used and the percentage change in serum 25-hydroxyvitamin D concentration in 3 months (rho = 0.099, P = 0.528, Spearman's rank correlation).


Discussion
Sunscreens are prescribed to patients with skin diseases caused or aggravated by sunlight, besides being used by normal individuals mainly over the face to prevent signs of ageing. Whether use of sunscreens would lead to suppression of vitamin D synthesis is a serious issue and needs careful attention as evidence indicates inverse associations of circulating 25-hydroxyvitamin D with risks of death due to cardiovascular disease, cancer and other causes.[1] In the present study, we examined the effect of use of a high sun protection factor (SPF 50+, PA++++) sunscreen on face in patients with melasma on serum concentration of 25-hydroxyvitamin D. Forty-five Indian patients (Fitzpatrick skin types III and IV) with melasma were advised to use a sunscreen for a total of 3 months; 43 of them completed the study. Serum concentration of 25-hydroxyvitamin D was tested before and after the study period. Compliance of the patients was good as the amount of sunscreen advised and the actual amount used did not differ significantly. There was no significant difference between the serum concentrations of 25-hydroxyvitamin D at baseline and at 3 months. In addition, no correlation was found between the amount of sunscreen used and the percentage change in serum 25-hydroxyvitamin D concentration in 3 months.
There are a few Western studies which have addressed this issue [Table - 3].[5],[6],[7],[8],[9],[10],[11],[12] In three of the eight studies, artificial light exposure was used instead of the real-life sun exposure and the highest SPF used, among all studies, has been mentioned as more than 30 in the latest study published in 2012.[12] In none of the studies was it ascertained whether the patients used adequate amount of sunscreen, leaving one unable to decide what would happen if the correct amount of sunscreen is used. Although these studies provide important information, understanding the effect of sunscreen on vitamin D concentration in a pragmatic setting requires further studies. Moreover, the results of these studies are not applicable to the Indian population as the Indian conditions, including climatic conditions, are different.
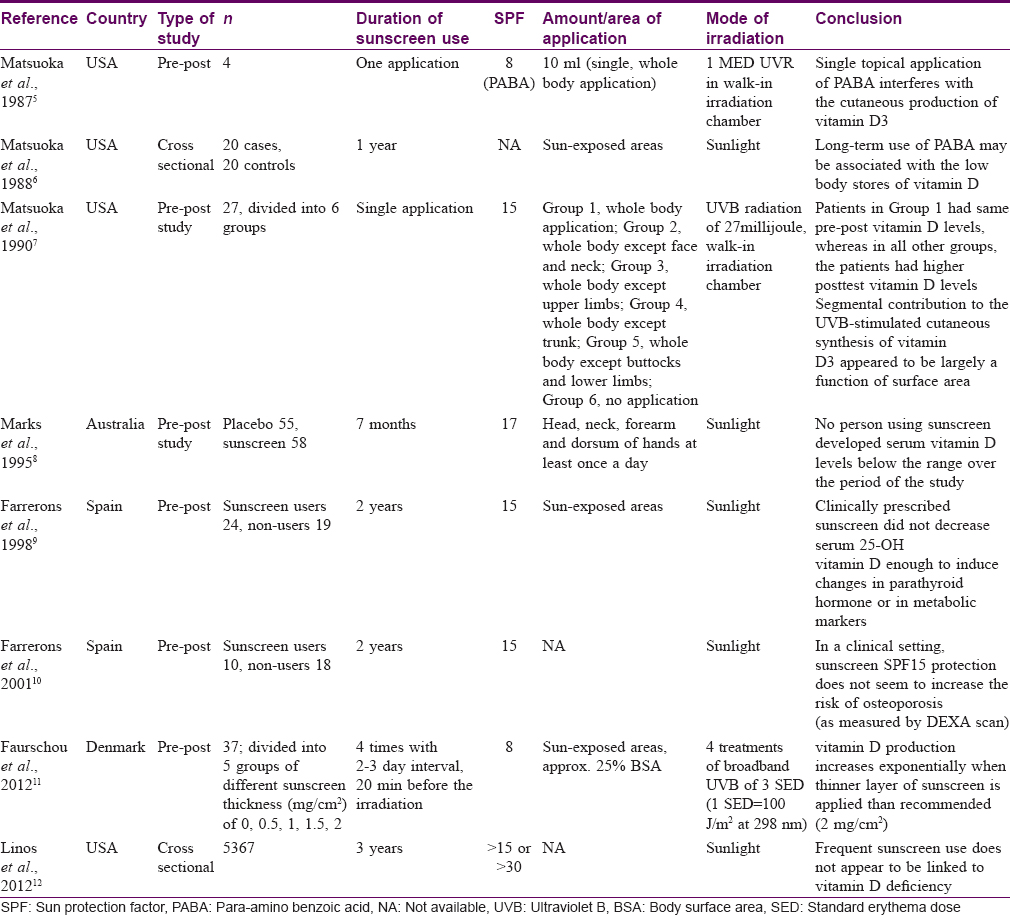
Some of the patients in the present study had used unprescribed sunscreen made by cosmetic companies on and off and not regularly. Furthermore, as the present study aimed to examine the effect of adequate sunscreen use on change in vitamin D concentration, the previous (inadequate) use of sunscreen was not expected to influence the results.
The study period was three months; therefore it is possible that sunscreen application for longer time may cause changes in serum vitamin D concentration. Also, we studied only 43 patients, so a study with a larger sample size may detect some changes which were not detected in the present study. In the absence of studies on the addressed problem, for the purpose of the present study, subjects were selected on the basis of convenience sampling to get an idea of the trend of results. Patients in the study were prescribed kojic acid 2% gel for application at night on the area affected by melasma; patients' compliance was not measured. It may be argued that some of the patients may have had a good response to kojic acid and the resulting decreased level of pigmentation could have led to higher vitamin D synthesis. However, the median area of involvement by melasma in the patients was 20.0% of the face (interquartile range, 12.5% to 25.0%) (data not shown). It appears unlikely that a decrease in pigmentation over such a small area would meaningfully affect the results. Furthermore, the patients were applying a sunscreen with SPF 50+ and PA++++, for which they had measured good compliance. Such a sunscreen would provide >98% UVB protection and excellent UVA protection. Taken together (i.e., suspected improvement in pigmentation on a median area of only 20% of face when applying sun protection factor 50+ and PA++++ sunscreen adequately), it appears highly unlikely that it would affect the study results. Patients in the study were advised to use an umbrella, stole or cap, whenever possible and appropriate, as would be done in a pragmatic setting. This advise was given only on their first visit, and patients' compliance with regard to this advice was not assessed. Further, in this pragmatic study, we wanted to know what would happen to the vitamin D concentration in a real-world setting, and it would have been unethical to not advise them about physical sun protection.
Conclusion
Results of the present study indicate that using a high sun protection factor sunscreen on the face, along with physical photoprotection advice, in patients with melasma for three months does not change serum 25-hydroxyvitamin D concentration in Indian conditions.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patients have given their consent for their images and other clinical information to be reported in the journal. The patients understand that name and initials will not be published and due efforts will be made to conceal identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Institute of Medical Sciences, Banaras Hindu University
Conflicts of interest
There are no conflicts of interest.
| 1. |
Chowdhury R, Kunutsor S, Vitezova A, Oliver-Williams C, Chowdhury S, Kiefte-de-Jong JC, et al. Vitamin D and risk of cause specific death: Systematic review and meta-analysis of observational cohort and randomised intervention studies. BMJ 2014;348:g1903.
[Google Scholar]
|
| 2. |
Srinivas CR, Lal S, Thirumoorthy M, Sundaram SV, Karthick PS. Sunscreen application: Not less, not more. Indian J Dermatol Venereol Leprol 2006;72:306-7.
[Google Scholar]
|
| 3. |
EDI Total 25-OH Vitamin D EIA Kit. Enzyme Immunoassay (EIA) for the Quantitative Measurement of total 25-OH D2/3 Levels in Serum or Plasma. Available from: http://www.epitopediagnostics.com/s/KT-815-Total-25-OH-Vitamin-D-EIA-V2.pdf. [Last accessed on 2017 Jul 27].
[Google Scholar]
|
| 4. |
Schmidt-Gayk H, Bouillon R, Roth HJ. Measurement of Vitamin D and its metabolites (calcidiol and calcitriol) and their clinical significance. Scand J Clin Lab Invest Suppl 1997;227:35-45.
[Google Scholar]
|
| 5. |
Matsuoka LY, Ide L, Wortsman J, MacLaughlin JA, Holick MF. Sunscreens suppress cutaneous Vitamin D3 synthesis. J Clin Endocrinol Metab 1987;64:1165-8.
[Google Scholar]
|
| 6. |
Matsuoka LY, Wortsman J, Hanifan N, Holick MF. Chronic sunscreen use decreases circulating concentrations of 25-hydroxyvitamin D. A preliminary study. Arch Dermatol 1988;124:1802-4.
[Google Scholar]
|
| 7. |
Matsuoka LY, Wortsman J, Hollis BW. Use of topical sunscreen for the evaluation of regional synthesis of Vitamin D3. J Am Acad Dermatol 1990;22:772-5.
[Google Scholar]
|
| 8. |
Marks R, Foley PA, Jolley D, Knight KR, Harrison J, Thompson SC. The effect of regular sunscreen use on Vitamin D levels in an Australian population. Results of a randomized controlled trial. Arch Dermatol 1995;131:415-21.
[Google Scholar]
|
| 9. |
Farrerons J, Barnadas M, Rodríguez J, Renau A, Yoldi B, López-Navidad A, et al. Clinically prescribed sunscreen (sun protection factor 15) does not decrease serum Vitamin D concentration sufficiently either to induce changes in parathyroid function or in metabolic markers. Br J Dermatol 1998;139:422-7.
[Google Scholar]
|
| 10. |
Farrerons J, Barnadas M, López-Navidad A, Renau A, Rodríguez J, Yoldi B, et al. Sunscreen and risk of osteoporosis in the elderly: A two-year follow-up. Dermatology 2001;202:27-30.
[Google Scholar]
|
| 11. |
Faurschou A, Beyer DM, Schmedes A, Bogh MK, Philipsen PA, Wulf HC. The relation between sunscreen layer thickness and Vitamin D production after ultraviolet B exposure: A randomized clinical trial. Br J Dermatol 2012;167:391-5.
[Google Scholar]
|
| 12. |
Linos E, Keiser E, Kanzler M, Sainani KL, Lee W, Vittinghoff E, et al. Sun protective behaviors and Vitamin D levels in the US population: NHANES 2003-2006. Cancer Causes Control 2012;23:133-40.
[Google Scholar]
|
Fulltext Views
3,524
PDF downloads
1,035





