Translate this page into:
Dramatic response of propranolol in hemangioma: Report of two cases
Correspondence Address:
Vikrant M Jadhav
Plot No. 29, "Maitreya", Near Sai-Smith Row House, Kalpataru Nagar, Wadala Shiwar, Nasik-11, Maharashtra
India
| How to cite this article: Jadhav VM, Tolat SN. Dramatic response of propranolol in hemangioma: Report of two cases. Indian J Dermatol Venereol Leprol 2010;76:691-694 |
Abstract
Hemangiomas are indolent birthmarks of vascular origin, which are known to appear soon after birth, proliferate for 8-18 months, and then slowly regress over the next 5-8 years, leaving behind normal or slightly blemished skin. In rare instances, hemangiomas may encroach upon and endanger vital structures with a mortality of up to 60%. Multiple therapeutic modalities are available for hemangiomas with variable results and associated with side effects. We report two cases of hemangioma, successfully treated with propranolol. Case 1 was a 5-month-old female child who presented with a giant segmental hemangioma since birth. She was unable to open her left eye over the past 7 days. Within 48 hours of administering full dose of oral propranolol (2 mg/kg/day), the lesion decreased considerably, and the patient was able to open her eye. Case 2 was a 1-year-old female child who presented with hemangioma over the danger area of face. Oral propranolol was given for a period of 6 months with monthly follow up. Both the cases showed dramatic response, with more than 80% regression, without any relapse after stopping the treatment.Introduction
Hemangiomas are common benign tumors of the vascular endothelium in infancy. The incidence ranges from 1% in neonates to 12% at 1 year, [1],[2],[3],[4] with predominance in premature newborns and chorionic villus sampling exposed infants. [5] Hemangiomas characteristically undergo postnatal growth for 8-12 months (proliferative phase), followed by regression over the next 1-5 years (involuting phase), with continuous improvement until 6-12 years (involuted phase). Large size or specific location or both may carry complications such as ulceration, vital structure compression, and visceral involvement. [6],[7],[8],[9] Large nodular superficial hemangiomas of face are at particular risk for disfiguring and scarring. [3],[6],[7] Even uneventful facial hemangiomas are known to cause parental anxiety. [6],[10]
Masterly inactivity continues to be the mainstay of therapy for majority of uncomplicated cases. Current medical therapeutic modalities for complicated hemangiomas include topical, intralesional and systemic steroids, recombinant interferons alpha-2a and 2b, vincristine, imiquimod, etc. Interventional therapies include cryotherapy, Argon, Nd-YAG, flashlamp-pumped pulse dye laser, embolization, sclerotherapy, surgery, and radiotherapy. [3],[4],[5],[6],[7],[8],[9],[11],[12],[13] The results with above therapies are variable and associated with side effects. We report two cases of hemangioma, treated successfully with propranolol.
Case Reports
Case 1
A 5-month-old female child presented with a giant segmental hemangioma since birth, involving left side of scalp, face, left arm, forearm and chest, with history of bleeding following minor trauma. She was unable to open her left eye over the past 7 days and had developed a large ulcer over lesion on scalp. Despite being on oral prednisolone 3 mg/kg/day since 2 months, the lesion was increasing rapidly in size. In view of development of ulcer over scalp, it was difficult to continue with systemic steroid, which would have predisposed her to secondary bacterial infection and delayed wound healing. On admission, a thorough physical examination was performed followed by electrocardiogram, blood sugar level, chest x-ray and local ultrasonography of the lesion using a high frequency probe. After ruling out asthma and atopy, the patient was started on oral propranolol 0.5 mg/kg/day with monitoring of heart rate and blood sugar level. After the patient tolerated the dose for 3 days without side effects, the dose was increased to 2 mg/kg/day given in three divided doses. Within 48 hours of administering full dose, the lesion decreased considerably. The patient was able to open her eye; color of swelling changed from red to purpuric and the consistency became soft [Figure - 1]. Oral prednisolone was rapidly tapered off over the next 2 weeks and propranolol was continued at a dose of 2 mg/kg/day for a period of 6 months. At the end of 6 months, reduction of 80% in size of hemangioma was observed and confirmed by local ultrasonography.
 |
| Figure 1 :Case 1: (a) pretreatment; (b) pretreatment; (c) after 48 hours of full dose of propranolol; (d) after 5 months; (e) after 5 months |
Case 2
A 1-year-old female child presented with a treatment naοve, gradually increasing hemangioma, measuring 2 cm Χ 2 cm over the danger area of face. The patient was investigated as in case 1, and the same dose regimen for propranolol was followed (2 mg/kg/day in three divided doses). Within 5 days of giving this dose, the swelling decreased [Figure - 2].
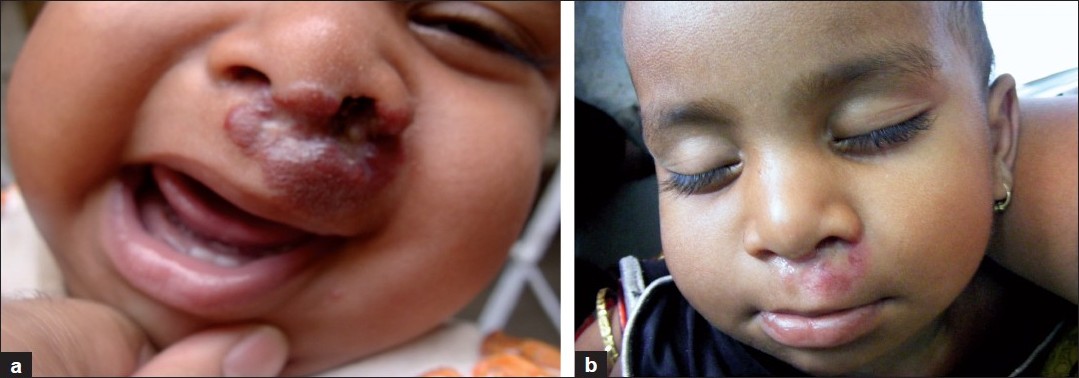 |
| Figure 2 :Case 2: (a) pretreatment; (b) after 5 months |
Both the cases were admitted and treated under observation for the first month. Subsequently, on discharge, oral propranolol was continued for 6 months with monthly follow up and monitoring of safety parameters. Thereafter, the dose was gradually tapered over 2 weeks and stopped. Regular monthly post-treatment follow up of both the patients did not show any rebound of hemangioma.
Discussion
Capillary hemangiomas are composed of a complex mixture of clonal endothelial cells, associated with pericytes, dendritic cells, and mast cells. [14] Regulators of hemangioma growth and involution are poorly understood. During the growth phase, two major proangiogenic factors are involved: basic fibroblast growth factor (bFGF) and vascular endothelial growth factor (VEGF). [14] Histological studies have shown active division of both endothelial and interstitial cells during this phase. During the involution phase, apoptosis has been shown. [14] Potential explanations for the therapeutic effect of propranolol (a non-selective beta-blocker) on infantile capillary hemangiomas include: vasoconstriction, which is immediately visible as a change in color, associated with a palpable softening of the hemangioma; decreased expression of VEGF and bFGF genes through the downregulation of the RAF-mitogen -activated protein kinase pathway [15] (which explains the progressive improvement of the hemangioma); and the triggering of apoptosis of capillary endothelial cells. [16]
The response of infantile hemangiomas to propranolol has been reported by Lιautι-Labrθze et al. [17] Investigators serendipitously discovered that propranolol effectively treated hemangiomas in two infants who received the drug for cardiac complications while on corticosteroid therapy. Index case was a 1-month-old infant with a rapidly growing segmental facial hemangioma, in whom propranolol was started for increased cardiac output. Seven days later, hemangioma was significantly smaller and no rebound occurred despite discontinuation of propranolol after 6 months. Subsequently, an additional nine infants with severe or disfiguring hemangiomas were treated successfully with propranolol.
The most common serious adverse effects with propranolol are bradycardia and hypotension. Propranolol may mask the clinical signs of early cardiac failure and diminish cardiac performance in infants with very large hemangiomas or miliary hemangiomatosis who are at risk for high-output cardiac compromise. [18] Propranolol may also blunt the clinical features of hypoglycemia. Sustained hypoglycemia in infancy has been associated with long-term neurologic sequelae. [19] To keep a check on side effects in our patients, regular blood sugar level monitoring and monthly electrocardiogram were done. None of the above mentioned side effects occurred in either of our patients and there was no rebound of the lesion. In case 1, there was obvious advantage of propranolol over steroids which were relatively contraindicated due to ulceration and subsequent risk of infection.
Conclusions
Amongst the armamentarium of available therapeutic options, propranolol appears to be a promising drug in the treatment of hemangioma. This molecule has been shown to have a rapid onset of clinical response with fewer side effects. However, comparative studies enrolling large series of patients are warranted to confirm the efficacy of propranolol before considering it as a possible first line drug in the treatment of infantile hemangiomas.
| 1. |
Jacobs AH, Walton RG. The incidence of birthmarks in the neonate. Pediatrics 1976;58:218-22.
[Google Scholar]
|
| 2. |
Metry DW, Hebert AA. Benign Cutaneous vascular tumors of infancy. When to worry, what to do. Arch Dermatol 2000;136:905-14.
[Google Scholar]
|
| 3. |
Frieden IJ, Eichenfield LF, Esterly NB, Geronemus R, Mallory SB. Guidelines of care for hemangiomas of infancy. J Am Acad Dermatol 1997;37:631-7
[Google Scholar]
|
| 4. |
Werner J, Dunne A, Lippert B, Folz B. Optimal treatment of vascular birthmarks. Am J Clin Dermatol 2003;4:745-56.
[Google Scholar]
|
| 5. |
Garzon MC, Frieden IJ. Hemangiomas: When to worry. Pediatr Ann 2000;29:58-67.
[Google Scholar]
|
| 6. |
Drolet BA, Esteryl NB, Frieden IJ. Hemangiomas in children. N Engl J Med 1999;341:173-81.
[Google Scholar]
|
| 7. |
Enjolras O, Gelbert F. Superficial hemangiomas: Association and management. Pediatr Dermatol 1997;14:173-9.
[Google Scholar]
|
| 8. |
Freiden IJ, Reese V, Cohen D. PHACE syndrome. Arch Dermatol 19996;132:307-11.
[Google Scholar]
|
| 9. |
Metry DW, Dowd CF, Barkovich AJ, Frieden IJ. The many faces of PHACE syndrome. J Pediatr 2001;139:117-23.
[Google Scholar]
|
| 10. |
Tanner JL, Dechert MP, Freiden IJ. Growing up with a facial hemangioma: parent and child coping and adaptation. Pediatrics 1998;101:446-83.
[Google Scholar]
|
| 11. |
Enjolras O, Riche MC, Merland JJ, Escande JP. Management of alarming hemangiomas in infancy: a review of 25 cases. Pediatrics 1990;85:491-8.
[Google Scholar]
|
| 12. |
Special symposium. Management of hemangiomas Pediatr Dermatol 1997;14:57-83.
[Google Scholar]
|
| 13. |
Poetke M, Philipp C, Berlin HP. Flashlamp-pumped pulse dye laser for hemangiomas in infancy. Arch Dermatol 2000;136:628-32.
[Google Scholar]
|
| 14. |
Frieden IJ, Haggstrom AN, Drolet BA, Mancini AJ, Friedlander SF, Boon L. Infantile hemangiomas: current knowledge, future directions: Proceedings of a research workshop on infantile hemangiomas, April 7-9, 2005, Bethesda, Maryland, USA. Pediatr Dermatol 2005;22:383-406.
[Google Scholar]
|
| 15. |
D'Angelo G, Lee H, Weiner RI. cAMP-dependent protein kinase inhibits the mitogenic action of vascular endothelial growth factor and fibroblast growth factor in capillary endothelial cells by blocking Raf activation. J Cell Biochem 1997;67:353-66.
[Google Scholar]
|
| 16. |
Sommers Smith SK, Smith DM. In vitro Beta blockade induces apoptosis in cultured capillary endothelial cells. Cell Dev Biol Anim 2002;38:298-304.
[Google Scholar]
|
| 17. |
Lιautι-Labrθze C, Dumas de la Roque E, Hubiche T, Boralevi F, Thambo J B, Taοeb A. Propranolol for severe hemangiomas of infancy. N Engl J Med 2008;358:2649-51.
[Google Scholar]
|
| 18. |
Gottschling S, Schneider G, Meyer S, Reinhard H, Dill-Mueller D, Graf N. Two infants with life-threatening diffuse neonatal hemangiomatosis treated with cyclophosphamide. Pediatr Blood Cancer 2006;46:239-42.
[Google Scholar]
|
| 19. |
Burns CM, Rutherford MA, Boardman JP, Cowan FM. Patterns of cerebral injury and neurodevelopmental outcomes after symptomatic neonatal hypoglycemia. Pediatrics 2008;122:65-74.
[Google Scholar]
|
Fulltext Views
2,432
PDF downloads
1,724





