Translate this page into:
Familial Bowen's disease
Correspondence Address:
Ping Fu
Department of Dermatology, Kunming Medical University, No. 374, Dianmian Road, Wuhua District, Kunming, Yunnan 650101
China
| How to cite this article: Fu P, Liu XH, Liu R, Suo GY. Familial Bowen's disease. Indian J Dermatol Venereol Leprol 2013;79:717-719 |
Sir,
Bowen′s disease has its highest incidence in the older age groups, while it rarely occurs in younger patients. We report a case of a pair of father and daughter suffering from Bowen′s disease. A genetic basis for Bowen′s disease should be considered and investigated.
A 21-year-old Chinese woman presented to our department in 2007 with papules and plaques on trunk and limbs for more than 10 years. The patient first presented with an asymptomatic plaque about the size of a coin at the age of 10. The number of lesions gradually increased before generalizing rapidly during her pregnancy in 2004. The patient denied a history of exposure to radiation and immunosuppressive therapy. The patient lived in Jianshui County in Yunnan, China, where arsenic pollution is not significantly present. The physical examination revealed brownish, scaling patches and plaques [Figure - 1]a. Cutaneous signs of arsenism, [1] including hyperkeratosis of the palms and soles and cutaneous hyperpigmentation with "raindrop" depigmentation, were absent. There was also no peripheral lymphadenopathy. Routine laboratory examinations were within normal limits. The biopsy of the lesions over the back and abdomen both revealed epidermal thickening with hyperkeratosis, dyskeratosis, loss of polarity, cellular derangement with atypia and many mitoses, along with variable degrees of acanthosis and chronic inflammatory infiltrates in the upper dermis [Figure - 1]b. The immunohistochemical stains for human papillomavirus (HPV) types 16 and 18 were negative [Figure - 1]c. Electron microscopy examination revealed intercellular inflammatory infiltrates in the epidermis with mild edema. The basement membrane was intact with the absence of viral particles [Figure - 1]d.
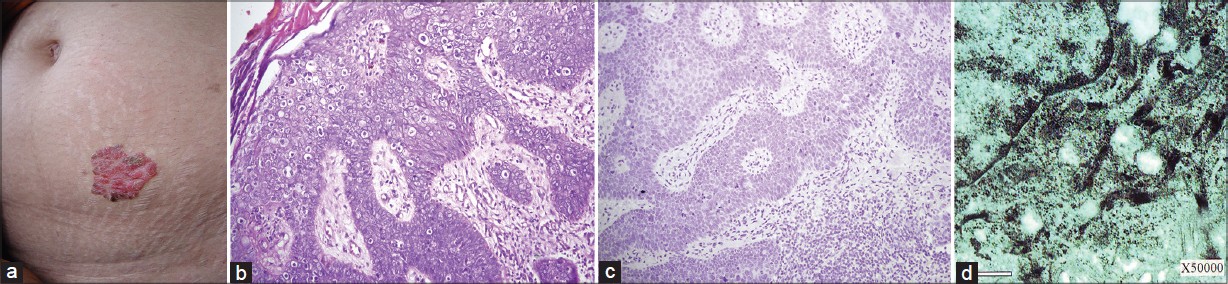 |
| Figure 1: (a) Dark-red, hyperkeratotic flat scaling papules coalescing to form a papillomatous hyperplastic plaque with a discrete border and overlying crust, (b) Cellular derangement with atypia and many mitoses, multinucleated oncocyte (H and E, ×200), (c) The immunohistochemical stains for human papillomavirus (HPV) types 16 and18 were negative (×200) , (d) No viral particles seen at ×50,000 magnification under electron microscopy |
The father of the patient (mentioned in the first case) complained of an asymptomatic salmon-red plaque on his left upper arm for 3 years. He is a farmer by occupation and both patients denied the pre-existence of freckles/tendency to sunburn. Physical examination revealed an erythematous, dome-shaped plaque with crusting on his left upper arm [Figure - 2]a. Laboratory examinations were normal. The biopsy of the lesions revealed the same results as the first case [Figure - 2]b. The immunohistochemical staining for HPV type 16 and 18 and electron microscopy examination also revealed the same results as the first case [Figure - 2]c.
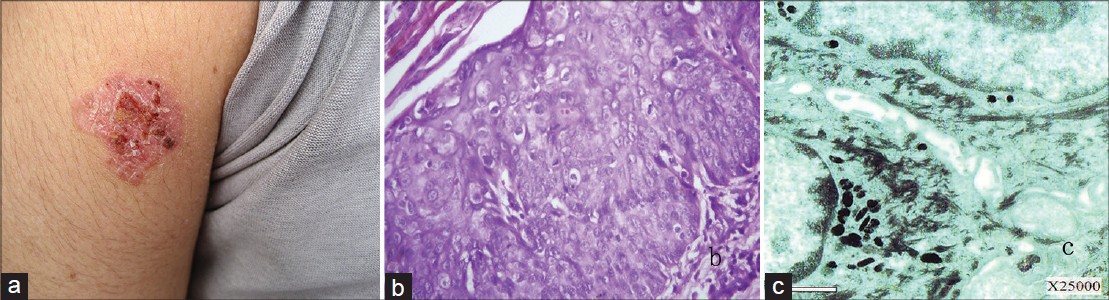 |
| Figure 2: (a) Salmon-red hyperkeratotic flat scaling papules coalescing to form a hyperplastic plaque with a discrete border and overlying crust that measures 2 cm by 2 cm in size, (b) Epidermal thickening with hyperkeratosis, dyskeratosis, papillary hyperplasia, loss of polarity, cellular derangement with atypia and many mitoses, along with chronic inflammatory infiltrates in the upper corium (H and E, ×400), (c) No viral particles were seen at ×25,000 magnification under electron microscopy |
The exact cause of Bowen′s disease remains unclear, although arsenic poisoning, sun exposure, and HPV infection are likely to be responsible for it. The incidence of Bowen′s disease in the Chinese population remains unclear. We report a father and daughter pairing with lesions occurring mainly on the trunk and extremities which were not sun exposed. The patients presented sequentially. The daughter presented earlier than her father. Till the time of reporting, no other family members had been found with the same lesions. Arsenic exposure has demonstrated an increased risk of cancer of the skin in humans. [2],[3] Both our patients denied a history of cryptic arsenic ingestion. More than 70% of all HPV-related anogenital cancers contain DNA from either HPV-16 or HPV-18. [4] We employed immunohistochemical methods to detect the presence of HPV-16 and HPV-18 DNA in the lesions, which turned out negative. Electron microscopy examination was also negative for viral particles in the lesions at ×40,000 magnification. We, therefore, conclude that there is no evidence to support an oncogenic viral cause for the occurrence in our two patients. It has been reported that the standardized incidence ratios for squamous cell carcinoma and Bowen′s disease increased to 1.95-4.30 times for individuals with affected siblings or parents. [5] The characteristics of our two patients include an early age of disease onset, and multiple and widely distributed lesions on the body. We therefore, postulate the possibility of a familial genetic susceptibility playing a part in the pathogenesis of the lesions in our reported patients. However, due to the fact that molecular and genetic studies were not performed, we could not completely exclude the possibility that this reported concurrence may be a coincidental finding. Future studies should explore hereditary, molecular, and genetic factors in the familial clustering of this disease.
| 1. |
Cöl M, Cöl C, Soran A, Sayli BS, Oztürk S. Arsenic-related Bowen's disease, palmar keratosis and skin cancer. Environ Health Perspect 1999;107: 687-9.
[Google Scholar]
|
| 2. |
Lien HC, Tsai TF, Lee YY, Hsiao CH. Merkel cell carcinoma and chronic arsenicism. J Am Acad Dermatol 1999;41:641-3.
[Google Scholar]
|
| 3. |
Schwartz RA. Arsenic and the skin. Int J Dermatol 1997;36:241-50.
[Google Scholar]
|
| 4. |
Cobb MW. Human papillomavirus infection. J Am Acad Dermatol 1990;22:547-66.
[Google Scholar]
|
| 5. |
Hussain SK, Sundquist J, Hemminki K. The effect of having an affected parent or sibling on invasive and in situ skin cancer risk in Sweden. J Invest Dermatol 2009;129:2142-7.
[Google Scholar]
|
Fulltext Views
1,870
PDF downloads
739





