Translate this page into:
Insect bite reactions
Correspondence Address:
Sanjay Singh
C-23, Swastik Towers, Lanka, Varanasi-221005, Uttar Pradesh
India
| How to cite this article: Singh S, Mann BK. Insect bite reactions. Indian J Dermatol Venereol Leprol 2013;79:151-164 |
Abstract
Insects are a class of living creatures within the arthropods. Insect bite reactions are commonly seen in clinical practice. The present review touches upon the medically important insects and their places in the classification, the sparse literature on the epidemiology of insect bites in India, and different variables influencing the susceptibility of an individual to insect bites. Clinical features of mosquito bites, hypersensitivity to mosquito bites Epstein-Barr virus NK (HMB-EBV-NK) disease, eruptive pseudoangiomatosis, Skeeter syndrome, papular pruritic eruption of HIV/AIDS, and clinical features produced by bed bugs, Mexican chicken bugs, assassin bugs, kissing bugs, fleas, black flies, Blandford flies, louse flies, tsetse flies, midges, and thrips are discussed. Brief account is presented of the immunogenic components of mosquito and bed bug saliva. Papular urticaria is discussed including its epidemiology, the 5 stages of skin reaction, the SCRATCH principle as an aid in diagnosis, and the recent evidence supporting participation of types I, III, and IV hypersensitivity reactions in its causation is summarized. Recent developments in the treatment of pediculosis capitis including spinosad 0.9% suspension, benzyl alcohol 5% lotion, dimethicone 4% lotion, isopropyl myristate 50% rinse, and other suffocants are discussed within the context of evidence derived from randomized controlled trials and key findings of a recent systematic review. We also touch upon a non-chemical treatment of head lice and the ineffectiveness of egg-loosening products. Knockdown resistance (kdr) as the genetic mechanism making the lice nerves insensitive to permethrin is discussed along with the surprising contrary clinical evidence from Europe about efficacy of permethrin in children with head lice carrying kdr-like gene. The review also presents a brief account of insects as vectors of diseases and ends with discussion of prevention of insect bites and some serious adverse effects of mosquito coil smoke.Introduction
Insects are a class of living creatures within the arthropods with a chitinous exoskeleton, three-part body, three pairs of jointed legs, compound eyes, and two antennae [Table - 1]. [1] Insects (Latin insectum, meaning "cut into sections") may be considered to "cut into" three sections, head, thorax, and abdomen. Bite is a wound produced by the mouth parts of an animal. Some animals have a special structure called sting through which they inflict wound and inject venom. All insects do not bite. Some non-biting insects are beetles, locusts, moths, and butterflies, although these may produce skin reactions by other means such as allergic reactions to their body parts, faeces, or body fluids. Bees, wasps, and ants also do not bite, but produce dermatological reactions by their stings.

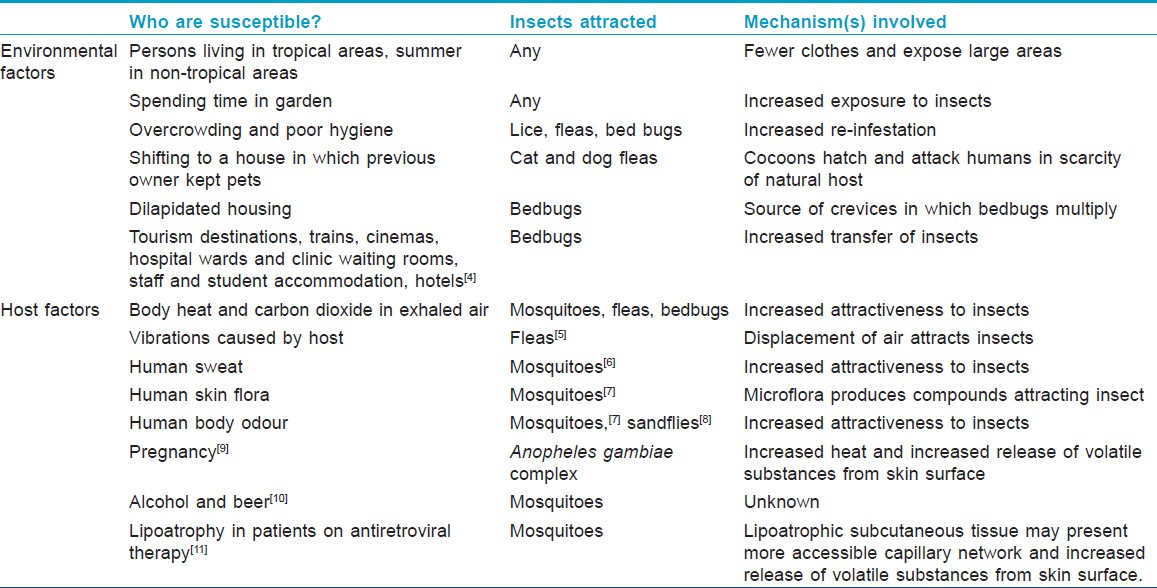
Epidemiology in India
Insect bite reactions are common, but information about their prevalence is limited. Children <14 years of age in dermatology outpatient clinic in Pondicherry had a prevalence of 5.3%. [2] Children <5 years of age attending skin outpatient clinic in Calcutta had 10.6% prevalence of papular urticaria, [3] with seasonal variation (rainy season 16.7%, summer 6.7%, winter 5.8%). Variables affecting susceptibility to insect bites have been described [Table - 2]. [4],[5],[6],[7],[8],[9],[10],[11]
Mosquito Bites
Mosquitoes belong to order Diptera, suborder Nematocera, and family Culicidae [Figure - 1]. Bite reactions due to insects of order Diptera are mostly due to allergens in saliva of insect and not due to toxin. Saliva of mosquitoes contains pharmacologically active compounds inhibiting body′s innate immune responses, causing anticoagulation, impaired platelet formation, vasodilation and anti-inflammatory activities. [12],[13] Allergens in mosquito whole body extract and saliva have been studied for developing diagnostic tests and immunotherapy for mosquito bite allergy. These approaches are used infrequently and mosquito whole body extracts are ineffective in down regulating specific immune responses to salivary allergens and may enhance sensitization. [14] Salivary gland surface proteins are major immunogenic components. [15] Species-specific and species-shared allergens of mosquitoes have been identified, which should facilitate production of specific recombinant allergens and improvement in diagnosis and specific immunotherapy. [16]
 |
| Figure 1: Aedes aegypti biting a human (source: http://commons. wikimedia.org/wiki/File:Aedes_aegypti_biting_human.jpg) |
Human sweat contains odorous organic compounds, produced by skin microflora, determining its attractiveness to mosquitoes. Variation in sweat composition causes differential attractiveness to mosquitoes within and between individuals. [6] Higher probability of mosquito bites is associated with increasing age in children, [17] male gender, [18] large body size, [19] pregnancy, [9] and alcohol ingestion. [10]
Common reaction pattern
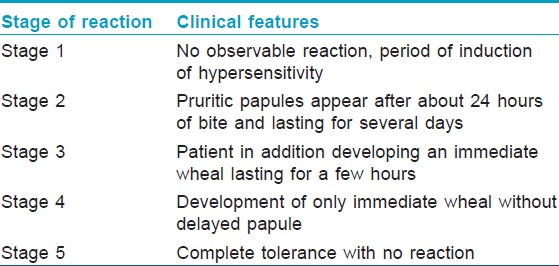
Mellanby [20] has described 5 stages of reaction to repeated mosquito bites [Table - 3].
Anaphylactic reaction
Severe IgE-mediated anaphylactic reactions rarely occurs, characterized by rapid onset of skin lesions which may be associated with respiratory compromise and may result in death. It is highly recommended that individuals at risk of anaphylaxis carry an epinephrine auto-injector whenever they are likely to encounter mosquitoes. [21],[22]
Hypersensitivity to Mosquito Bite Epstein-Barr Virus Natural Killer (HMB-EBV-NK) Disease
This disease is mainly reported from Japan, Korea, and China. Persons infected with EBV may develop a severe reaction to mosquito bites. In 1990, Tokura et al described a patient with HMB with 50% to 60% of peripheral blood mononuclear cells being NK cells. [23] Mean age of onset is 6.7 years without any gender predominance. [24] EBV infection immortalizes NK cells, which are activated by injection of some mosquito constituents, thereby exhibiting HMB as primary clinical manifestation. [24]
NK cell predominant mononuclear cells are infiltrated into bite sites. Typical clinical course includes three stages, initial exaggerated reaction to mosquito bites, deterioration of general systemic symptoms particularly in elderly, and the hemophagocytic syndrome (or malignant histiocytosis) with major life-threatening complications at the terminal stages. [24] A recurrent and prolonged activated state of NK cells may induce additional genetic damage leading to leukemias or lymphomas. [25] Some patients may already have leukemia or lymphoma at first episode of HMB. [24]
HMB-EBV-NK disease is included under "T/NK cell chronic active EBV disease" (CAEBV), which also includes hydroa vacciniforme (HV), HV-like lymphoma, and systemic EBV+T-cell lymphoproliferative disease of childhood. [26]
Eruptive Pseudoangiomatosis
Millet-sized erythema or small shiny red papules of angioma-like appearance with an anaemic halo occur on exposed parts resolving within a few days. [27],[28] The disease is possibly due to mosquito bites, as lesions have been elicited by Culex pipiens bites. [27] It overlaps with a disease known in Japan as erythema punctatum Higuchi, possibly caused by Culex pipiens pallens. [29] Histopathological findings are unremarkable with noticeable capillary dilation with plump endothelial cells protruding into lumen and perivascular predominantly lymphohistiocytic infiltrate without vasculitis. [29]
Skeeter Syndrome
Skeeter syndrome is a mosquito saliva-induced large local inflammatory reaction clinically resembling cellulites. [30] Lesions are red, itchy, warm swellings appearing within minutes of bite and itchy papules, ecchymotic, vesiculated, and bullous reactions appearing 2 to 6 hours afterwards and persisting for days or weeks with or without fever. [31] Severe reactions are treated with systemic prednisone. [30]
Papular Pruritic Eruption of HIV/AIDS
It is characterized by symmetrically distributed pruritic 2 to 5 mm papules on extremities and trunk sparing palms, soles, and digital web-spaces, [32] usually in advanced HIV disease, and is more prevalent in less developed regions of the world. [33] The condition, which tends to wax and wane and is resistant to oral antihistamine and topical corticosteroid therapy, [33] responds to HAART. [34] Increasing rash severity has been significantly associated with lower CD4 cell counts and higher absolute peripheral eosinophil counts. [33] Most of the patients had moderately dense to dense, superficial and deep, perivascular and interstitial infiltrates of lymphocytes and many eosinophils beneath an epidermis that was slightly hyperplastic. [33] History and histopathology suggest it to be an abnormal response to arthropod, mainly mosquito bites. [33]
Black Flies
Small blood crust with surrounding ecchymosis appears at bite site. Within a few hours, small, pruritic papules develop lasting several days. Severe reactions with marked oedema of limbs and constitutional upset occasionally occur and sometimes nodules and eczematous areas persist for several months. [35]
Two patients with Simulium dermatitis from North-Eastern region of India had intense itching, excoriations, scarring, and hyperpigmentation. Histopathology showed vesicles, dermal oedema, and perivascular infiltrates rich in eosinophils and lymphocytes. [36]
Blandford Flies
Bites produce skin swellings and sometimes fever or joint pain. [37]
Horse Flies
Bites produce painful lesions and rarely anaphylactic reaction with generalized itching, urticaria, paresthesia, and unconsciousness. [38]
Louse Flies
Deer ked (Lipoptena cervi L.), a haematophagous louse fly of deer, causes pruritic papules, usually in forests. [39] Lesions appear mostly on head and back, are resistant to treatment and persist for weeks to months. Direct immunofluorescence may show deposits of C3 in dermal vessel walls. [39] IgE, complement and cell-mediated mechanisms are involved. [39]
Tsetse Fly
Tsetse includes all species in genus Glossina, generally placed in their own family, Glossinidae. [40] Confined to Africa, tsetse flies are vectors of trypanosomiasis. Almost no information is available about lesions produced by bite. An allergen found in stinging insects (TAg5) was detected in tsetse fly saliva. [41] Anaphylactic reaction occurred following Glossina morsitans bites in a laboratory worker. [42]
Midges
Biting midges prefer certain human hosts determined by body odour, with non-attractive people producing natural "repellents". [43] In areas where midges are found, they are abundant at heights of 1 to 4 meters above ground [44] and hence bite taller people first. [45] Strong association between probability of bite and increasing height in men and body mass index in women has been shown. [45] This study found no association between bites and eating strongly flavoured foods (garlic, chilli, and onion), contrary to popular belief that garlic makes one less attractive to biting insects.
Bites may manifest as IgE-mediated urticaria or as presumably delayed-type reactions with papules, ulcers, or bullae persisting for weeks [46] IFN-γ, IL-6 and TNF-α are involved in delayed reactions. [47]
Bugs
Bugs are insects of the order Hemiptera with a common arrangement of sucking mouth parts; their hindwings are smaller than forewings. All bugs of family Cimicidae are flattened, oval and have no hind wings; the front wings are vestigial, hence they do not fly. Adult bedbugs are about 5 mm long and may be confused with booklice, carpet beetles, and small cockroaches [Figure - 2]. Cimicids are obligate haematophagous ectoparasites. [48] Bedbugs may survive up to a year without feeding. [49]
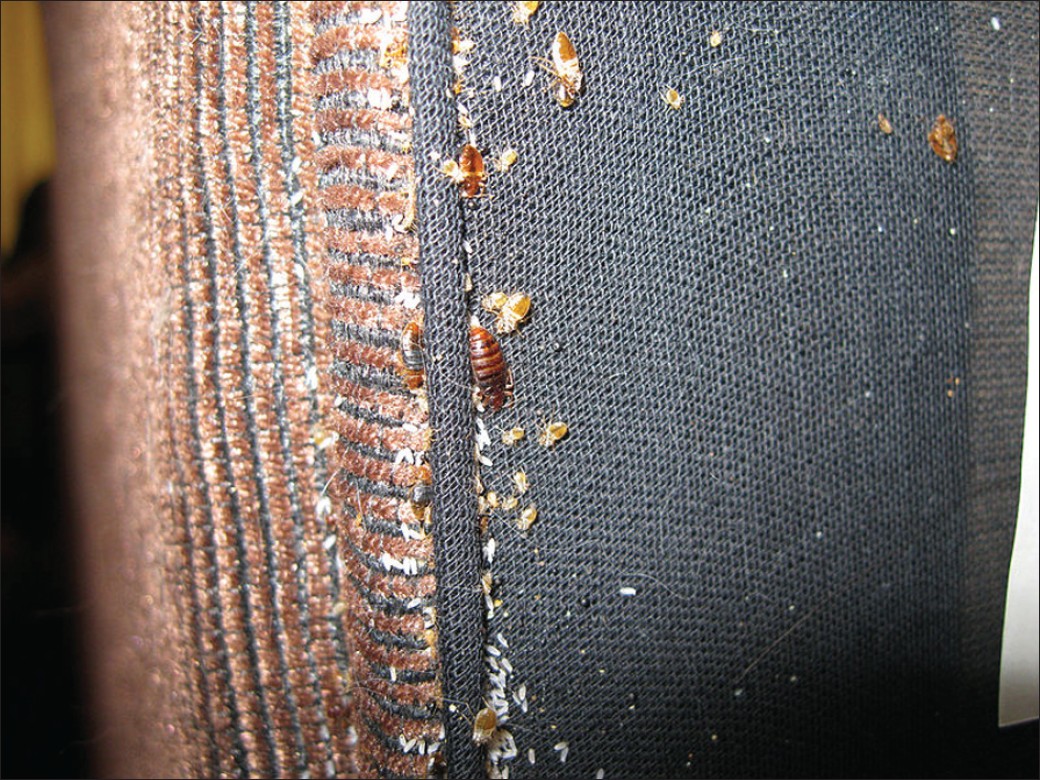 |
| Figure 2: Bedbugs and their eggs on a mattress (source: http:// commons.wikimedia.org/wiki/File:Bedbugs.jpg ) |
Bedbugs
Common species found in India are common bedbug and tropical bedbug. [1] Bedbug bites are known as cimicosis. On first exposure most individuals do not develop lesions. With further bites, most develop an obvious skin reaction and latency for previously reacting persons decreases substantially. [50] Few may not be sensitized even after repeated exposures as happened in a voluntarily-exposed researcher. [49] Three salivary proteins of bedbugs, a nitric oxide-liberating heme protein (nitrophorin), [51] a 17-kDa anticoagulant (Factor X), [52] and a 40-kDa apyrase-like nucleotide-binding enzyme, may be important immunologically. [53]
Bedbug infestation is facilitated by poor sanitation, overcrowding of residences and trade in second-hand furniture. [54] Infestation in high turnover locations (hotel rooms, school hostels) may spread the disease, [55] bedbugs being transferred with luggage to homes.
Bedbugs avoid light and feed at night. [49] Patient develops itch or a barely visible punctum. [49] This, if not abraded, resolves within a week. [56] Other lesions are pruritic, usually painless, erythematous macules, papules, nodules, urticarial wheals, and blisters. [49],[57] Bullous rashes occurring days later [58] may represent late-phase response of IgE-mediated hypersensitivity to salivary protein. [57] Common sites are arms, shoulders, and legs. Bites may produce anxiety, insomnia [59] or delusions in a cured patient. [60] Heavy infestation may cause significant blood loss and anemia in children. [57]
Rare systemic reactions include generalized urticaria, asthma, and anaphylaxis. [49] Bedbugs are suspected to transmit ≥40 human pathogens, [49] however, there is no proven case. [61]
Exposing suspected infested household materials to sunlight has little effect as bedbugs move away to dark crevices. [62]
Mexican chicken bugs
Bites of Mexican chicken bugs Haematosiphon inodorus (haematosiphoniasis) produce polymorphic lesions (wheals, papules, vesicles, pustules, and scabs). [63]
Assassin bugs
Assassin bug bites are defensive and extremely painful. [64]
Kissing bugs
Bites of kissing bugs (Triatoma sanguisuga) are painless, allowing them to feed undisturbed. Initial bites produce little reaction, with repeated exposure reactions ranging from pruritic papules with central punctum to haemorrhagic nodules and bullae may occur. [65] Patients have multiple clustered bites, especially on face, hence the name kissing bugs. [66] After additional bites, reaction may "accelerate" with local to diffuse urticaria and even erythema multiforme. [65] Rarely anaphylactic reaction may occur, usually as urticaria or angioedema. [67]
Often insects defecate while feeding, and parasite-laden faeces from infected bugs are source of Trypanosoma cruzi inocula, the causative organism of Chagas′ disease. [66] Other insects of subfamily Triatominae may also transmit T. cruzi.
Kissing bug bite on face may produce Romaña sign, consisting of unilateral swelling of eye at site of initial infection with T. cruzi with localized lymphadenopathy. Swelling persists for weeks. Acute stage of Chagas is followed by indeterminate stage lasting ≥10 years. [66] Although considered pathognomonic of T. cruzi infection, the sign may occur after a bite in absence of T. cruzi transmission. [68]
Fleas
Fleas are wingless haematophagous ectoparasites of birds and mammals, such as bats and humans [Figure - 3]. Animal fleas bite humans coming in contact with infested animals. Flea bites produce maculopapular or papular rashes and severe pruritus (pulicosis). [69],[70] Diagnosis is confirmed by examination of debris from pet′s beddings [71] or presence of flea faeces, eczematous lesions or alopecia on pet′s body. [1] Bites may produce psychological distress. [72] Treatment consists of eradication of flea infestation in the source animal.
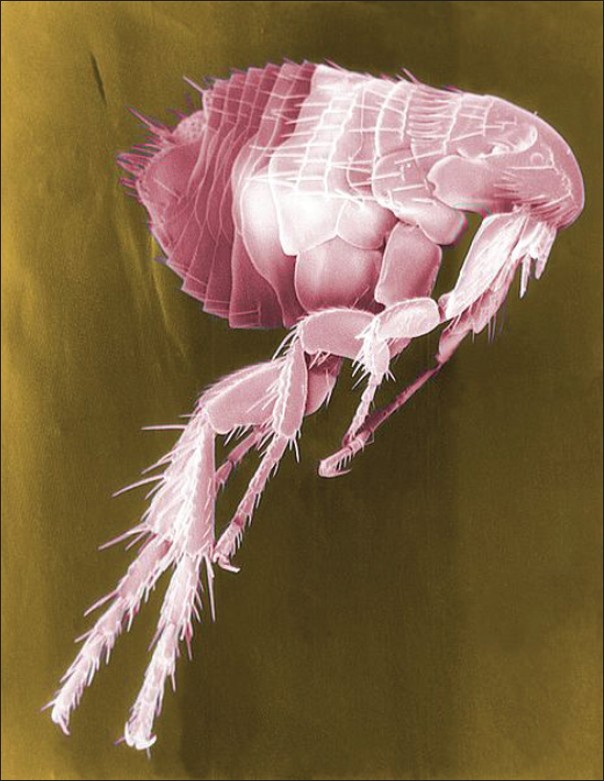 |
| Figure 3: Scanning electron micrograph of a flea with false colours (source: http://commons.wikimedia.org/wiki/File:Flea_Scanning_ Electron_Micrograph_False_Color.jpg ) |
Thrips
Bites produce tiny puncta and small, pink macules or papules. [73],[74]
Papular Urticaria
Epidemiology
Usually occurring in 2 to 10 years old children, papular urticaria occurs occasionally in adolescents and adults. [75] Sensitization takes time; hence it is not seen in newborns. [76] Higher prevalence in children may result from immune mechanisms and/or behaviours predisposing them to contact with insects. Most children outgrow the disease probably due to desensitization by repeated exposures. There are no racial or gender predilections. The disease is common in summer and spring seasons when opportunities of bites are more.
Insects
Papular urticaria results from hypersensitivity reaction to bites of mosquitoes, fleas, bed bugs, midges, biting flies, and other arthropods (mites and ticks). Some authors also consider cutaneous hypersensitivity reactions following stings and contact with body parts of arthropods (spiders, caterpillars) under papular urticaria. [77] Different insects may be responsible in different regions.
Pathogenesis
First exposure to the bites does not produce reaction. On subsequent exposures, host response to salivary or contactant proteins causes eruption. [77] Exact immune mechanism is unknown; evidence supports involvement of types I, III, and IV hypersensitivity.
Evidence supporting involvement of type I hypersensitivity includes time frame of immediate reaction, formation of wheals, elevated mosquito saliva-specific IgE in papular urticaria caused by mosquito bites, correlation of immediate wheals and flares with mosquito salivary gland-specific IgE levels, and high levels of saliva-specific IgE levels in systemic reactions. [30]
Mosquito saliva-specific IgG antibodies, mainly IgG4 and IgG1, are raised in individuals with positive mosquito bite tests and those with severe local reactions, but not systemic reactions, to mosquito bites. Levels of mosquito saliva-specific IgG correlate with sizes of both immediate and delayed skin reactions and with saliva-specific IgE levels. [30]
Immunoglobulin and complement deposits have been detected in skin suggesting that lesions may result from vasculitis. [78],[79]
Predominance of CD4+ T cells in all lesions (vesicles, wheals, and papules) after cutaneous injection of flea antigens in patients suggests delayed-type hypersensitivity. [76] Furthermore, presence of abundant eosinophils throughout dermis in all lesions, as earlier reported, [80] supported involvement of both immediate and delayed mechanisms. [76]
Peripheral blood mononuclear cells from patients with papular urticaria after polyclonal stimulation predominantly respond with Th2 cytokine (IL-4) production, indicating that patients underwent an atopic stage predisposing them to the disease. [81] Similar Th2 predominant response occurred on stimulation of dendritic cells. [82]
Type I hypersensitivity reaction may cause the wheal, while delayed papule may result from type IV hypersensitivity.
Appearance of new lesions following new bites may be accompanied by lesions appearing on previously sensitized sites, indicating role of circulating antigen(s) which stimulate cutaneous T cells in previously sensitized sites. [77]
Clinical features
Patients present with chronic or recurrent eruption of wheals and itchy papules [Figure - 4]. A mnemonic SCRATCH aids in diagnosis (S, symmetric distribution; C, crops of different coloration with erythema and pigmentary changes; R, pet (rover) unnecessary for diagnosis; A, age 2 to 10 years; T, target lesions and time taking weeks to years to resolve; C, confused paediatrician or parent; and H, household with single family member affected). [76]
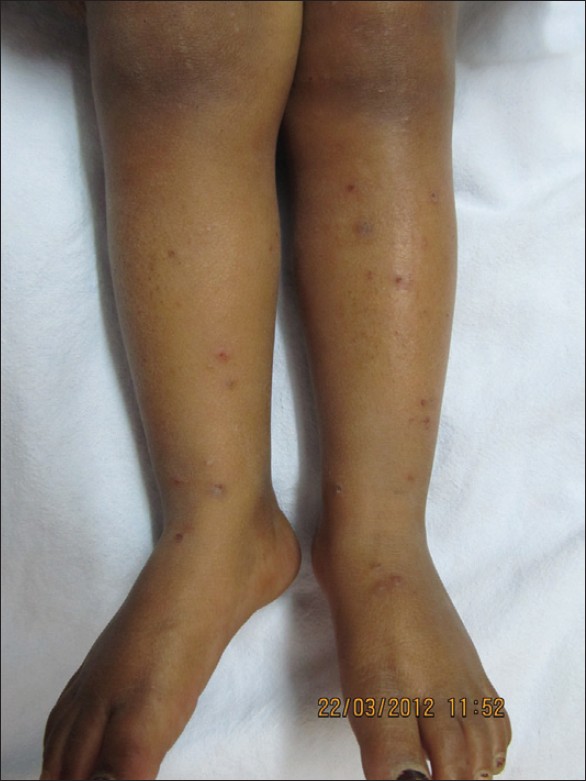 |
| Figure 4: Lesions of papular urticaria in a 3 year old girl |
Identifying culprit insect is difficult. Bullous papular urticaria, sometimes extensive, seen commonly in Iraq, may simulate serious bullous skin diseases. [83]
An eruption called harara (urticaria multiformis endemic), reported mainly in immigrants and children from Palestine after Phlebotomus bites, may be a form of papular urticaria. [84]
Histopathology
Mild acanthosis and spongiosis, exocytosis of lymphocytes, mild subepidermal edema, extravasation of erythrocytes, superficial and deep mixed inflammatory cell infiltrate of moderate density, and interstitial eosinophils are present. Depending on the predominant cellular infiltrate 4 subtypes (lymphocytic, eosinophilic, neutrophilic, and mixed) may be recognized. [80]
Treatment
Three "Ps" of management include protective clothing, pruritus control, and patience. [76] Treatment of insect bite reactions is symptomatic, with topical corticosteroids and antihistamines for mild reactions, short course of systemic corticosteroid for severe reactions, and management of anaphylaxis if it occurs.
Pediculosis
Head and body lice were designated Pediculus capitis and P. corporis but they are now known to belong to same species, P. humanus.[85] Under the body louse genome project, genome of the body louse, smallest among insects (108 Mb), has been sequenced, offering unique information and tools for understanding co-evolution among vectors, symbionts, and pathogens. [86]
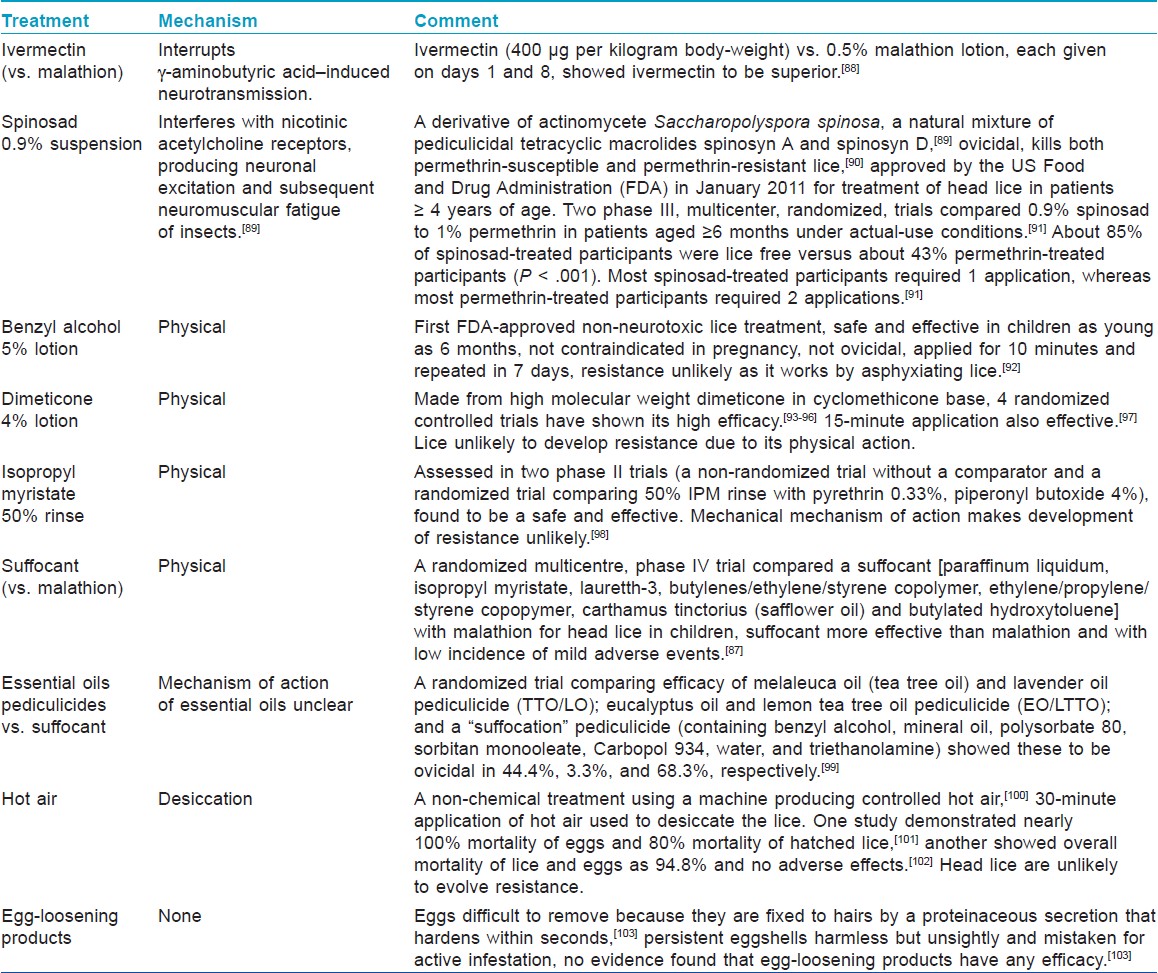
Treatment
Increasing resistance and adverse effects have raised concerns about head lice treatments, [87] encouraging search for new treatments [Table - 4]. [88],[89],[90],[91],[92],[93],[94],[95],[96],[97],[98],[99],[100],[101],[102],[103] No available pediculicide is 100% ovicidal, and resistance to lindane, pyrethrins, permethrin, and malathion has been reported. [100] Nits persist after the treatment and may be mistaken for active infestation [Figure - 5].
 |
| Figure 5: Numerous nits in a 15 year old girl with pediculosis |
A recent systematic review on pediculosis capitis including articles published till June 2010 concluded: malathion lotion may increase lice eradication compared with placebo, phenothrin, or permethrin; current best practice is to treat with two applications 7 days apart, and to check for cure at 14 days; studies comparing malathion or permethrin with wet combing have given conflicting results, possibly because of varying insecticide resistance; oral ivermectin may be more effective than malathion in people with previous failed treatment with insecticides; although tested in a clinical trial, oral ivermectin is not currently licensed for treating head lice, and generally its likely usefulness has been superseded by introduction of physically acting chemicals that are not affected by resistance and are considered safer; permethrin may be more effective than placebo or lindane; eradication may be increased by adding trimethoprim-sulfamethoxazole to topical permethrin, although this increases adverse effects; we don′t know whether combinations of insecticides are beneficial compared with single agents or other treatments; dimeticone may be more effective compared with malathion or permethrin; dimeticone and phenothrin have produced similar results, but this may be because of varying insecticide resistance and formulation of phenothrin used; we don′t know whether pyrethrum is beneficial compared with other insecticides; some herbal and essential oils may be beneficial compared with other treatments but this likely depends upon the compound(s) or extracts used; isopropyl myristate may be more effective than permethrin; benzyl alcohol may be more effective than placebo, however, we don′t know whether benzyl alcohol is more effective than insecticides or other treatments; and spinosad may be more effective at eliminating lice than permethrin. [104]
Permethrin resistance
Permethrin acts by its agonist action at voltage-gated sodium channels (VGSC) in nervous systems of insects. Selective point mutations in α-subunit gene of VGSC result in nerve insensitivity, resulting in resistance known as knockdown resistance (kdr). [105],[106] For efficient monitoring of head lice resistance in field based on kdr genotype molecular tools (quantitative sequencing, real-time PCR amplification of specific allele, serial invasive signal amplification reaction) have been developed. [105]
Surprisingly, a recent report from Germany has questioned that kdr gene is responsible for treatment failure. [107] Kdr-like gene was detected in up to 95% of head lice in Europe, which contrasts with reported low rates of treatment failure with permethrin. Treatments with 0.5% permethrin and 0.3% pyrethrin were effective in 93% and 74% of children, respectively, whose head lice carried kdr-like gene. Authors concluded that in Pediculus capitis populations examined, the kdr-like gene did not correlate with failure of permethrin or pyrethrin treatment and that further studies are necessary to identify all possible contributors to pyrethroid resistance, including attributes of head louse and host factors. [107] It may be possible that permethrin is acting by some hitherto unknown mechanism or its vehicle is acting as a suffocant.
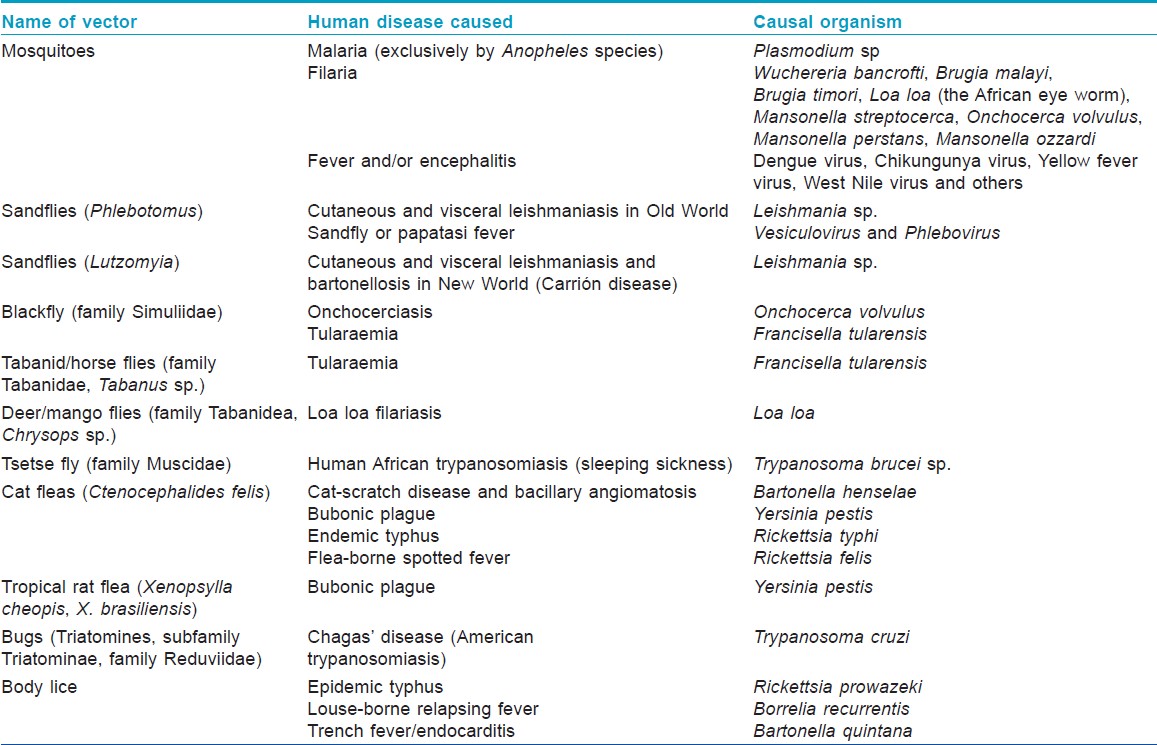
Insects as Vectors of Diseases [Table - 5]
Prevention of insect bites
This is achievable by protective clothing, insecticide sprays, residential insecticides, repellents, or by physical means using nets which may be insecticide-treated. Major classes of insecticides used to control mosquitoes are pyrethroid, organophosphate, carbamate and dichloro-diphenyl-trichloroethane (DDT). [108] Repellents are substances applied to skin preventing insects from biting. Most efficient repellents are diethyl-m-toluamide (DEET) and dimethylphthalate (DMP), [109] others being N, N-diethylphenylacetamide (DEPA), [110] para-Menthane-3, 8-diol (PMD), [111] picardin (icaridin, KB 3023), [112] and IR3535. [113] Neem oil has been found to have larvicidal activity in field conditions, [114] and has been shown to possess weak mosquito repellent activity. [115]
Rational repellent prescription for a child must take into account: age, active substance concentration, topical substance tolerance, nature and surface of the skin, number of daily applications, and the length of use in a benefit-risk ratio assessment. [113] The 4 repellents currently recommended by the WHO for their long lasting efficacy and tolerance are PMD, DEET, icaridin, and IR3535. [113] Minimum effective concentration of each of these agents for 3 hours efficacy against most arthropods is 20%. [113] Although the toxicity of topical repellent used sub-chronically and chronically is not well studied in paediatric age groups, current French recommendations are as follows: once daily use of topical repellent in infants above 6 months, 2 applications daily from ages 1 to 12 years, and 3 applications daily after 12 years. [113]
Synthetic pyrethroids are now commonly used as they cause less ecological problems and are available as sprays, vaporizing mats, mosquito coils or in combination with physical means such as bed-nets. [109] Although ineffective against mosquitoes, systemic vitamin B1, acoustic devices and so-called electrocuting light traps are still sold and used. [109]
Pyrethroids resistance among insects is increasing, necessitating study of its implications for insect bites and insect-borne diseases. [116],[117]
Long-term exposure to mosquito coil smoke (MCS) might induce asthma and persistent wheeze in children. [118] Mosquito coils containing pyrethroid insecticides, particularly d-allethrin, may contain octachlorodipropyl ether (S-2, S-421). Their use likely exposes individuals to an extremely potent lung carcinogen bis(chloromethyl)ether (BCME) formed from combustion of coils. [119] Prolonged exposures that recur in homes must be studied. Use of mosquito coils containing S-2 is illegal in US. [119] Case control studies have linked exposure to MCS to lung cancer [120] and nasopharyngeal carcinoma. [121] It may be prudent to instead use physical means such as bed-nets and window-nets for prevention of insect bites.
Topical ivermectin and 1,2-octanediol for head lice
Two studies, reporting 3 randomized controlled trials, have shown about 74% cure rate with a single 10-minute application of 0.5% ivermectin lotion. [122],[123] Another randomized controlled trial showed that two 8-hour 7 days apart applications of a physically active surfactant, 5% 1,2-octanediol lotion, cured around 80% patients with superiority over 0.5% malathion liquid. [124] Octanediol is the first surface active chemical to be tested for head lice. [124]
Acknowledgment
Grateful acknowledgment is made of Wikimedia Commons ( http://commons.wikimedia.org/wiki/Main_Page ), an online database of freely usable media files, for [Figure - 1], [Figure - 2] and [Figure - 3]. We also acknowledge kindness of United States Department of Agriculture, Charlysays, and Centers for Disease Control and Prevention (Janice Haney Carr) for contributing [Figure - 1], [Figure - 2] and [Figure - 3] respectively to Wikimedia Commons.
| 1. |
Burns DA. Diseases caused by arthropods and other noxious animals. In: Burns T, Breathnach S, Cox N, Griffiths C, editors. Rook′s textbook of dermatology. 8 th ed. Chichester: Wiley-Blackwell; 2010. p. 38.1-38.61.
[Google Scholar]
|
| 2. |
Karthikeyan K, Thappa DM, Jeevankumar B. Pattern of pediatric dermatoses in a referral center in South India. Indian Pediatr 2004; 41:373-7.
[Google Scholar]
|
| 3. |
Banerjee S, Gangopadhyay DN, Jana S, Chanda M. Seasonal Variation in pediatric dermatosis. Indian J Dermatol 2010;55:44-6.
[Google Scholar]
|
| 4. |
Doggett SL, Russell R. Bed bugs - what the GP needs to know. Aust Fam Physician 2009;38:880-4.
[Google Scholar]
|
| 5. |
Marshall AG. The ecology of ectoparasitic insects. London: Academic Press, 1981.
[Google Scholar]
|
| 6. |
Smallegange RC, Verhulst NO, Takken W. Sweaty skin: an invitation to bite? Trends Parasitol 2011;27:143-8.
[Google Scholar]
|
| 7. |
Keystone JS. Of bites and body odour. Lancet 1996;347:1423.
[Google Scholar]
|
| 8. |
Hamilton JG, Ramsoondar TM. Attraction of Lutzomyia longipalpis to human skin odours. Med Vet Entomol 1994;8:375-80.
[Google Scholar]
|
| 9. |
Lindsay S, Ansell J, Selman C, Cox V, Hamilton K, Walraven G. Effect of pregnancy on exposure to malaria mosquitoes. Lancet 2000;355:1972.
[Google Scholar]
|
| 10. |
Shirai O, Tsuda T, Kitagawa S, Naitoh K, Seki T, Kamimura K, et al. Alcohol ingestion stimulates mosquito attraction. J Am Mosq Control Assoc 2002;18:91-6.
[Google Scholar]
|
| 11. |
Greub G, Fellay J, Telenti A. HIV lipoatrophy and mosquito bites. Clin Infect Dis 2002;34:288-9.
[Google Scholar]
|
| 12. |
James AA, Rossignol PA. Mosquito salivary glands: parasitological and molecular aspects. Parasitol Today 1991;7:267-71.
[Google Scholar]
|
| 13. |
Ribeiro JM. Vector salivation and parasite transmission. Mem Inst Oswaldo Cruz 1987;82 Suppl 3:1-3.
[Google Scholar]
|
| 14. |
Peng Z, Simona FE. Comparison of proteins, IgE, and IgG binding antigens, and skin reactivity in commercial and laboratory made mosquito extracts. Ann Allergy Asthma Immunol 1996;77:371-6.
[Google Scholar]
|
| 15. |
King JG, Vernick KD, Hillyer JF. Members of the salivary gland surface protein (SGS) family are major immunogenic components of mosquito saliva. J Biol Chem 2011;286:40824-34.
[Google Scholar]
|
| 16. |
Wongkamchai S, Khongtak P, Leemingsawat S, Komalamisra N, Junsong N, Kulthanan K, et al. Comparative identification of protein profiles and major allergens of saliva, salivary gland and whole body extracts of mosquito species in Thailand. Asian Pac J Allergy Immunol 2010;28:162-9.
[Google Scholar]
|
| 17. |
Michael E, Ramaiah KD, Hoti SL, Barker G, Paul MR, Yuvaraj J, et al. Quantifying mosquito biting patterns on humans by DNA fingerprinting of bloodmeals. Am J Trop Med Hyg 2001;65:722-8.
[Google Scholar]
|
| 18. |
Carnevale P, Frézil JL, Bosseno MF, Le Pont F, Lancien J. The aggressiveness of Anopheles gambiae A in relation to the age and sex of the human subjects. Bull World Health Organ 1978;56:147-54.
[Google Scholar]
|
| 19. |
Port GR, Boreham PFL, Bryan JH. The relationship of host size to feeding by mosquitoes of the Anopheles gambiae Giles Complex (Diptera, Culicidae). Bull Entomol Res 1980;70:133-44.
[Google Scholar]
|
| 20. |
Mellanby K. Man′s reaction to mosquito bites. Nature 1946;158:554.
[Google Scholar]
|
| 21. |
Simons F, Peng Z. Mosquito allergy. In: Levine M, Lockey R, editors. American Academy of Allergy, Asthma and Immunology monograph on insect allergy. 4 th ed. Milwaukee: American Academy of Allergy, Asthma and Immunology; 2003. p. 175-203.
[Google Scholar]
|
| 22. |
Karppinen A, Brummer-Korvenkontio H, Petman L, Kautiainen H, Hervé JP, Reunala T. Levocetirizine for treatment of immediate and delayed mosquito bite reactions. Acta Derm Venereol 2006;86:329-31.
[Google Scholar]
|
| 23. |
Tokura Y, Tamura Y, Takigawa M, Koide M, Satoh T, Sakamoto T, et al. Severe hypersensitivity to mosquito bites associated with natural killer cell lymphocytosis. Arch Dermatol 1990;126:362-8.
[Google Scholar]
|
| 24. |
Tokura Y, Ishihara S, Tagawa S, Seo N, Ohshima K, Takigawa M. Hypersensitivity to mosquito bites as the primary clinical manifestation of a juvenile type of Epstein-Barr virus associated natural killer cell leukemia/lymphoma. J Am Acad Dermatol 2001;45:569-78.
[Google Scholar]
|
| 25. |
Tsuge I, Morishima T, Morita M, Kimura H, Kuzushima K, Matsuoka H. Characterization of Epstein-Barr virus infected natural killer cell proliferation in patients with severe mosquito allergy: Establishment of an IL-2-dependent NK-like cell line. Clin Exp Immunol 1999;115:385-92.
[Google Scholar]
|
| 26. |
Cohen JI, Kimura H, Nakamura S, Ko YH, Jaffe ES. Epstein- Barr virus-associated lymphoproliferative disease in non-immunocompromised hosts: A status report and summary of an international meeting, 8-9 September 2008. Ann Oncol 2009;20:1472-82.
[Google Scholar]
|
| 27. |
Oka K, Ohtaki N, Kasai S, Takayama K, Yokozeki H. Two cases of eruptive pseudoangiomatosis induced by mosquito bites. J Dermatol 2012;39:301-5.
[Google Scholar]
|
| 28. |
Romero A, Martín L, Hernández-Núñez A, Arias D, Castaño E, Borbujo J. Eruptive pseudoangiomatosis. Actas Dermosifiliogr 2007;98:138-9.
[Google Scholar]
|
| 29. |
Pérez-Barrio S, Gardeazábal J, Acebo E, Martínez de Lagrán Z, Díaz-Pérez JL. Eruptive pseudoangiomatosis: study of 7 cases. Actas Dermosifiliogr 2007;98:178-82.
[Google Scholar]
|
| 30. |
Peng Z, Simons FE. Mosquito allergy: Immune mechanisms and recombinant salivary allergens. Int Arch Allergy Immunol 2004;133:198-209.
[Google Scholar]
|
| 31. |
Peng Z, Simons FE. Advances in mosquito allergy. Curr Opin Allergy Clin Immunol 2007;7:350-4.
[Google Scholar]
|
| 32. |
Bason MM, Berger TG, Nesbitt LT Jr. Pruritic papular eruption of HIV-disease. Int J Dermatol 1993;32:784-9.
[Google Scholar]
|
| 33. |
Resneck JS Jr, Van Beek M, Furmanski L, Oyugi J, LeBoit PE, Katabira E, et al. Etiology of pruritic papular eruption with HIV infection in Uganda. JAMA 2004;292:2614-21.
[Google Scholar]
|
| 34. |
Castelnuovo B, Byakwaga H, Menten J, Schaefer P, Kamya M, Colebunders R. Can response of a pruritic papular eruption to antiretroviral therapy be used as a clinical parameter to monitor virological outcome? AIDS 2008;22:269-73.
[Google Scholar]
|
| 35. |
Gudgel EF, Grauer FH. Acute and chronic reactions to black fly bites (Simulium fly). Arch Dermatol Syphilol 1954;70:609-15.
[Google Scholar]
|
| 36. |
Borah S, Goswami S, Agarwal M, Rahman I, Deka M, Chattopadhyay P, et al. Clinical and histopathological study of Simulium (blackfly) dermatitis from North-Eastern India-A report. Int J Dermatol 2012;51:63-6.
[Google Scholar]
|
| 37. |
Inskip H, Campbell L, Godfrey K, Coggon D. A survey of the prevalence of biting by the Blandford fly during 1993. Br J Dermatol 1996;134:696-9.
[Google Scholar]
|
| 38. |
Quercia O, Emiliani F, Foschi FG, Stefanini GF. A case of anaphylaxis: Horse-fly or hymenoptera sting? Eur Ann Allergy Clin Immunol 2009;41:152-4.
[Google Scholar]
|
| 39. |
Rantanen T, Reunala T, Vuojolahti P, Hackman W. Persistent pruritic papules from deer ked bites. Acta Derm Venereol 1982;62:307-11.
[Google Scholar]
|
| 40. |
Anonymous. Tsetse fly. Available from: http://en.wikipedia.org/wiki/Tse_tse_fly. [ Last Accessed 2011 Feb 17].
[Google Scholar]
|
| 41. |
Caljon G, Broos K, De Goeyse I, De Ridder K, Sternberg JM, Coosemans M, et al. Identification of a functional Antigen5-related allergen in the saliva of a blood feeding insect, the tsetse fly. Insect Biochem Mol Biol 2009;39:332-41.
[Google Scholar]
|
| 42. |
Stevens WJ, Van den Abbeele J, Bridts CH. Anaphylactic reaction after bites by Glossina morsitans (tsetse fly) in a laboratory worker. J Allergy Clin Immunol 1996;98:700-1.
[Google Scholar]
|
| 43. |
Logan JG, Seal NJ, Cook JI, Stanczyk NM, Birkett MA, Clark SJ, et al. Identification of human-derived volatile chemicals that interfere with attraction of the Scottish biting midge and their potential use as repellents. J Med Entomol 2009;46:208-19.
[Google Scholar]
|
| 44. |
Carpenter S, Mordue W, Mordue J. Selection of resting areas by emerging Culicoides impunctatus (Diptera: Ceratopogonidae) on downy birch (Betula pubescens). Int J Pest Manag 2008;54:39-42.
[Google Scholar]
|
| 45. |
Logan JG, Cook JI, Stanczyk NM, Weeks EN, Welham SJ, Mordue Luntz AJ. To bite or not to bite! A questionnaire-based survey assessing why some people are bitten more than others by midges. BMC Public Health 2010;10:275.
[Google Scholar]
|
| 46. |
Steffen C. Clinical and histopathologic correlation of midge bites. Arch Dermatol 1981;117:785-7.
[Google Scholar]
|
| 47. |
Chen YH, Lee MF, Tsai JJ, Wu HJ, Hwang GY. Specific IgE and IgG responses and cytokine profile in subjects with allergic reactions to biting midge Forcipomyia taiwana. Int Arch Allergy Immunol 2009;150:66-74.
[Google Scholar]
|
| 48. |
Anonymous. Cimicidae. Available from: http://en.wikipedia.org/wiki/Cimicidae. [Last Accessed 2011 Feb 17].
[Google Scholar]
|
| 49. |
Goddard J, de Shazo R. Multiple feeding by the common bed bug, Cimex lectularius, without sensitization. Midsouth Entomol 2009;2:90-2.
[Google Scholar]
|
| 50. |
Reinhardt K, Kempke D, Naylor R, Siva-Jothy MT. Sensitivity to bites by the bedbug, Cimex lectularius. Med Vet Ent 2009;23:163-6.
[Google Scholar]
|
| 51. |
Valenzuela JG, Ribeiro JM. Purification and cloning of the salivary nitrophorin from the hemipteran Cimex lectularius. J Exp Biol 1998;201: 2659-64.
[Google Scholar]
|
| 52. |
Valenzuela JG, Guimaraes JA, Ribeiro JM. A novel inhibitor of factor X activation from the salivary glands of the bed bug Cimex lectularius. Exp Parasitol 1996;83:184-90.
[Google Scholar]
|
| 53. |
Valenzuela JG, Charlab R, Galperin MY, Ribeiro JM. Purification, cloning, and expression of an apyrase from the bed bug Cimex lectularius: A new type of nucleotide-binding enzyme. J Biol Chem 1998;273:30583-90.
[Google Scholar]
|
| 54. |
Omudu EA, Kuse CN. Bedbug infestation and its control practices in Gbajimba: A rural settlement in Benue state, Nigeria. J Vector Borne Dis 2010;47:222-7.
[Google Scholar]
|
| 55. |
Boase CJ. Bedbugs: Reclaiming our cities. Biologists 2004;51:9-12.
[Google Scholar]
|
| 56. |
Masetti M, Bruschi F. Bedbug infestations recorded in Central Italy. Parasitol Int 2007;56:81-3.
[Google Scholar]
|
| 57. |
Leverkus M, Jochim RC, Schad S, Brocker EB, Anderson JF, Valenzuela JG. Bullous allergic hypersensitivity to bedbug bites mediated by IgE against salivary nitrophorin. J Invest Dermatol 2006;126:91-6.
[Google Scholar]
|
| 58. |
Cooper DL. Can bedbug bites cause bullous erythema? JAMA 1948;138:1206.
[Google Scholar]
|
| 59. |
Doggett SL, Geary MJ, Russell RC. The resurgence of bedbug in Australia: With notes on their ecology and control. Environ Health 2004;4:30-7.
[Google Scholar]
|
| 60. |
Vandam J. Sleep tight, and don′t let...Oh, just forget about it. New York Times 2003, Nov 2.
[Google Scholar]
|
| 61. |
Goddard J. Do bed bugs carry human diseases? A controversy. Pest Control Technol 2003;31:38-40.
[Google Scholar]
|
| 62. |
Paul J, Bates J. Is infestation with common bedbug increasing? BMJ 2000;320:1141.
[Google Scholar]
|
| 63. |
Andrade RN. Haematosiphoniasis. In: Simons RD, editors. Handbook of Tropical Dermatology. Vol. 2. Amsterdam: Elsevier; 1953. p. 905-7.
[Google Scholar]
|
| 64. |
Smith FD, Miller NC, Camazzo SJ, Eaton WB. Insect bite by Argilus cristatus, a North American reduviid. AMA Arch Dermatol 1958;77:324-30.
[Google Scholar]
|
| 65. |
Shields TS, Walsh EN. ′Kissing bug′ bite. AMA Arch Dermatol 1956;74:14-21.
[Google Scholar]
|
| 66. |
Klotz JH, Dorn PL, Logan JL, Stevens L, Pinnas JL, Schmidt JO, et al. "Kissing bugs": Potential disease vectors and cause of anaphylaxis. Clin Infect Dis 2010;50:1629-34.
[Google Scholar]
|
| 67. |
Klotz J, Klotz S, Pinnas J. Animal bites and stings with anaphylactic potential. J Emerg Med 2009;36:148-56.
[Google Scholar]
|
| 68. |
Lumbreras H, Flores W, Escallon A. Allergic reactions to prints of reduviiden and their significance in Chagas disease. Zeits fur Tropenmed und Parasit 1959;10:6-19.
[Google Scholar]
|
| 69. |
Chin HC, Ahmad NW, Lim LH, Jeffery J, Hadi AA, Othman H, et al. Infestation with the cat flea, Ctenocephalides felis felis (Siphonaptera: Pulicidae) among students in Kuala Lumpur, Malaysia. Southeast Asian J Trop Med Public Health 2010;41:1331-4.
[Google Scholar]
|
| 70. |
Chua EC, Goh KJ. A flea-borne outbreak of dermatitis. Ann Acad Med Singapore 1987;16:648-50.
[Google Scholar]
|
| 71. |
Burns DA. The investigation and management of arthropod bite reactions acquired in the home. Clin Exp Dermatol 1987;12:114-20.
[Google Scholar]
|
| 72. |
Naumann DN, Baird-Clarke CD, Ross DA. Fleas on operations in Afghanistan--environmental health measures on the front line. J R Army Med Corps 2011;157:226-8.
[Google Scholar]
|
| 73. |
Bailey SF. Thrips attacking man. Can Entomol1936;68:95-8.
[Google Scholar]
|
| 74. |
Fishman HC. Thrips. Arch Dermatol1987;123:993-4.
[Google Scholar]
|
| 75. |
Steen CJ, Carbonaro PA, Schwartz RA. Arthropods in dermatology. J Am Acad Dermatol 2004;50:819-62.
[Google Scholar]
|
| 76. |
Hernandez RG, Cohen BA. Insect bite-induced hypersensitivity and the SCRATCH principles: A new approach to papular urticaria. Pediatrics 2006;118:e189-96.
[Google Scholar]
|
| 77. |
Demain JG. Papular urticaria and things that bite in the night. Curr Allergy Asthma Rep 2003;3:291-303.
[Google Scholar]
|
| 78. |
Heng MC, Kloss SG, Haberfelde GC. Pathogenesis of papular urticaria. J Am Acad Dermatol 1984;10:1030-4.
[Google Scholar]
|
| 79. |
Alexander JO. Papular urticaria and immune complexes. J Am Acad Dermatol 1985;12:374-5.
[Google Scholar]
|
| 80. |
Jordaan HF, Schneider JW. Papular urticaria:a histopathologic study of 30 patients. Am J Dermatopathol 1997;19:119-26.
[Google Scholar]
|
| 81. |
Cuéllar A, Rodríguez A, Rojas F, Halpert E, Gómez A, García E. Differential Th1/Th2 balance in peripheral blood lymphocytes from patients suffering from flea bite-induced papular urticaria. Allergol Immunopathol (Madr) 2009;37:7-10.
[Google Scholar]
|
| 82. |
Cuellar A, García E, Rodríguez A, Halpert E, Gómez A. Functional dysregulation of dendritic cells in patients with papular urticaria caused by fleabite. Arch Dermatol 2007;143:1415-9.
[Google Scholar]
|
| 83. |
Dilaimy M. Bullous papular urticaria. Cutis 1978;21:666-8.
[Google Scholar]
|
| 84. |
Jacobsohn F. Harara (urticaria multiformis endemica). Proc R Soc Med 1939;32:1587-8.
[Google Scholar]
|
| 85. |
Light JE, Toups MA, Reed DL. What′s in a name: The taxonomic status of human head and body lice. Mol Phylogenet Evol 2008;47:1203-16.
[Google Scholar]
|
| 86. |
Kirkness EF, Haas BJ, Sun W, Braig HR, Perotti MA, Clark JM, et al. Genome sequences of the human body louse and its primary endosymbiont provide insights into the permanent parasitic lifestyle. Proc Natl Acad Sci U S A 2010;107:12168-73.
[Google Scholar]
|
| 87. |
Greive KA, Lui AH, Barnes TM, Oppenheim VM. A randomized, assessor-blind, parallel-group, multicentre, phase IV comparative trial of a suffocant compared with malathion in the treatment of head lice in children. Australas J Dermatol 2010;51:175-82.
[Google Scholar]
|
| 88. |
Chosidow O, Giraudeau B, Cottrell J, Izri A, Hofmann R, Mann SG, et al. Oral ivermectin versus malathion lotion for difficult-to-treat head lice. N Engl J Med 2010;362:896-905.
[Google Scholar]
|
| 89. |
McCormack PL. Spinosad: In pediculosis capitis. Am J Clin Dermatol 2011;12:349-53.
[Google Scholar]
|
| 90. |
Cueto GM, Zerba E, Picollo MI. Permethrin-resistant head lice (Anoplura: Pediculidae) in Argentina are susceptible to spinosad. J Med Entomol 2006;43:634-5.
[Google Scholar]
|
| 91. |
Stough D, Shellabarger S, Quiring J, Gabrielsen AA Jr. Efficacy and safety of spinosad and permethrin creme rinses for pediculosis capitis (head lice). Pediatrics 2009;124:e389-95.
[Google Scholar]
|
| 92. |
Meinking TL, Villar ME, Vicaria M, Eyerdam DH, Paquet D, Mertz- Rivera K, et al. The clinical trials supporting benzyl alcohol lotion 5% (Ulesfia): A safe and effective topical treatment for head lice (pediculosis humanus capitis). Pediatr Dermatol 2010;27:19-24.
[Google Scholar]
|
| 93. |
Burgess IF, Brown CM, Lee PN. Treatment of head louse infestation with 4% dimeticone lotion: Randomised controlled equivalence trial. BMJ 2005;330:1423.
[Google Scholar]
|
| 94. |
Burgess IF, Lee PN, Matlock G. Randomised, controlled, assessor blind trial comparing 4% dimeticone lotion with 0.5% malathion liquid for head louse infestation. PLoS One 2007;2:e1127.
[Google Scholar]
|
| 95. |
Kurt O, Balcioðlu IC, Burgess IF, Limoncu ME, Girginkardeþler N, Tabak T, et al. Treatment of head lice with dimeticone 4% lotion: Comparison of two formulations in a randomised controlled trial in rural Turkey. BMC Public Health 2009;9:441.
[Google Scholar]
|
| 96. |
Heukelbach J, Pilger D, Oliveira FA, Khakban A, Ariza L, Feldmeier H. A highly efficacious pediculicide based on dimeticone: R andomized observer blinded comparative trial. BMC Infect Dis 2008;8:115.
[Google Scholar]
|
| 97. |
Burgess IF, Burgess NA. Dimeticone 4% liquid gel found to kill all lice and eggs with a single 15 minute application. BMC Res Notes 2011;4:15.
[Google Scholar]
|
| 98. |
Kaul N, Palma KG, Silagy SS, Goodman JJ, Toole J. North American efficacy and safety of a novel pediculicide rinse, isopropyl myristate 50% (Resultz). J Cutan Med Surg 2007;11:161-7.
[Google Scholar]
|
| 99. |
Barker SC, Altman PM. An ex vivo, assessor blind, randomised, parallel group, comparative efficacy trial of the ovicidal activity of three pediculicides after a single application-melaleuca oil and lavender oil, eucalyptus oil and lemon tea tree oil, and a "suffocation" pediculicide. BMC Dermatol 2011;11:14.
[Google Scholar]
|
| 100. |
Frankowski BL, Bocchini JA Jr. Head lice. Pediatrics 2010;126:392-403.
[Google Scholar]
|
| 101. |
Goates BM, Atkin JS, Wilding KG, Birch KG, Cottam MR, Bush SE, et al. An effective nonchemical treatment for head lice: A lot of hot air. Pediatrics 2006;118:1962-70.
[Google Scholar]
|
| 102. |
Bush SE, Rock AN, Jones SL, Malenke JR, Clayton DH. Efficacy of the LouseBuster, a new medical device for treating head lice (Anoplura: Pediculidae). J Med Entomol 2011;48:67-72.
[Google Scholar]
|
| 103. |
Burgess IF. Do nit removal formulations and other treatments loosen head louse eggs and nits from hair? Med Vet Entomol 2010;24:55-61.
[Google Scholar]
|
| 104. |
Burgess IF. Head lice. Clin Evid (Online). 2011;2011.pii:1703.
[Google Scholar]
|
| 105. |
Clark JM. Determination, mechanism and monitoring of knockdown resistance in permethrin-resistant human head lice, Pediculus humanus capitis. J Asia Pac Entomol 2009;12:1-7.
[Google Scholar]
|
| 106. |
Burton MJ, Mellor IR, Duce IR, Davies TG, Field LM, Williamson MS. Differential resistance of insect sodium channels with kdr mutations to deltamethrin, permethrin and DDT. Insect Biochem Mol Biol 2011;41:723-32.
[Google Scholar]
|
| 107. |
Bialek R, Zelck UE, Fölster-Holst R. Permethrin treatment of head lice with knockdown resistance-like gene. N Engl J Med 2011;364:386-7.
[Google Scholar]
|
| 108. |
Nauen R. Insecticide resistance in disease vectors of public health importance. Pest Manag Sci 2007;63:628-33.
[Google Scholar]
|
| 109. |
Holzer RB. Malaria prevention without drugs. Schweiz Rundsch Med Prax 1993;82:139-43.
[Google Scholar]
|
| 110. |
Klun JA, Khrimian A, Rowton E, Kramer M, Debboun M. Biting deterrent activity of a deet analog, two DEPA analogs, and SS220 applied topically to human volunteers compared with deet against three species of blood-feeding flies. J Med Entomol 2006;43:1248-51.
[Google Scholar]
|
| 111. |
Carroll SP, Loye J. PMD, a registered botanical mosquito repellent with deet-like efficacy. J Am Mosq Control Assoc 2006;22:507-14.
[Google Scholar]
|
| 112. |
Anonymous. Picardin-A new insect repellent. Med Lett Drugs Ther 2005;47:46-7.
[Google Scholar]
|
| 113. |
Sorge F. Prevention with repellent in children. Arch Pediatr 2009;16:S115-22.
[Google Scholar]
|
| 114. |
Dua VK, Pandey AC, Raghavendra K, Gupta A, Sharma T, Dash AP. Larvicidal activity of neem oil (Azadirachta indica) formulation against mosquitoes. Malar J 2009;8:124.
[Google Scholar]
|
| 115. |
Vatandoost H, Hanafi-Bojd AA. Laboratory evaluation of 3 repellents against Anopheles stephensi in the Islamic Republic of Iran. East Mediterr Health J 2008;14: 260-7.
[Google Scholar]
|
| 116. |
Ranson H, N′guessan R, Lines J, Moiroux N, Nkuni Z, Corbel V. Pyrethroid resistance in African anopheline mosquitoes: What are the implications for malaria control? Trends Parasitol 2011;27:91-8.
[Google Scholar]
|
| 117. |
Tawatsin A, Thavara U, Chompoosri J, Phusup Y, Jonjang N, Khumsawads C, et al. Insecticide resistance in bedbugs in Thailand and laboratory evaluation of insecticides for the control of Cimex hemipterus and Cimex lectularius (Hemiptera: Cimicidae). J Med Entomol 2011;48:1023-30.
[Google Scholar]
|
| 118. |
Fagbule D, Ekanem EE. Some environmental risk factors for childhood asthma: A case-control study. Ann Trop Paediatr 1994;14:15-9.
[Google Scholar]
|
| 119. |
Krieger RI, Dinoff TM, Zhang X. Octachlorodipropyl ether (s-2) mosquito coils are inadequately studied for residential use in Asia and illegal in the United States. Environ Health Perspect 2003;111:1439-42.
[Google Scholar]
|
| 120. |
Chen SC, Wong RH, Shiu LJ, Chiou MC, Lee H. Exposure to mosquito coil smoke may be a risk factor for lung cancer in Taiwan. J Epidemiol 2008;18:19-25.
[Google Scholar]
|
| 121. |
West S, Hildesheim A, Dosemeci M. Non-viral risk factors for nasopharyngeal carcinoma in the Philippines: Results from a case-control study. Int J Cancer 1993;55:722-7.
[Google Scholar]
|
| 122. |
Pariser DM, Meinking TL, Bell M, Ryan WG. Topical 0.5% ivermectin lotion for treatment of head lice. N Engl J Med 2012;367:1687-93.
[Google Scholar]
|
| 123. |
Meinking TL, Mertz-Rivera K, Villar ME, Bell M. Assessment of the safety and efficacy of three concentrations of topical ivermectin lotion as a treatment for head lice infestation. Int J Dermatol 2013;52:106-12.
[Google Scholar]
|
| 124. |
Burgess IF, Lee PN, Kay K, Jones R, Brunton ER. 1,2-Octanediol, a novel surfactant, for treating head louse infestation: identification of activity, formulation, and randomised, controlled trials. PLoS One 2012;7:e35419.
[Google Scholar]
|
Fulltext Views
38,861
PDF downloads
5,554





