Translate this page into:
Metastatic Merkel cell carcinoma of the face
2 Department of Biomedical Sciences and Morphological and Functional Images, University of Messina, Messina, Italy
3 Department of Human Pathology, Gaetano Barresi, University of Messina, Messina, Italy
Correspondence Address:
Mario Vaccaro
Department of Clinical and Experimental Medicine, Dermatology, University of Messina, Via C. Valeria, Gazzi, 98125 Messina
Italy
| How to cite this article: Vaccaro M, Magaudda E, Brianti P, Licata L, Cardia R, De Ponte FS. Metastatic Merkel cell carcinoma of the face. Indian J Dermatol Venereol Leprol 2016;82:562-564 |
Sir,
Merkel cell carcinoma is a rare malignant neuroendocrine skin tumor, most common in the elderly and immunocompromised, with an overall 5-year survival rate. It occurs predominantly on the sun-exposed skin of the head and neck. Lesions have no distinctive clinical features but usually present as reddish-purple, rapidly growing, painless nodules or plaques. Although the molecular pathogenesis remains largely unknown, ultraviolet radiation and immune suppression are likely predisposing factors.[1],[2]
We report a case of an 85 year old man who presented with a 2 month history of 0.5-5cm sized bluish-purple, painless nodules on the chin and submental region associated with palpable lymph nodes. Clinical examination revealed a swollen, congested and fissured lower lip with a red-violet lesion fixed to underlying tissue. The lesion extended from the left parasymphysis to the contralateral mandibular angle [Figure - 1]a, [Figure - 1]b. A subcutaneous swelling was observed in the middle third of the sternal head of the right sternocleidomastoid muscle.
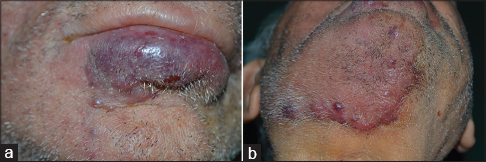 |
| Figure 1: Merkel cell carcinoma: Clinical presentation. (a) Congested and fissured lower lip with a red-violet lesion fixed to underlying layers; (b) bluish-purple painless nodules of variable size in the chin and submental region |
Microscopically, the neoplastic proliferation was localized to the superficial and deep dermis with an organoid solid infiltrative pattern characterized by neoangiogenesis. A high degree of nuclear pleomorphism and mitotic rate were noted [Figure - 2]a, [Figure - 2]b. Intense cytoplasmic immunoreactivity was evident with chromogranin A, while a granular, non-homogeneous immunoreactivity was found with neuron-specific enolase [Figure - 3]a, [Figure - 3]b, [Figure - 3]c,[Figure - 3]d. The neoplastic elements were unreactive to CK, CK7, S-100, CD68, CD99 and CD34 [Figure - 3]a, [Figure - 3]b, [Figure - 3]c,[Figure - 3]d. Ultrathin sections stained with uranyl acetate and lead citrate were observed using an electron microscope (Elmiskop II, Siemens): the most relevant feature was the presence of a dense core of neurosecretory granules (100–300 nm) located in the peri-nuclear cytoplasmic area [Figure - 4].
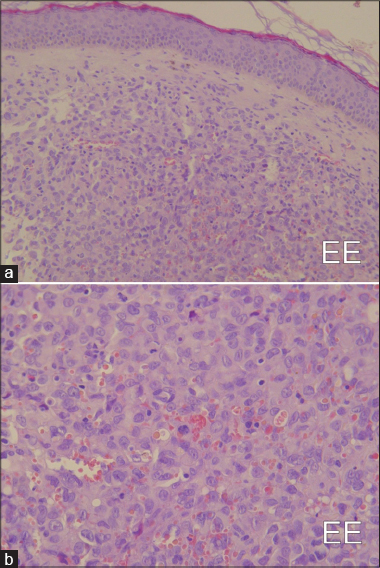 |
| Figure 2: Merkel cell carcinoma: Histopathological findings. (a) Lesion extensively infiltrating the dermis (H and E, ×80 ); (b) proliferating elements showing round/oval hyperchromatic, pleomorphic nuclei,(H and E, ×160) |
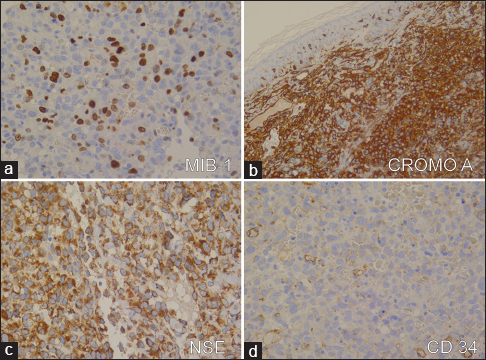 |
| Figure 3: Merkel cell carcinoma: Immunohistochemistry findings. (a) High nuclear immunoexpression of MIB-1 in more than 20% of neoplastic elements (MIB-1 antiserum, Mayer's hemalum nuclear counterstain, ×160 ); (b) Diffuse cytoplasmic immunoreactivity with chromogranin A (Mayer's hemalum nuclear counterstain, ×160 ); (c) granular non-homogeneous immunoreactivity with neuron-specific enolase staining (Mayer's hemalum nuclear counterstain, ×160 ); and (d) newly formed vessels stained with CD34 (Mayer's hemalum nuclear counterstain, ×160) |
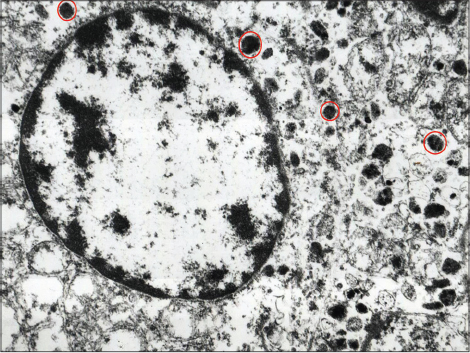 |
| Figure 4: Neoplastic merkel cell carcinoma cell exhibiting characteristic neurosecretory granules of different sizes (circled in red) (fragment retrieved from formalin-fixed paraffin-embedded tissue block, ×20,000) |
Computed tomography scan with contrast revealed parotid, submandibular and mediastinal lymphadenopathy. Soft-tissue metastases were present with invasion of temporalis, pterygoideus externus and sternocleidomastoid muscles, as well as distant metastases to the lung. Because of the aggressive nature of this tumor (T2N2M1), the patient was given chemotherapy with VP-16 (etoposide, topoisomerase inhibitor) to reduce the rate of progression.
Merkel cell carcinoma is an aggressive neoplasm with an incidence of around 3 per million cases/patients/year that increases to 11 more in acquired immune deficiency syndrome (AIDS) patients and 5 more in the organ transplanted. It is poorly responsive to anti-cancer therapies and thus prognosis is generally considered poor. The association with immunosuppression reported previously in the literature had raised the possibility of infection as the likely causative agent. Recently, a previously unknown polyomavirus, the Merkel cell polyomavirus, has been reported to be associated in about 80% of cases and thus thought to be the causative agent of this tumor.[3],[4]
Unfortunately, Merkel cell carcinoma has a non-specific clinical presentation (though rapid growth of a red violaceous papule or nodule is often noted) and thus diagnosis can be challenging.[3] The clinical differential diagnoses include basal cell carcinoma, squamous cell carcinoma, skin metastasis from another primary site, malignant lymphoma and amelanotic melanoma. Histologically, the carcinoma is composed of small round blue cells, with sparse cytoplasm, medium to large-sized hyperchromatic nuclei, multiple small nucleoli, delicately granular chromatin, abundant mitoses and numerous apoptotic figures.[5] CK20 positivity, in a distinctive peri-nuclear dot-like pattern, is a strongly suggestive of Merkel cell carcinoma.
Imaging modalities used in the staging of Merkel cell carcinoma include ultrasonography, computed tomography, magnetic resonance imaging and octreotide scans. Recently, 18F-fluorodeoxyglucose positron emission tomography has become a valuable and useful technique for staging in these patients. Staging is based on the traditional tumor, node, metastasis (TNM) classification system with prognostic data showing decreased overall survival with advancing stage of the disease.[3]
Management of Merkel cell carcinoma is largely dependent on tumor staging at presentation, with curative intent for locoregional disease and palliative intent for distant disease. A multidisciplinary approach to treatment is vital to optimize outcomes. Optimal management includes surgical excision of the primary tumor with wide resection margins and prophylactic dissection of regional nodes. In the case of metastasis to regional nodes, radiotherapy, chemotherapy (mostly using the combination of cyclophosphamide, doxorubicin and vincristine, or cisplatin/carboplatin with or without etoposide), immunotherapy, molecular-targeted therapy and lymphadenectomy may be considered.[6] This tumor is radiosensitive and radiotherapy is now mainly used as an adjuvant to surgery.
Several studies have shown that individuals with Merkel cell carcinoma are at a significantly higher risk for developing second malignancies (most commonly tumors of brain, salivary gland, biliary tract, malignant melanoma, laryngeal cancer and cutaneous basal cell carcinoma) when compared to the general population. A better knowledge of disease prognosis with regard to second malignancies is important to optimize medical and surgical management, radiotherapy, follow-up and surveillance procedures.[7]
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. |
Tang CK, Toker C. Trabecular carcinoma of the skin: An ultrastructural study. Cancer 1978;42:2311-21.
[Google Scholar]
|
| 2. |
Albores-Saavedra J, Batich K, Chable-Montero F, Sagy N, Schwartz AM, Henson DE. Merkel cell carcinoma demographics, morphology, and survival based on 3870 cases: A population based study. J Cutan Pathol 2010;37:20-7.
[Google Scholar]
|
| 3. |
Rockville Merkel Cell Carcinoma Group. Merkel cell carcinoma: Recent progress and current priorities on etiology, pathogenesis, and clinical management. J Clin Oncol 2009;27:4021-6.
[Google Scholar]
|
| 4. |
Feng H, Shuda M, Chang Y, Moore PS. Clonal integration of a polyomavirus in human Merkel cell carcinoma. Science 2008;319:1096-100.
[Google Scholar]
|
| 5. |
Andea AA, Coit DG, Amin B, Busam KJ. Merkel cell carcinoma: Histologic features and prognosis. Cancer 2008;113:2549-58.
[Google Scholar]
|
| 6. |
Bichakjian CK, Lowe L, Lao CD, Sandler HM, Bradford CR, Johnson TM, et al. Merkel cell carcinoma: Critical review with guidelines for multidisciplinary management. Cancer 2007;110:1-12.
[Google Scholar]
|
| 7. |
Saxena A, Rubens M, Ramamoorthy V, Khan H. Risk of second cancers in Merkel cell carcinoma: A meta-analysis of population based cohort studies. J Skin Cancer 2014;2014:184245.
[Google Scholar]
|
Fulltext Views
1,753
PDF downloads
671





