Translate this page into:
Photoprotection
Correspondence Address:
C R Srinivas
Department of Dermatology, PSG Hospitals, Peelamedu, Coimbatore-641 004, Tamil Nadu
India
| How to cite this article: Rai R, Srinivas C R. Photoprotection. Indian J Dermatol Venereol Leprol 2007;73:73-79 |
Abstract
The deleritious effect of ultraviolet radiation on humans has increased the need for photoprotection. Sunscreens are widely used as photo protective agents. They are divided into chemical sunscreens which absorb high-energy ultraviolet rays and physical blockers which reflect or scatter light. Effectiveness of sunscreens depends upon sun protection factor and its substantivity. Clothing is also important for sun protection and its effectiveness is measured by Ultraviolet Protection Factor. There are many other agents with photo protective properties, which range from antioxidants to plant extracts to DNA repair enzymes. Usage of wide brimmed hats and sunglasses, avoidance of solar exposure at times of peak intensity, use of cover-up garments and sunscreen lotions are effective for photo protection of the skin.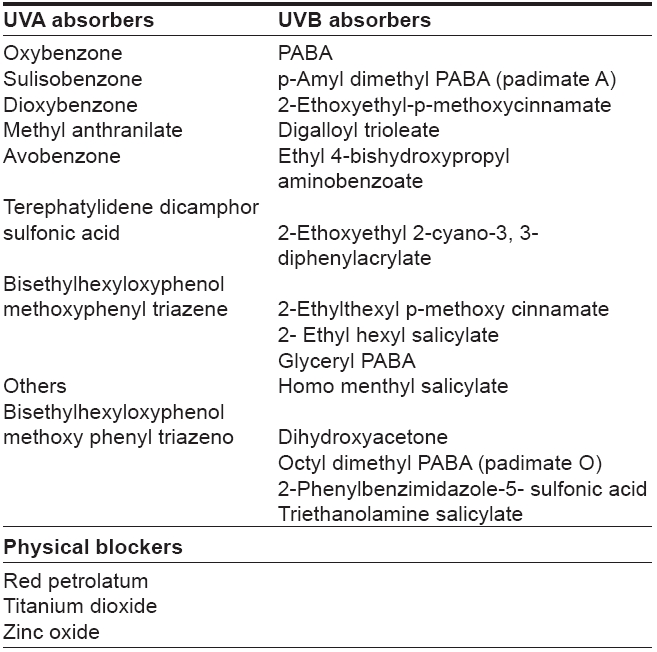


INTRODUCTION
The need to protect from sunlight has been recognized since long. The ancient Greeks used olive oil as a type of sunscreen but this was not very effective. In 1944 a pharmacist Benjamin Greene to save the soldiers from the sun′s harmful rays, used a sticky, red substance, which he called red vet pet (red veterinary petrolatum). This worked by physically blocking the sun′s rays. It did not work nearly as well as modern sunscreens, but it was a start. Since then sunscreens have come a long way.
ULTRAVIOLET RADIATION
Ultraviolet radiation is a small component of the electromagnetic spectrum with a narrow band of radiation from 200-400 nm. The UV spectrum is further divided into UVC (200-290 nm), UVB (290-320 nm) and UVA (320-400). UVA rays constitute 90-95% of the ultraviolet light reaching the earth. They have a relatively long wavelength (320-400 nm) and are not absorbed by the ozone layer. UVA light penetrates the furthest into the skin and is involved in sun tanning, UVA tends to suppress the immune function and is implicated in premature ageing of the skin. [1] UVB rays are partially absorbed by the ozone layer and have a medium wavelength (290-320 nm). They do not penetrate the skin as far as the UVA rays do and are the primary cause of sunburn. They are also responsible for photoaging, photo carcinogenesis and are implicated in cataract formation. [1] UVC rays have the shortest wavelength (below 290 nm) and are almost totally absorbed by the ozone layer. As the ozone layer thins UVC rays may begin to contribute to sunburning and premature ageing of the skin. [1]
To minimize the deleterious effect of ultraviolet rays, photoprotection is important.
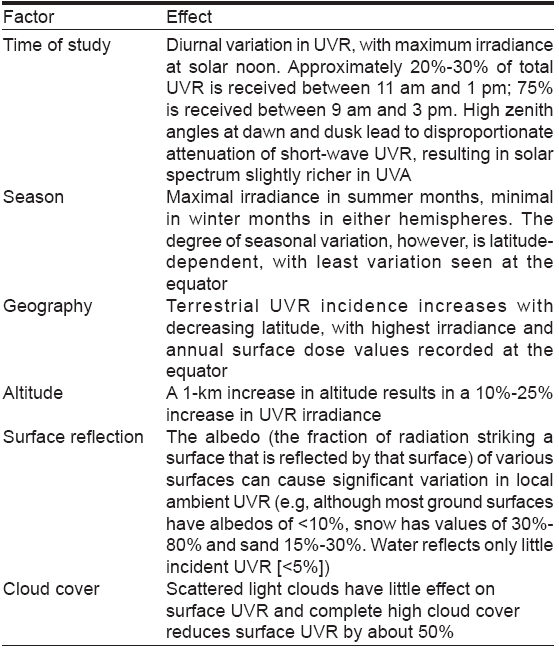
Variability of UV [2]
The various factors that determine quality and quantity of terrestrial ultraviolet radiation are given in [Table - 1]. [2]
SUNSCREENS [3]
Sunscreens have been divided into chemical absorbers and physical blockers on the basis of their mechanism of action. Chemical sunscreens are generally aromatic compounds conjugated with a carbonyl group. This general structure allows the molecule to absorb high-energy ultraviolet rays and release the energy as lower-energy rays, thereby preventing the skin-damaging ultraviolet rays from reaching the skin. So, upon exposure to UV light, most of the ingredients (with the exception of avobenzone) do not undergo significant chemical change. This allows these ingredients to retain the UV-absorbing potency without significant photodegradation, thereby preventing the skin-damaging ultraviolet rays from reaching the skin. Physical blockers or non chemical sunscreens reflect or scatter UVR. They contain inert minerals such as titanium dioxide or zinc oxide.
[Table - 2] lists the most common chemicals used in sunscreens. These compounds absorb a specific portion of the UVR spectrum. Depending on the details of each compound′s molecular structure, the absorbance may be effective for UVB alone or both UVB and to some extent UVA.
Ultraviolet B Blockers [3]
Para-aminobenzoic acid
This was one of the first chemical sunscreens to be widely available. The problems limiting its use include an alcoholic vehicle, staining of clothes and a number of adverse reactions. Ester derivatives, mainly padimate O or octyl dimethyl PABA is associated with greater compatibility in a variety of cosmetic vehicles, a lower potential for staining and adverse reactions. Padimate O is the most potent UV-B absorber. The decline in its use, along with the demand for higher SPF products, has led to the incorporation of multiple active ingredients into a single product to achieve the desired SPF, replacing sunscreens with single PABA esters.
Cinnamates
The cinnamates have largely replaced PABA derivatives as the next most potent UV-B absorbers. Octinoxate or Octyl methoxy cinnamate is the most frequently used sunscreen ingredient. Octinoxate is less potent than padimate O.
Octyl salicylate
Octisalate or octyl salicylate is used to augment the UV-B protection in a sunscreen. Salicylates are weak UV-B absorbers and they are generally used in combination with other UV filters. Other salicylates must be used in higher concentrations. They all have a good safety profile.
Octocrylene
Octocrylene may be used in combination with other UV absorbers to achieve higher SPF formulas. Octocrylene used in combination with other sunscreen ingredients, such as avobenzone, may add to the overall stability of these ingredients in a specific formula.
Phenyl benzimidazole sulfonic acid
Most chemical sunscreen ingredients are oils that are soluble in the oil phase of emulsion systems, accounting, in part, for the heavy, greasy aesthetics of many of these products. Ensulizole or phenyl benzimidazole sulfonic acid is water soluble and is used in products formulated to feel lighter and less oily, such as daily use cosmetic moisturizers. It is a selective UV-B filter, allowing almost all UV-A transmission.
Ultraviolet-A Blockers [3]
Benzophenone
Although benzophenones are primarily UV-B absorbers, oxybenzone absorbs well through UV-A2. Oxybenzone can be considered a broad-spectrum absorber
Anthranilate
Anthranilates are weak UV-B filters and they absorb mainly in the UV-A2 portion of the spectrum. Anthranilate are less effective in this range than benzophenones and they are less widely used.
Avobenzone
It provides superior protection through a large portion of the UV-A range, including UV-A1. Potentially a significant addition to sunscreen products for true broad-spectrum UV protection, concerns have been raised regarding its photostability and its potential to degrade other sunscreen ingredients.
Terephthalylidene dicamphor sulfonic acid or Mexoryl SX
This provides protection within the near UV-A range (320- to 340-nm); it is water soluble and less water resistant.
Bisethylhexyloxyphenol methoxy phenyl triazene
This broadband sunscreen filter lends photostability to avobenzone containing sunscreens.
PHYSICAL BLOCKERS [3]
Physical sunscreens contain inert minerals such as titanium dioxide or zinc oxide. The particles scatter and reflect the solar radiation. The most common type used is ultrafine titanium dioxide, which provides broad-spectrum protection against UV and due to the particle size, it is invisible when applied in cosmetic formulations. They are chemically stable and do not cause photo-allergic or contact dermatitis and do not break down over time and are far less liable to cause skin irritation.
SYSTEMIC PHOTOPROTECTION [3]
Systemic agents have been investigated for photoprotection since they provide protection for the entire body and are likely to eliminate the problem of substantivity which is important for topical products.
Oral agents that have been tried include PABA, indomethacin, retinol, steroids, psoralen, antimalarials and antioxidants like vitamin A, vitamin C, vitamin E and beta-carotene. Antioxidants are less potent than sunscreens in preventing sunburn. [4]
Measuring and Rating Effectiveness: UVB and UVA
The concept of the sun protection factor (SPF) was originally proposed by the Austrian scientist Franz Greiter and subsequently adopted by many regulatory authorities in the cosmetic and pharmaceutical industries. It tells us about how much longer skin covered with sunscreen takes to burn as compared to unprotected skin. SPF is a numerical rating system to indicate the degree of protection provided by a sun care product. It is defined as the ratio of the least amount of ultraviolet energy (UVB) required to produce minimal erythema on sunscreen-protected skin to the amount of energy required to produce the same erythema on unprotected skin. [5] Internationally agreed procedures define protected skin as that to which a 2 mg/cm 2 layer of sunscreen has been applied. [5]
The efficacy of a product is related not only to its SPF but also to its substantivity. [5] Substantivity is the ability of a sunscreen to remain effective under the stress of prolonged exercise, sweating and swimming. The following three labeling recommendations have been suggested to help clarify substantivity:
- Sweat-resistant: protects up to 30 minutes of continuous heavy perspiration.
- Water-resistant: protects up to 40 minutes of continuous water exposure; and
- Waterproof: protects for up to 80 minutes of continuous water exposure. PABA and its esters demonstrate more resistance to sweating and/or water immersion than do other chemical sunscreens. [6]
UVA Protection of Sunscreen
Currently there is no uniformly accepted standard method for measuring UVA protection of sunscreen. [7] The more commonly used in vivo methods are immediate pigment darkening (IPD), [8] persistent pigment darkening (PPD) [9] and protection factor in UVA. [10] The UV erythema protection factor assesses the time to induce erythema in UVA exposed skin. Among these methods, PPD is most commonly used because pigmentation remains stable between 2 and 24 hours and it is sensitive for all UVA filters irrespective of their range of absorbency within the UVA range. [10] Broad-spectrum protective effect can be evaluated in vitro by using spectrophotometry to assess the critical wavelength value, which is defined as the wavelength below which 90% of sunscreen′s UV absorbency occurs as measured at 290 to 400 nm. [11] It has been demonstrated that critical wavelength value is commensurate with SPF. [11] A consensus conference recommended that sunscreens with broad-spectrum label should have a critical wavelength of more than 370 nm and a PPD or protection factor in UVA greater than 4. [7]
SIDE EFFECTS OF SUNSCREENS
Sunscreens that contain aminobenzoic acid and its esters (PABA), cinnamates and oxybenzone can cause contact dermatitis or photosensitivity reactions. [12],[13] Aminobenzoic acid is chemically similar to other drugs that cause photosensitivity reactions and so these individuals should not use a sunscreen containing aminobenzoic acid or one of its derivatives (aminobenzoate, menthyl anthranilate or padimate A or O) due to cross-reactivity. A sunscreen containing oxybenzone or cinoxate should be recommended for these individuals instead. [14] Miscellaneous compounds, such as fragrances, lanolin, alcohol and preservatives may also cause skin and eye irritation or sensitization.
A new nitrosamine known as NPABAO was found in certain sunscreens containing padimate-O. [15] Nitrosamines themselves can be carcinogenic but it is uncertain whether this is present in sufficient quantities in sunscreens to be of concern. [15]
CONTROVERSIES CONCERNING SUNSCREENS
Skin under the age of six months may have different absorptive characteristics than that of adults and the biological systems that metabolize and excrete drugs absorbed through the skin may not be fully developed in children. Hence, it is recommended that sunscreens containing aminobenzoic acid should be avoided in children younger than six months of age and that no sunscreens be used on children during the first six months of life. [16]
Sunscreens, when liberally and frequently applied, reduce sunburn. Sunscreens will also protect against other forms of damage but evidence in humans for a cancer-preventive effect against cutaneous malignant melanoma and basal cell carcinoma is inadequate. [17]
Some sunscreens such as PABA, oxybenzone, padimate O interact with the skin to produce free radicals and may damage cellular DNA. [18],[19],[20] Some studies state that PABA is mutagenic but others indicate it is neither mutagenic nor photomutagenic. [21] It has been reported that regular use of sunscreens can affect vitamin D synthesis, [22] although several clinical trials have shown that there was no effect on vitamin D levels. [23]
SUNSCREEN APPLICATION
The ideal sunscreen should have a high SPF rating, be well-tolerated, cosmetically pleasant, non-toxic, equally effective against UVA and UVB, photostable, water-resistant and inexpensive.
Unfortunately, no single such sunscreen currently exists. Sunscreen should be applied 20 to 30 minutes before sun exposure so the product has a chance to bond with the skin. Products containing PABA and PABA-like chemicals, however, may need to be applied up to two hours in advance of sun exposure in order to achieve their maximal effect. [16] Contrary to the common advice that sunscreen should be reapplied every 2-3 hours, research has shown that the best protection is achieved by application 15-30 minutes before exposure, followed by one reapplication 15-30 minutes after the sun exposure begins. Further reapplication is only necessary after activities such as swimming, sweating and rubbing. [24]
Most individuals do not apply enough sunscreen to achieve adequate protection. It has been found that 20-50% of the required amount of sunscreen is usually used by most individuals. To cover the average 1.73 m 2 adult, approximately 35ml of sunscreen is required. The teaspoon rule of applying sunscreen is as follows: Apply slightly more than ½ teaspoon (~3ml) to each arm, to the face and the neck. On each leg, the chest and back, apply slightly more than a teaspoon (~6ml). [25] Using an adequate amount of sunscreen (2 mg/cm 2 ) provides greater sun protection than using an inadequate amount of a sunscreen with a higher SPF rating.
Patients should select broad-spectrum sunscreens that contain agents that effectively block both UVB and UVA rays with an SPF of 30 or greater. A sunscreen with a SPF of 15 filters out approximately 94% of the UVB rays. One with a SPF of 30 filters out 97%. The SPF applies for UVB rays only. The protection provided against UVA rays in chemical sunscreens is about 10% of the UVB rating. [26]
Natural pigments such as titanium dioxide and zinc oxide are good UVB and UVA blockers. Today, certain chemicals can be added to sunscreen products to give them good UVA ray blocking abilities. Such chemicals include avobenzone and Mexoryl SX. Foundation makeup products without sunscreen may not offer more than a SPF of 4 via its pigment content. However, most of cosmetic products that contain sunscreen chemicals offer various SPF levels up to 15-30.
PHOTOPROTECTION FOR HAIR
The primary photoaging effect of sunlight on hair is oxymelanin production which results in lightening of hair, and causing disulphide bond disruption resulting in physical weakening of hair shaft. [27],[28]
Various hair care products like shampoos, instant conditioners, deep conditioners and hairstyling products can provide some hair photoprotection. [29] The degree of photo protection offered by a hair care product is minimal. A more thorough, thicker application will result in better photoprotection, but this is difficult given the massive surface area of a head of hair. Many currently available sunscreen actives are not substantive to hair, meaning that they do not stick or adhere to the hair well, preventing sunscreen deposition and facilitating removal. A better approach to hair photoprotection is the use of clothing, such as a cap, hat, scarf or umbrella.
CLOTHING FOR SUN PROTECTION
Clothing for sun protection is another approach to ultraviolet ray protection. Comparisons between effectiveness of fabrics are difficult since some tests measure SPF factors or sun-burning time while others measure actual transmission of ultraviolet rays, ultraviolet protection factor (UPF). [30]
Fabric SPF is the ratio of the radiation dose needed to produce just perceptible erythema under fabric covered skin to the radiation dose needed to produce just perceptible erythema of uncovered skin. The definition of UPF is that it is the ratio of average effective UV irradiance transmitted without fabric to the average effective UV irradiance transmitted through fabric. The UPF value calculated therefore indicates how much longer a person can stay in the sun when fabric covers the skin as compared with the length of time in the sun without fabric covering to obtain the same perceptible skin reddening.
Fabrics are placed into classes based on the calculated label UPF value. [31] The good protection is composed of fabrics with label UPF value of 15 to 24, the very good protection class is composed of fabrics with label UPF values of 40 to 50 and 50+ (the highest value permitted on a label).
Fibers are classified in three distinct groups based on their UV absorbing properties, [32]
Group 1: Polyester, which is the best UV absorber
Group 2: Wool, silk and nylon
Group 3: Cotton and rayon (cellulosic fibers), which are the poorest absorbers
UV absorbers
Factors that affect UPF of a fabric are: [33]
- Tightly woven thicker fibers have better UPF.
- Washing increases UPF because of shrinkage whereas hydration decreases it.
- Fabrics prone to stretch have lower UPF
- Chemical treatment with bleaching agents results in attenuation of UV rays.
- Colored fabric has greater UPF.
- Greater the distance of a fabric from the skin, better is the photoprotection.
Cover-up garments, garments that cover the arms, legs and torso, wide brimmed hats and sunglasses are also recommended for greater UV protection.
FUTURE TRENDS IN PHOTOPROTECTION
There are number of agents that have been shown to have photo protective properties; most are in various stages of investigation. The antioxidants are caffeic acid, poly podium leukotomes, zinc, polyphenolic compounds, isoflavone, N-acetylcysteine and butyrated hydroxytoluene (synthetic antioxidant). [34],[35],[36],[37],[38],[39] Calcitriol and citrus which are free radical scavengers. [40] Plant oligosaccharides and genistein help in prevention of UV induced immunosuppression. [41]
Sunscreens alone may provide insufficient protection from UVR as they prevent sunburn from UV-B radiation and provide more limited protection from UV-A radiation. Avoiding solar exposure at times of peak intensity, using cover-up garments and sunscreen lotions is an effective combination for protection of the skin on sunny and cloudy days.
| 1. |
Larsen HR. Sunscreens and Cancer. Intl J Alternative Complement Med 1994;12:17-9.
[Google Scholar]
|
| 2. |
Matts PJ. Solar ultraviolet radiation: Definitions and terminology. Dermatol Clin 2006;24:1-8.
[Google Scholar]
|
| 3. |
Lowe NJ. An overview of ultraviolet radiation, Sunscreens and photo-induced dermatoses. Dermatol Clin 2006;24:9-17.
[Google Scholar]
|
| 4. |
Hamanaka H, Miyachi Y, Imamura S. Photo protective effect of topically applied superoxide dismutase on sunburn reaction in comparison with sunscreen. J Dermatol 1990;17:595-8.
[Google Scholar]
|
| 5. |
Griffin ME, Bourget TD, Lowe NJ. Sun protection factor determination in the United States. In: Lowe NJ, Pathak M, Shaath N, editors. Sunscreens, development, evaluation and regulatory aspects. 2nd ed. Marcel Dekker: New York; 1997. p. 499-512.
[Google Scholar]
|
| 6. |
Lester RS. Topical Formulary for the Pediatrician. Ped Clin North Am 1983;30:749-50.
[Google Scholar]
|
| 7. |
Lim HW, Naylor M, Honigsmann H, Gilchrest BA, Cooper K, Morison W, et al. American academy of dermatology consensus conference on UVA protection of sunscreens: Summary and recommendations. J Am Acad Dermatol 2001;44:505-8.
[Google Scholar]
|
| 8. |
Kaidbey KH, Barnes A. Determination of UVA protection factors by means of immediate pigment darkening in normal skin. J Am Acad Dermatol 1991;25:262-6.
[Google Scholar]
|
| 9. |
Moyal D, Chardon A, Kollias N. UVA protection efficacy of sunscreens can be determined by the persistent pigment darkening (PPD) method. (Part 2). Photodermatol Photoimmunol Photomed 2001;16:250-5.
[Google Scholar]
|
| 10. |
Cole C. Multicenter evaluation of sunscreen UVA protectiveness with the protection factor test method. J Am Acad Dermatol 1994;30:729-36.
[Google Scholar]
|
| 11. |
Diffey BL, Tanner PR, Matts PJ, Nash JF. In vitro assessment of the broad-spectrum ultraviolet protection of sunscreen products. J Am Acad Dermatol 2000;43:1024-35.
[Google Scholar]
|
| 12. |
Dromgoole SH, Maibach HI. Contact sensitization and photocontact sensitization of sunscreening agents. In: Lowe NJ, Shaath N, editors, Sunscreens, development, evaluation and regulatory aspects. Marcel Dekker: New York; 1990. p. 313-40.
[Google Scholar]
|
| 13. |
Dromgoole SH, Maibach HI. Sun-screening intolerance: Contact and photocontact sensitization and contact urticaria. J Am Acad Dermatol 1990;22:1068-78.
[Google Scholar]
|
| 14. |
DeSimone EM 2nd. Sunscreen and sunscreen products. In: Handbook of nonprescription drugs. 10th ed. Feldman EG, Davidson DE, editors. American Pharmaceutical Association: Washington, DC; 1986. p. 575-87.
[Google Scholar]
|
| 15. |
Pathak MA, Robins P. A response to concerns about sunscreens: A report from the skin cancer foundation. J Dermatol Surg Oncol 1989;15:486-7.
[Google Scholar]
|
| 16. |
Hurwitz S. The sun and sunscreen protection: Recommendations for children. J Dermatol Surg Oncol 1988;14:657-60.
[Google Scholar]
|
| 17. |
Garland CF, Garland FC, Gorham ED. Could sunscreens increase melanoma risk? Am J Public Health 1992;82:614-5.
[Google Scholar]
|
| 18. |
Schallreuter KU, Wood JM, Farwell DW, Moore J, Edwards HG. Oxybenzone oxidation following solar irradiation of skin: Photoprotection versus antioxidant inactivation. J Invest Dermatol 1996;106:583-6.
[Google Scholar]
|
| 19. |
Sutherland JC, Griffin KP. P-aminobenzoic acid can sensitive the formation of pyrimidine dimmers in DNA: Direct chemical evidence. Photochem Photobiol 1984;40:391-4.
[Google Scholar]
|
| 20. |
Gulston M, Knowland J. Illumination of human keratinocytes in the presence of the sunscreen ingredient Padimate -O and through an SPF-15 sunscreen reduces direct photodamage to DNA but increases strand breaks. Mutat Res 1999;444:49-60.
[Google Scholar]
|
| 21. |
Naylor MF, Farmer KC. The case for sunscreens a review of their use in preventing actinic damage and neoplasia. Arch Dermatol 1997;133:1146-54.
[Google Scholar]
|
| 22. |
Matsuoka LY, Wortsman J, Hanifan N, Holick MF. Chronic sunscreen use decreases circulating concentrations of 25- hydroxyvitamin D: A preliminary study. Arch Dermatol 1988;124:180-24.
[Google Scholar]
|
| 23. |
Farrerons J, Barnadas M, Rodriguez J, Renau A, Yoldi B, Lopez-Navidad A, et al. Clinically prescribed sunscreen (sun protection factor 15) does not decrease serum vitamin D concentration sufficiently either to induce changes in parathyroid function or in metabolic markers. Br J Dermatol 1998;139:422-7.
[Google Scholar]
|
| 24. |
Diffey BL. When should sunscreen be reapplied? J Am Acad Dermatol 2001;45:882-5.
[Google Scholar]
|
| 25. |
Schneider J. The teaspoon rule of applying sunscreen. Arch Dermatol 2002;138:838-9.
[Google Scholar]
|
| 26. |
Kaidbey K, Gange RW. Comparison of methods of assessing photoprotection against ultraviolet A in vivo. J Am Acad Dermatol 1987;16:346-53.
[Google Scholar]
|
| 27. |
Jachowicz J. Hair damage and attempts at its repair. J Soc Cosmet Chem 1987;38:263-86.
[Google Scholar]
|
| 28. |
Hoting E, Zimmerman M, Hilterhaus-Bong S. Photochemical alterations in human hair: I. Artificial irradiation and investigation of hair proteins. J Soc Cosmet Chem 1995;46:85-99.
[Google Scholar]
|
| 29. |
Draelos ZD. Sunscreen and Hair photoprotection. Dermatol Clin 2006;24:81-4.
[Google Scholar]
|
| 30. |
Hatch KL, Osterwalder U. Garments as solar ultraviolet radiation screening materials. Dermatol Clin 2006;24:85-100.
[Google Scholar]
|
| 31. |
American Society for Testing and Materials (ASTM International). Standard D 6603-00, Standard guide for labeling of UV-protective textiles. In: Bailey SJ, Baldwin NC, McElrone EK, et al, editors. ASTM standard. 2004. 7:03. p. 1187-91.
[Google Scholar]
|
| 32. |
Crews PC, Kachman S, Beyer AG. Influence on UVR transmission of undyed woven fabrics. Textile Chem Colorist 1993;31:17-26.
[Google Scholar]
|
| 33. |
Kullavanijaya P, Lim HW. Photoprotection. J Am Acad Dermatol 2005;52:937-58.
[Google Scholar]
|
| 34. |
Emonet-Piccardi N, Richard MJ, Ravanat JL, Signorini N, Cadet J, Beani JC. Protective effects of antioxidants against UVA-induced DNA damage in human skin fibroblasts in culture. Free Radic Res 1998;29:307-13.
[Google Scholar]
|
| 35. |
Rostan EF, DeBuys HV, Madey DL, Pinnell SR. Evidence supporting zinc as an important antioxidant for skin. Int J Dermatol 2002;41:606-11.
[Google Scholar]
|
| 36. |
Elmets CA, Singh D, Tubesing K, Matsui M, Katiyar S, Mukhtar H. Cutaneous photoprotection from ultraviolet injury by green tea polyphenols. J Am Acad Dermatol 2001;44:425-32.
[Google Scholar]
|
| 37. |
van den Broeke LT, Beijersbergen van Henegouwen GM. Topically applied N-acetylcysteine as a protector against UVB-induced systemic immunosuppression. J Photochem Photobiol B 1999;27:61-5.
[Google Scholar]
|
| 38. |
Widyarini S, Spinks N, Husband AJ, Reeve VE. Isoflavonoid compounds from red clover (Trifolium pretense) protect from inflammation and immune suppression induced by UV radiation. Photochem Photobiol 2001;74:465-70.
[Google Scholar]
|
| 39. |
Koone MD, Black HS. A mode of action of butylated hydroxytoluene - mediated photoprotection. J Invest Dermatol 1986;87:343-7.
[Google Scholar]
|
| 40. |
Attaguile G, Russo A, Camposo A, Savoca F, Acqiaviva R, Ragua N, et al. Antioxidant activity and protective effect on DNA cleavage of extracts from Cistus incanus L and Cistus monospeliensis L. Cell Biol Toxicol 2000;16:83-90.
[Google Scholar]
|
| 41. |
Strickland FM, Darvill A, Albersheim P, Eberhard S, Pauly M, Pelley RP. Inhibition of UV-induced immune suppression and interleukin-10 production by plant oligosaccharides and polysaccharides. Photochem Photobiol 1999;69:141-7.
[Google Scholar]
|
Fulltext Views
9,374
PDF downloads
3,855





