Translate this page into:
Small-fibre neuropathy in leprosy the role of in vivo confocal microscopy: A cross-sectional study
-
Received: ,
Accepted: ,
How to cite this article: Cajueiro MA, Cavalcanti BM, Cavalcanti CM, Brito MFM, Bonfim C, Cunha Correia CD. Small-fibre neuropathy in leprosy the role of in vivo confocal microscopy: A cross-sectional study. Indian J Dermatol Venereol Leprol 2023;89:524–9.
Abstract
Background
Leprosy (or Hansen’s disease) continues to present considerable challenges regarding containment and early diagnosis. Leprosy is considered to be primarily a neural disease that first affects the sensory function of small fibres. Although the condition is well described in terms of clinical manifestations and histology, few studies have been undertaken to detect damage done to small-fibre sensory nerves. In vivo confocal microscopy is a useful tool for conducting a detailed evaluation of these structures, although its use in individuals affected by leprosy has still not been explored.
Objective
To evaluate in vivo confocal microscopy findings in Hansen’s disease patients and their association with clinical variables relating to this disease.
Method
A cross-sectional case-series type study was carried out between October 2019 and May 2021, in Recife, Pernambuco, Brazil. Socio-demographic and clinical data were gathered from 21 patients with leprosy. The douleur neuropathique 4 neuropathic pain questionnaire was used to evaluate pain. In vivo confocal microscopy of the cornea was employed to evaluate the small-calibre fibres. Findings were compared with those for a control group of 23 healthy individuals.
Results
In relation to clinical parameters, 90.5% of the patients were classified as “multibacillary” according to the World Health Organization criteria, and 70% as dimorphic or borderline, in accordance with the Madrid classification. Around 52.4% had received a diagnosis after one year or less of living with the disease, while 95.2% presented alterations in small-fibre sensory function and 35% presented such alterations in the large fibre. Neuropathic pain was present in 81% of the patients. In vivo confocal microscopy found no statistically significant difference in mean age and distribution according to sex between the Hansen disease patients and the control group of healthy individuals. The median-of-means for dendritic cells and volume of sub-basal nerve fibres in the control group were used to test for normality. Both eyes of all leprosy patients examined contained higher number of dendritic cells than the median value and a volume of sub-basal nerve fibres lower than the mean. These differences were statistically significant (P < 0.001 and P < 0.001, respectively). Multibacillary individuals had a median number of dendritic cells two times that of paucibacillary individuals (P = 0.035).
Limitations
No association was found between the variables examined using in vivo confocal microscopy and clinical variables relating to small-fibre damage, the neuropathic pain questionnaire or alterations detected by the neurological examination. We believe, however, that Cochet-Bonnet esthesiometry of the cornea may have revealed such an association.
Conclusion
In vivo confocal microscopy is a useful diagnostic tool for detecting small fibre loss in individuals affected by leprosy and may constitute a useful addition to the range of tools available to help curb the effects of neuropathy in these patients.
Keywords
Leprosy
diagnosis
small-fibre neuropathy
in vivo confocal microscopy
Plain Language Summary
Containment and early diagnosis of leprosy continue to pose a challenge. Leprosy is a neural disease that initially affects the sensory function of small fibres. In vivo confocal microscopy is a useful tool for conducting a detailed evaluation of these structures, although its use in individuals affected by leprosy has still not been explored. The aim of the present study was to evaluate this method in leprosy and its association with clinical variables. Clinical data were gathered for 21 patients, including the douleur neuropathique 4 neuropathic pain questionnaire. The in vivo confocal microscopy findings were compared to those of 23 healthy individuals. The multibacillary form was found to be present in 90.5% of patients, 95.2% of patients had alterations in small fibre sensory function, and 81% experienced neuropathic pain. In vivo confocal microscopy found an increased number of dendritic cells and a decrease in the number of sub-basal nerve fibres. Multibacillary individuals presented a median number of dendritic cells twice as high as that of paucibacillary individuals. In vivo confocal microscopy is, therefore, a useful diagnostic tool for the detection of small fibre loss in individuals affected by leprosy. We recommend that it be applied early on in the course of the disease to help curb the effects of neuropathy in such patients.
Introduction
Leprosy is primarily a disorder of the peripheral nerves, initially affecting the sensory small fibres.1-3 The World Health Organisation classification identifies as “paucibacillary” those individuals who present with five skin lesions or less and test negative on the bacilloscopy test. Those with five skin lesions or more testing positive on this test are considered ‘multibacillary’. On the other hand, the Madrid classification (1953) takes into consideration the clinical characteristics of the disease, classifying it as indeterminate, tuberculoid, dimorphic or borderline and Virchow-type or lepromatous.4,5
Despite the existence of programs aiming to control this disease, early diagnosis and containment of leprosy still pose an enormous challenge, since current treatment options fail to break the chain of transmission.6
Recent research has begun to study small-fibre nerves in individuals affected by leprosy.7 This pattern of involvement determines hypoesthesia and pain, which can be measured using the DN4 neuropathic pain questionnaire. This is a tool that provides better characterization of pain and treatment.8,9
Diagnosis of small fibre neuropathy is challenging since the clinical picture may be difficult to interpret and the results of nerve conduction studies are often normal. Further complimentary methods are therefore essential. In vivo confocal microscopy is one of the recommended clinical examinations that is currently receiving greater attention.10
It is estimated that 70-75% of leprosy patients will present with eye disorders. Hansen bacilli proliferate in the eyeball, especially the anterior chamber. This involves small fibre autonomic nerves and degeneration of axonal fibre and may lead to damage, especially in the cornea.11,12
Detailed examination of these corneal nerves can be carried out using in vivo confocal microscopy, a non-invasive procedure that is capable of producing images with a quasi-histological resolution to examine the number, length, density and ramification of the nerve fibres.13-16 This procedure is increasingly being used for diagnosis and ancillary treatment in patients with systemic diseases and eye disorders. Its use as an ancillary tool in the treatment of leprosy is, however, yet to be explored.17,18
The aim of the present study was to evaluate in vivo confocal microscopy findings and their association with clinical variables in individuals affected by leprosy.
Methods
A case series study was carried out with leprosy outpatients attending Policlínica Lessa de Andrade, Hospital Universitário Oswaldo Cruz and Hospital das Clínicas de Pernambuco, Recife, Pernambuco, Brazil, between October 2019 and June 2021.
The study included individuals aged 18 years or older of both sexes who had received a diagnosis of leprosy. Individuals were excluded if they reported other systemic diseases associated with peripheral neuropathy or severe cognitive disorders or were undergoing chemotherapy.
The pain was evaluated using the douleur neuropathique 4 neuropathic pain questionnaire. A score higher than four on this scale distinguishes neuropathic from nociceptive pain.
The sensitivity of small fibres was assessed using cotton for touch perception and a needle for the perception of pain. The sensitivity of large fibres was evaluated using a 128 Hz tuning fork.
Each participant was invited to attend the Pernambuco Eye Hospital to undergo a confocal microscopy examination.
Patients were excluded from the in vivo confocal microscopy examination if they had previously undergone a cornea transplant or intraocular surgery. They were also excluded if they have had infectious keratitis and/or corneal scarring, dry eye secondary to systemic diseases other than leprosy, or other disorders of the ocular surface.
An in vivo confocal microscopy examination was carried out using an HRT3 laser platform (Rostock Cornea Module with Heidelberg Retina Tomograph 3®; Heidelberg Engineering GmbH, Dossenheim, Germany) in the central region of the cornea in both eyes. A 63x immersion objective lens (Olympus 63x®, Tokyo, Japan), captured images of a 400 × 400 µm area with 800x total magnification and axial resolution of one micrometre. Three representative images were selected for each eye. A drop of fast-acting local anaesthetic was placed in each eye. Better quality images were obtained by applying a thin layer of 0.2% polyacrylic acid ophthalmic gel to the front of the objective lens, to help attach it to the surface of the eye.
Free software distributed by the National Institute of Health, ImageJ and the NeuronJ plugin were used to measure the number of sub-basal nerve fibres and the density of dendritic cells.19
Descriptive analysis was carried out to compare the case and control groups in relation to age and sex, using Student’s t-test and Pearson’s chi-square test, respectively. Normality was established for the variables relating to the number of dendritic cells and the density of sub-basal nerve fibres, with log-transformation for the number of dendritic cells. Student’s comparison of means t-test was then applied to compare these variables in the group of leprosy patients with those in the control group and to determine associations with clinical characteristics. Medians and interquartile ranges were obtained for the number of dendritic cells. Statistical analysis was carried out using Stata Version 14 software with a level of significance of 5%.
The research ethics committee of Oswaldo Cruz University Hospital, Recife-Brazil approved this study under CAAE number 16493619.0.0000.5192.
Results
The control group comprised 23 individuals, 65.2% of whom were female, with a mean age of 56.2 ± 8.8 years. There was no statistically significant difference in mean age or distribution according to sex between leprosy and the control groups. The socio-demographic data showed that 40% of participants had a household income of less than or equal to one minimum wage and 47.6% had received no more than four years of schooling.
Clinical data revealed that 90.5% were classified as “multibacillary,” 70% were borderline, 4.8% were purely neural. Alterations in the small and large fibres were found in 95.2% and 35% of patients respectively. Eighty one percent of the patients had developed neuropathic pain, as determined using the douleur neuropathique 4 questionnaire [Table 1].
| Characteristics | Number (%) |
|---|---|
| Sex: Female, male | 15 (71.4%), 6 (28.6%) |
| Age (mean ± standard deviation) | 49.4 ± 15.0 |
| Household income | |
| No income | 3 (15.0%) |
| ≤1 minimum wage | 8 (40.0%) |
| More than one minimum wage | 9 (45.0%) |
| Schooling (in years) | |
| 0-4 years | 10 (47.6%) |
| 5-9 years | 3 (14.3%) |
| More than 9 years | 8 (38.1%) |
| Clinical | |
| Clinical form (World Health Organization) | |
| “Paucibacillary” | 2 (9.5%) |
| “Multibacillary” | 19 (90.5%) |
| Time of Diagnosis | |
| <1 year | 11 (52.4%) |
| 2-3 years | 5 (23.8%) |
| 4-7 years | 2 (9.5%) |
| Over 8 years | 3 (14.3%) |
| 4-7 years | 2 (9.5%) |
| Over 8 years | 3 (14.3%) |
| Purely neural: Yes | 1 (4.8%) |
| Presence of sensory alterations in small fibres on neurological evaluation | 20 (95.2%) |
| Presence of sensory alterations in large fibres on neurological evaluation | 7 (35.0%) |
| Neuropathic pain | |
| Nociceptive | 4 (19.0%) |
| Neuropathic | 17 (81.0%) |
The median-of-means for dendritic cells and sub-basal nerve fibres in the control group was used to establish normality. Accuracy means were calculated to measure the diagnostic utility of in vivo confocal microscopy in individuals affected by leprosy. Eye examinations in the leprosy group found the presence of corneal neuromas in 86.7%. The number of dendritic cells was higher than the median for the control group (37/41). This indicates that 90.2% of the eyes of patients examined had a volume of sub-basal fibres lower than the mean for the control group, as shown in Table 2.
| Variables | Patients with leprosy | Control group | P-value |
|---|---|---|---|
| Dendritic cells | |||
| Median (P25 - P75) | 16.7 (10.4-43.7) | 2.62 (0.67-5.67) | <0.001a |
| Sub-basal nerve fibres | |||
| Mean ± standard deviation | 14.341 ± 4.875 | 22.588 ± 3.593 | <0.001 |
a Logarithmic transformation for normalization and application of the parametric test
Cross-tabulation of the in vivo confocal microscopy variables for leprosy provided one statistically significant finding regarding the World Health Organisation’s operational classification and the median number of dendritic cells. Multibacillary individuals presented a median of around two times greater in relation to the number of cells (18.7) compared to paucibacillary individuals (P = 0.035). Although none of the other associations were found to be statistically significant, interpretation of the other data presented is also clinically important [Table 3].
| Clinical Data | Density of cornea fibres (mean ± standard deviation) | P-value | Dendritic Cells Median (P25 - P75) | P-valuea | |
|---|---|---|---|---|---|
| Sex | |||||
| Female | 18,152 ± 6,169 | 0.198 | 9.4 (9.0-10.2) | 0.612 | |
| Male | 19,921 ± 5,197 | 18.7 (12.0-43.7) | |||
| Age (in years) | |||||
| Correlation | 0.0736 | 0.498 | -0.1627 | 0.132 | |
| Clinical form (Word Health Organization) | |||||
| Paucibacillary | 11,975 ± 542 | 0.313 | 9.4 (9.0-10.2) | 0.035 | |
| Multibacillary | 14,597 ± 5,069 | 18.7 (12.0-43.7) | |||
| Clinical form (Madrid) | |||||
| Tuberculoid | 11,975 ± 542 | 0.533 | 9.4 (9.0-10.2) | 0.082 | |
| Borderline | 14,395 ± 5,470 | 27.1 (10.4-56.2) | |||
| Lepromatous-type | 15,469 ± 4,450 | 13.5 (12.5-17.7) | |||
| Time of diagnosis | |||||
| ≤1 year | 13,941 ± 5,485 | 0.579 | 15.6 (9.5-31.2) | 0.663 | |
| >1 year | 14,803 ± 4,160 | 17.7 (11.0-43.7) | |||
| Small fibre sensory function | |||||
| Altered | 14,447 ± 4,978 | 0.545 | 17.7 (10.4-43.7) | 0.191 | |
| Normal | 12,271 ± 458 | 9.1 (8.7-9.5) | |||
| Large fibre sensory function | |||||
| Altered | 13,597 ± 4,316 | 0.504 | 16.7 (10.4-27.1) | 0.523 | |
| Normal | 14,752 ± 5,348 | 19.8 (10.4-45.8) | |||
| Evaluation of neuropathic pain | |||||
| Nociceptive | 15,271 ± 7,121 | 0.554 | 37.5 (11.8-77.1) | 0.134 | |
| Neuropathic | 14,115 ± 4,283 | 16.7 (10.4-31.2) |
aLogarithmic transformation for normalization and application of the parametric test
Discussion
The goals related to the elimination of leprosy established by the World Health Organisation have not yet been met. A study in Nepal found a higher prevalence among males (n = 58) and a mean age of 54.19 years, with 60% lacking schooling, and high levels of poverty. Such findings concur with our data in suggesting that a low level of education and low socio-economic status may cause individuals to be more susceptible to infection.20,21
We identified neuropathic pain in 81.5% of patients, noting that 62% had concluded the period of treatment but were still experiencing pain. This indicates that this symptom merits more extensive investigation, using easy-to-apply tools that may help to treat this neglected common symptom.22-24
In relation to data obtained using in vivo confocal microscopy, we found an increased number of dendritic cells and a reduced number of sub-basal nerve fibres in the individuals with leprosy compared to the control group. These differences were statistically significant (P < 0.001). We identified only one case report evaluating the presence and distribution of nerve thickening in leprosy patients, but the difference in study design makes it difficult to compare this result with ours25 [Figure 1].
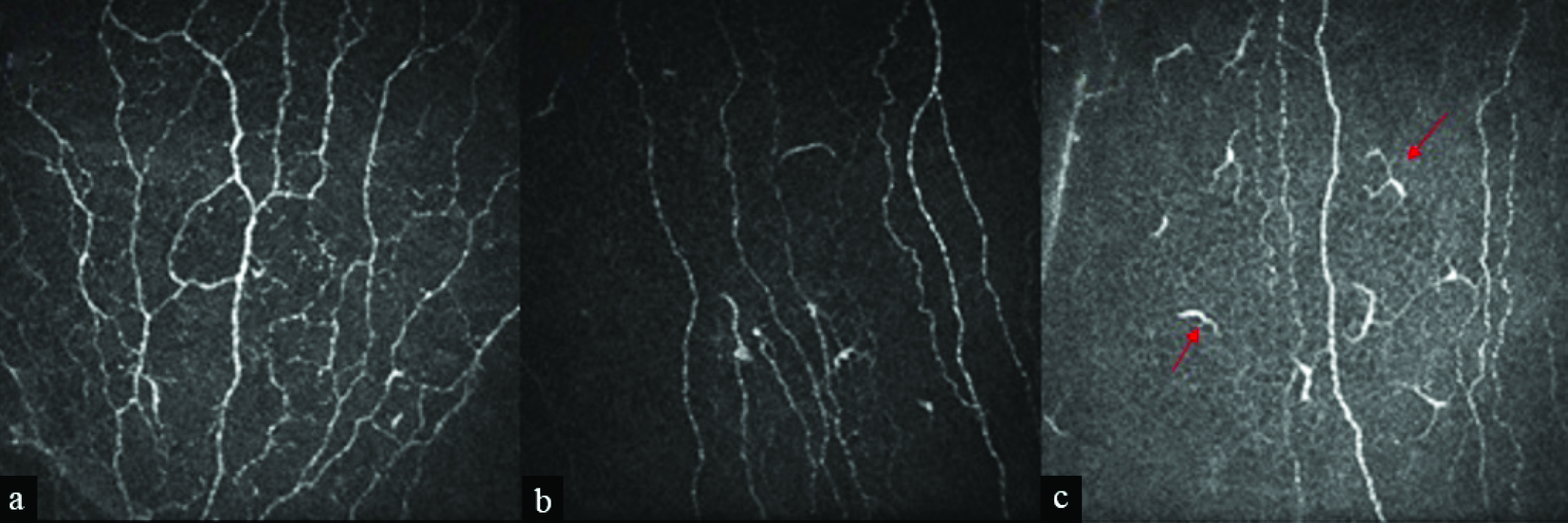
- (a) Nerve fibres preserved in healthy individuals. (b) Reduced numbers of nerve fibres in leprosy patient. (c) Reduced numbers of nerve fibres and the influx of dendritic cells in a leprosy patient. Dendritic cells are marked with red arrows. [×800 total magnification]
Dendritic cells are also known as antigen-presenting cells. Their function is to regulate and connect the innate and acquired immune responses. These cells are widely distributed throughout the body, including the cornea, and are capable of processing and presenting antigens to other immune cells, which initiate the acquired immune response. We believe that this function may explain the statistical significance found in the correlation between the median number of dendritic cells and the clinical presentation of multibacillary because these individuals present a higher number and greater proliferation of Hansen’s bacilli.26,27
Recent studies have shown significant changes in the number of sub-basal layer nerves in the cornea and in its morphology in patients with peripheral neuropathies of various etiologies, such as Sjögren’s syndrome, Crohn’s disease, diabetes and herpes zoster.28-32 Despite the absence of comparative data on leprosy, our results accord with the identification of a reduction in the density of sub-basal nerve fibres described in acute or chronic disorders affecting these structures. We also found a larger number of neuromas and this suggests that, despite the seriousness of the lesion, this insult may be followed by a regenerative process [Figure 2].
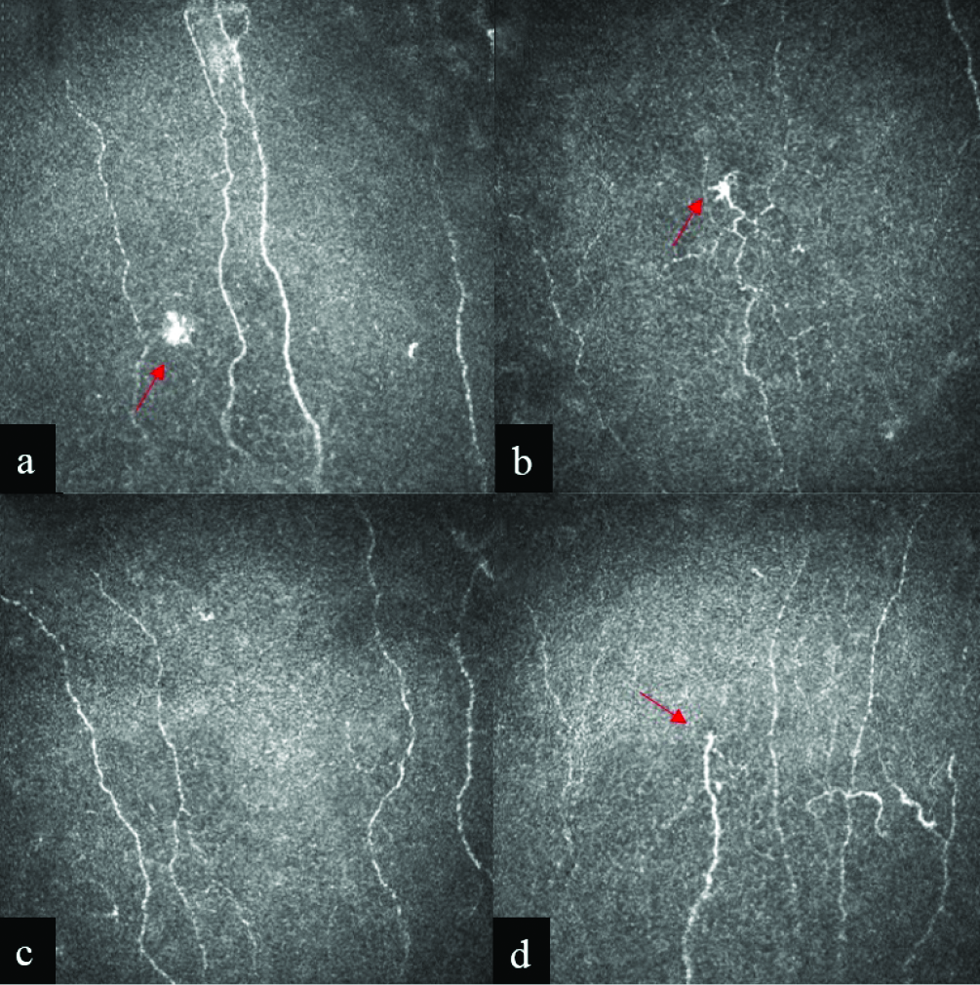
- (a) Confocal microscopy images of nerve fibres in the central region of the cornea; (b) Patients with lepromatous-type Hansen’s cells; (c, d) Patient with borderline type Hansen’s cells. Images suggesting neuroma marked with red arrow. Lens [× 800 total magnification]
Our data facilitate the identification of ocular involvement in leprosy of the kind which may result in signs and symptoms that may lead to disorders that impair vision and even blindness. Small fibre nerve destruction may produce diminished pupillary reactions and reduced accommodation. This may occur early and subtle signs can be identified during clinical evaluation.11
In vivo confocal microscopy of corneal nerves provides an opportunity to study the human tissue type that has the highest density of nerve fibres. This may aid the diagnosis of small fibre neuropathy, which still commonly poses a significant challenge in clinical practice.
Limitations
Although we did not find any association between the in vivo confocal microscopy variables examined and the alterations detected by neurological examination, we believe that Cochet-Bonnet esthesiometry may be able to provide evidence of such an association. It was not, however, possible to carry out such an evaluation in most patients. Even so, the results reported here are of great importance for the characterization and treatment of leprosy.
Conclusion
In vivo confocal microscopy is a useful additional diagnostic tool for the detection of small fibre loss in individuals affected by leprosy. We recommend that it be applied early on in the course of the disease to help curb the effects of neuropathy in such patients.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Sensitive damage in leprosy: A longitudinal and retrospective study. Int Arch Med. 2020;13:1-9.
- [CrossRef] [Google Scholar]
- Small-fiber polyneuropathy in leprosy without skin changes: Study of 17 cases. Arq Neuropsiquiatr. 2003;61:542-6.
- [CrossRef] [PubMed] [Google Scholar]
- Fatores de risco em contatos intradomiciliares de pacientes com hanseníase utilizando variáveis clínicas, sociodemográficas e laboratoriais. Rev Pan-Amaz Saude. 2017;8:23-30.
- [CrossRef] [Google Scholar]
- Clinical aspects of leprosy. Clinics in Dermatology. 2015;33:26-37.
- [CrossRef] [PubMed] [Google Scholar]
- Peripheral nerve biopsy: A tool still needed in the early diagnosis of neural leprosy? Trans R Soc Trop Med Hyg. 2020;114:792-7.
- [CrossRef] [PubMed] [Google Scholar]
- Mechanisms of nerve injury in leprosy. Clin Dermatol. 2015;33:46-54.
- [CrossRef] [PubMed] [Google Scholar]
- Definition and diagnosis of small fiber neuropathy: Consensus from the peripheral neuropathy scientific department of the Brazilian academy of neurology. Arq Neuropsiquiatr. 2018;76:200-8.
- [CrossRef] [PubMed] [Google Scholar]
- In vivo confocal microscopy of corneal nerves in health and disease. Ocul Surf. 2017;15:15-47.
- [CrossRef] [PubMed] [Google Scholar]
- Morphology of corneal nerves using confocal microscopy. Cornea. 2001;20:374-84.
- [CrossRef] [PubMed] [Google Scholar]
- In vivo confocal microscopy of corneal nerves: Analysis and clinical correlation. Semin Ophthalmol. 2010;25:171-7.
- [CrossRef] [PubMed] [Google Scholar]
- Assessing the reproducibility of quantitative in vivo confocal microscopy of corneal nerves in different corneal locations. Cornea. 2013;32:1331-8.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical presentation and diagnosis of neuropathic pain. Rev Dor. 2016;17:S15-9.
- [CrossRef] [Google Scholar]
- Diabetic neuropathy: A focus on small fibres. Diabetes Metab Res Rev. 2019;36:1-4.
- [CrossRef] [PubMed] [Google Scholar]
- Potential use of corneal confocal microscopy in the diagnosis of parkinson’s disease associated neuropathy. Transl Neurodegener. 2020;9:1-10.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- ImageJ. Bethesda, Maryland, USA: National Institutes of Health; http://imagej.nih.gov/ij
- Leprosy stigma & the relevance of emergent therapeutic options. Indian J Med Res. 2020;151:1-5.
- [CrossRef] [PubMed] [Google Scholar]
- From social curse to social cure: A self-help group community intervention for people affected by leprosy in Nepal. J Community Appl Soc Psychol. 2021;31:276-87.
- [CrossRef] [Google Scholar]
- Agreement between the douleur neuropathique in 4 questions and leeds assessment of neuropathic symptoms and signs questionnaires to classify neuropathic pain among patients with leprosy. Am J Trop Med Hyg. 2016;95:756-9.
- [CrossRef] [PubMed] [Google Scholar]
- Diagnosis and medical treatment of neuropathic pain in leprosy. Rev Lat Am Enfermagem. 2016;24:e2731.
- [CrossRef] [PubMed] [Google Scholar]
- Diagnosis and impact of neuropathic pain in leprosy patients in Nepal after completion of multidrug therapy. PLoS Negl Trop Dis. 2018;12:e0006610.
- [CrossRef] [PubMed] [Google Scholar]
- In Vivo Confocal Laser Scanning Microscopy of Corneal Nerves in Leprosy. . 2008;Arch Ophthalmol. 2008;126:282-4.
- [CrossRef] [PubMed] [Google Scholar]
- Corneal antigen-presenting cells. Chem Immunol Allergy. 2007;92:58-60.
- [CrossRef] [PubMed] [Google Scholar]
- The corneal stroma is endowed with a significant number of resident dendritic cells. Invest Ophthalmol Vis Sci. 2003;44:581-9.
- [CrossRef] [PubMed] [Google Scholar]
- Autologous serum tears for treatment of photoallodynia in patients with corneal neuropathy: Efficacy and evaluation with in vivo confocal microscopy. Ocul Surf. 2015;13:250-62.
- [CrossRef] [PubMed] [Google Scholar]
- In vivo confocal microscopy demonstrates a profound increase in immune dendritic cells and decrease in corneal nerves in patients with post-refractive surgery keratoneuralgia. Invest Ophthalmol Vis Sci. 2013;54:3711.
- [Google Scholar]
- In vivo confocal microscopy detects bilateral changes of corneal immune cells and nerves in unilateral herpes zoster ophthalmicus. Ocul Surf. 2018;16:101-1.
- [CrossRef] [PubMed] [Google Scholar]
- Corneal epithelial “neuromas”: A case of mistaken identity? Corne 202039:930-4.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]






