Translate this page into:
Stevens-Johnson syndrome after ChAdOx1 nCoV-19 vaccine
Corresponding author: Pedro Alves da Cruz Gouveia, Department of Internal Medicine, Oswaldo Cruz University Hospital, Pernambuco, Brazil. pedroalves@doctor.com
-
Received: ,
Accepted: ,
How to cite this article: Gouveia PAC, Cavalcanti LNF, Alves LCF, Almeida DM, Barretto LWA. Stevens-Johnson syndrome after ChAdOx1 nCoV-19 vaccine. Indian J Dermatol Venereol Leprol 2022;88:702-702.
Sir,
A 40-year-old black woman received the first dose of the ChAdOx1 nCoV-19 vaccine (AstraZeneca/Fiocruz) and two days later developed asthenia and a skin rash on her trunk and arms. She presented with a 48-hour progressive worsening of the skin rash to a widespread pattern, and she felt pain and irritation in the oral mucosa.
The patient was admitted to Oswaldo Cruz University Hospital, Brazil, with vesicles over confluent erythematous macules, found disseminated throughout the body [Figure 1], and having atypical target lesions on the palms and soles of the feet [Figure 2]. A biopsy of a flaccid blister on the left arm was performed. She also had oral mucosal lesions and had difficulty eating [Figure 3]. Routine laboratory tests were normal, except for inflammation markers such as high C-reactive protein and erythrocyte sedimentation rate.
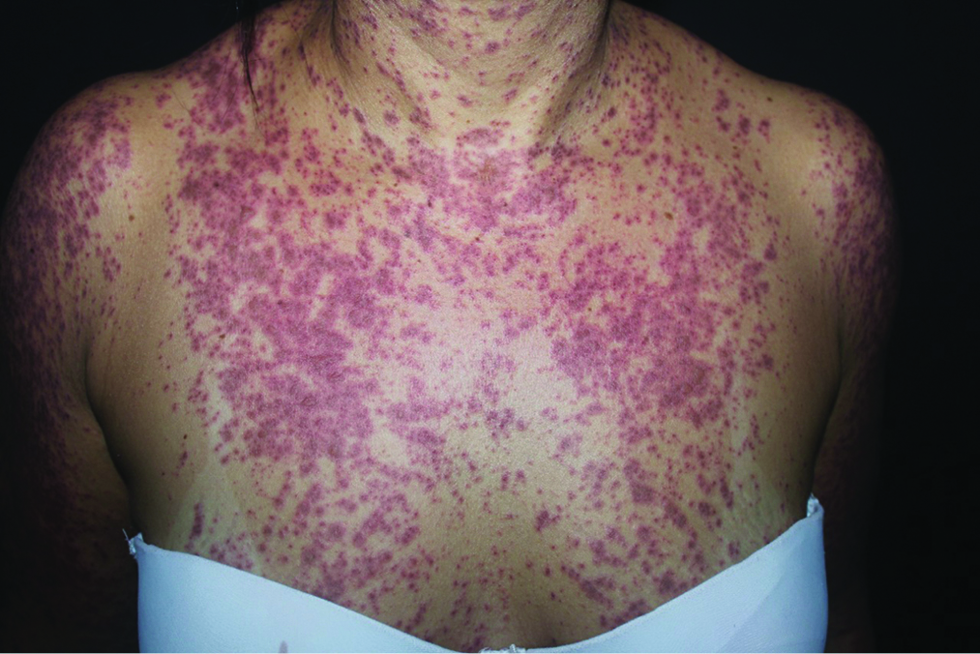
- Vesicles over confluent erythematous macules on the upper trunk
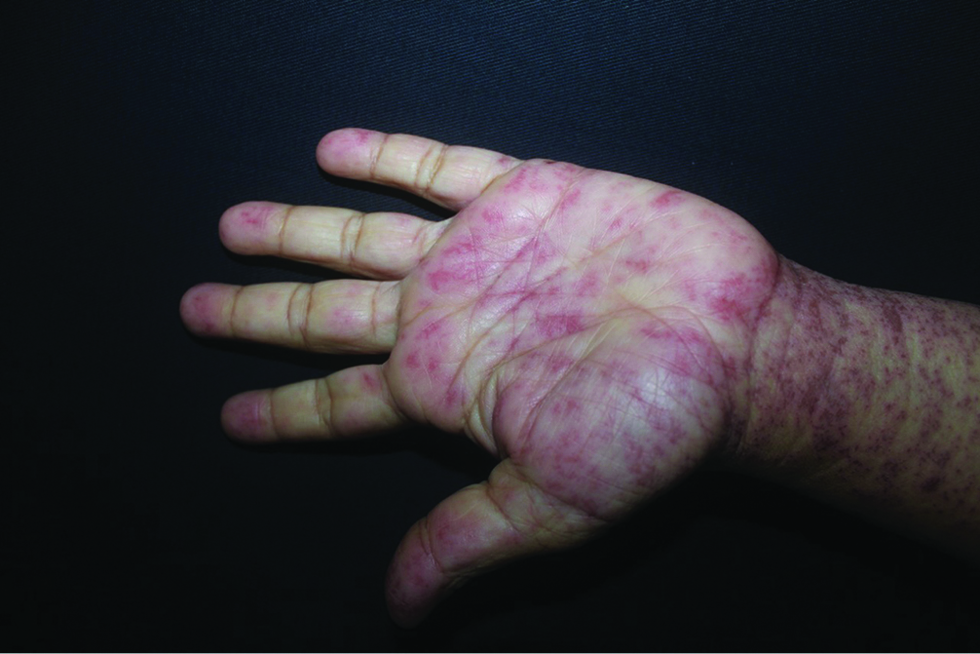
- Atypical target lesions on the palm of the hand
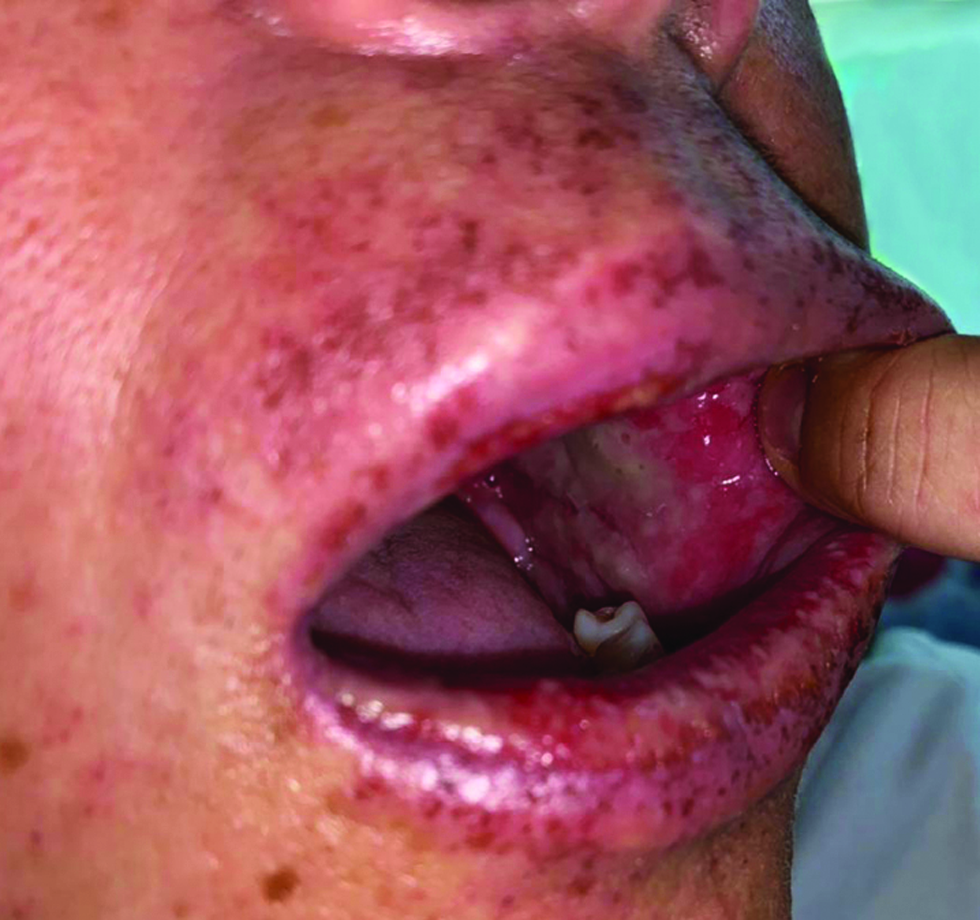
- Erosions on the oral mucosa covered with exudation
She denied taking any medication in the past few weeks as well as having any previous drug reaction or erythema multiforme. To rule out the activity and influence of infections, serological tests were collected for arboviruses, Epstein-Barr virus, HIV, HTLV, viral hepatitis, syphilis, toxoplasmosis, cytomegalovirus, with negative results for markers of acute phase of any infection.
Since there was no other exposure to drugs or infection, a diagnosis of Stevens-Johnson syndrome related to vaccination was made and treatment with prednisone was initiated. On the third day of hospitalization, she returned to an oral diet and vesicles converged to cause detachment of the epidermis, without new lesions. On the fifth day, she was discharged from the hospital with epidermal detachment <10% body surface area [Figure 4]. Skin biopsy results showed keratinocyte necrosis with a detachment of the epidermis and mild lymphocytic interface dermatitis, suggestive of Stevens-Johnson syndrome [Figure 5].
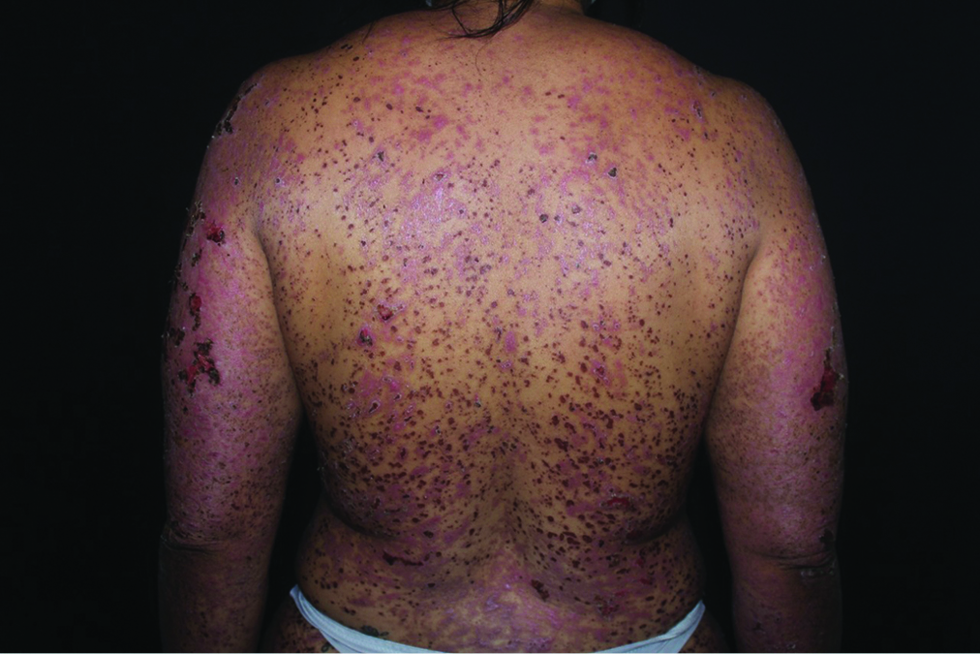
- Epidermal detachment on the back and arms after one week
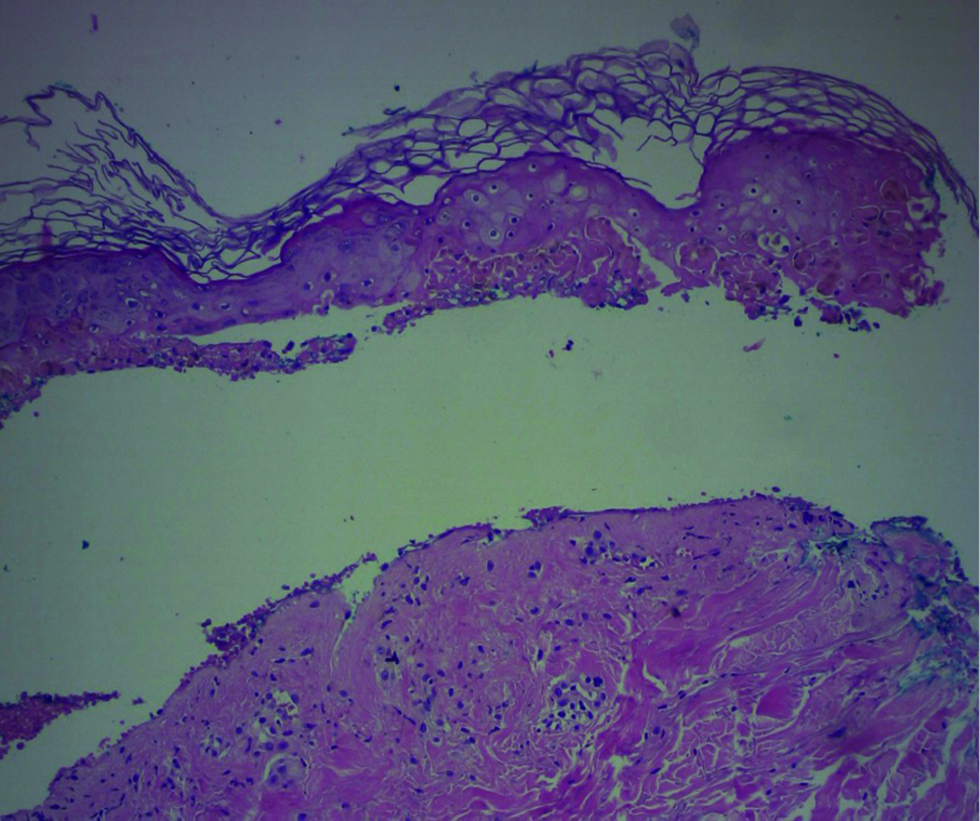
- Histopathology shows necrosis of keratinocytes with detachment of the epidermis accompanying a mild lymphocytic inflammatory infiltrate (HE × 100)
The patient received a letter of recommendation from health authorities advising avoidance of the second dose of the AstraZeneca/FioCruz vaccine. It was advised that the vaccination be completed two months later, with a second vaccine of different technology, such as Pfizer.
Differentiating erythema multiforme with mucous membrane involvement from Stevens-Johnson syndrome is a challenge. The SCAR group study differentiates these two entities according to a clinical classification using the pattern and distribution of lesions. Erythema multiforme has typical targets of acral distribution, whereas Stevens-Johnson syndrome has blisters on macules and flat atypical targets disseminated throughout the body, as happened in the present case. This distinction is used in several studies and in the UK guidelines for Stevens-Johnson syndrome.
Rare cases of post-vaccination Stevens-Johnson syndrome have been described with a questionable correlation. A 60-year-old man presenting with Stevens-Johnson syndrome, like the case reported here, was reported by Dash et al. in India three days after the first dose of the Astra-Zeneca vaccine.1 Bakir et al. described a middle-aged female patient in Saudi Arabia with severe cutaneous and mucosal lesions one week after the dose of the Pfizer COVID-19 vaccine. Lesions affected more than 30% of the body surface area, characterizing toxic epidermal necrolysis.2 And Mansouri et al. reported a patient in Iran with lesions of the oral and vaginal mucosa, and blisters on the palm of her hand three days after the second dose of the Sinopharm vaccine.3
A single dose of ChAdOx1 nCoV-19 vaccine can induce Th1 cell responses and can drive immune responses mediated by CD4+ T cells and CD8+ T cells of cytotoxic phenotypes.4 We hypothesize that the vaccine activates cytotoxic CD8+ T cells against keratinocytes and this results in consequent epidermal detachment from the skin. This cytotoxicity in Stevens-Johnson syndrome occurs via several pathways, such as perforin/granzyme B, granulysin and the Fas/FasL interaction.5
Dermatologists should be aware of this serious adverse effect, despite this being only the second case of Stevens-Johnson syndrome related to the Astra-Zeneca vaccine. The benefits outweigh the risks of this vaccine. Thus vaccination against COVID-19 should not be discouraged.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no Conflict of Interest.
References
- COVID-19 vaccine-induced stevens-johnson syndrome. Clin Exp Dermatol. 2021;46:1615-7.
- [CrossRef] [PubMed] [Google Scholar]
- Toxic epidermal necrolysis post covid-19 vaccination - first reported case. Cureus. 2021;13:e17215.
- [CrossRef] [PubMed] [Google Scholar]
- Stevens-johnson syndrome due to COVID-19 vaccination. Clin Case Rep. 2021;9:e05099.
- [CrossRef] [PubMed] [Google Scholar]
- T cell and antibody responses induced by a single dose of ChAdOx1 nCoV-19 (AZD1222) vaccine in a phase 1/2 clinical trial. Nat Med. 2021;27:270-8.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical and pathogenic aspects of the severe cutaneous adverse reaction epidermal necrolysis (EN) J Eur Acad Dermatol Venereol. 2020;34:1957-71.
- [CrossRef] [PubMed] [Google Scholar]





