Translate this page into:
TEN mimics: Classification and practical approach to toxic epidermal necrolysis-like dermatoses
Corresponding author: Prof. Eman Salah, Department of Dermatology, Venereology & Andrology, Faculty of Medicine, Zagazig University, Egypt. esmohamed@zu.edu.eg
-
Received: ,
Accepted: ,
How to cite this article: Salah E. TEN mimics: Classification and practical approach to toxic epidermal necrolysis-like dermatoses. Indian J Dermatol Venereol Leprol 2023;89:337-46.
Abstract
Toxic epidermal necrolysis (TEN) is an acute life-threatening dermatologic emergency. However, many dermatoses can present with a TEN-like eruption. Those “TEN-mimics” are a true diagnostic challenge and an alarming differential diagnosis to such a serious condition. Herein, we will expose and classify the landscape of TEN-mimics. Also, the key differentiating clinical and/or laboratory points will be highlighted to help an accurate diagnosis of either a TEN or a TEN-like presentation.
Keywords
Toxic epidermal necrolysis
Lupus
Nikolsky’s signs
SSSS
AGEP
Introduction
Toxic epidermal necrolysis (TEN) is a life-threatening T-cell mediated hypersensitivity reaction mostly to certain medications.1 The high-risk drugs include anti-epileptics, cotrimoxazole, sulfonamides and allopurinol.2,3 It takes between one and eight weeks to develop a TEN reaction after the 1 administration of the triggering medication and only a few hours in cases of drug re-intake.1,4 According to the classification by Bastuji-Garin et al., the features of Stevens-Johnson syndrome (SJS) can overlap with TEN.5
Toxic epidermal necrolysis may start with a maculopapular, purpuric or atypical targetoid rash. Eventually, cutaneous tenderness, blistering and denudation with a positive Nikolsky’s sign supervene. Painful erosive mucositis occurs in most patients with oral involvement in up to 100% of cases. Sepsis with multi-organ failure is the most common cause of death. Survivors may develop cutaneous scarring and/or ocular complications.1
The hallmark histopathological finding in TEN is “keratinocyte necrosis.” In the well-established lesions, full-thickness epidermal necrosis and sub-epidermal bullae can be seen. A scant perivascular lymphohistiocytic infiltrate with eosinophils is present in the superficial dermis.6
The standard management for TEN involves cessation of the offending agent and supportive therapy in a burn unit or an intensive care facility. Intravenous immune globulin (IVIG) with or without systemic corticosteroids has been used with conflicting results. In refractory cases; plasmapheresis could help.7
It may appear to some that a definite diagnosis of TEN is easily made based on the above-mentioned clinical and pathological presentation. However, many other dermatoses of an autoimmune or even infective aetiology can simulate TEN syndrome at different stages of evolution. Misdiagnosis of such conditions can potentially result in devastating outcomes.
Recently, a TEN-like presentation for different disorders or “TEN-mimics” has been increasingly reported in the literature. Herein, we will classify [Table 1] the landscape of TEN-mimics. Also, the key differentiating clinical and/or laboratory points [Table 2] will be highlighted to help an accurate diagnosis of either a TEN or another dermatosis with a TEN-like presentation.
| Etiology | TEN-mimics |
|---|---|
| 1-Infections | a-Bacterial/Bacterial-toxin: |
| Staphylococcal scalded skin syndrome (SSSS) Toxic shock syndrome (TSS) Purpura fulminans |
|
| b-Viral: | |
| Coxsackievirus-induced severe mucocutaneous disease Chikungunya fever Epstein-Barr virus) EBV (positive T-cell lymphoproliferative disease |
|
| 2-Immune-mediated | Lupus erythematosus Dermatomyositis Pemphigus vulgaris Paraneoplastic pemphigus Lichen Planus Pemphigoides Graft−versus−host−disease (GVHD) Inflammatory epidermolysis bullosa acquisita Inflammatory epidermolysis bullosa |
| 3-Drug-induced | Acute generalised exanthematous pustulosis (AGEP) Generalised bullous fixed drug eruption Drug-induced linear IgA bullous dermatosis Drug reaction with eosinophilia and systemic symptoms (DRESS) Contact dermatitis to dendrimers |
| 4-Metbolic | Pseudoporphyria |
| 5-Burn | a-Chemical: |
| Boric acid intoxication Bleaching agents |
|
|
b-Thermal: Fire or hot liquids injury |
| TEN/TEN-mimics | Cause | Age/Sex | Cutaneous lesions | Nikolsky’s sign | Mucosal lesions | Systemicsymptoms | Laboratory/pathology findings | Treatment |
|---|---|---|---|---|---|---|---|---|
| TEN | Drug-induced | Any age group, more in females | Epidermal detachment according to the classification of Bastuji-Garin et al. | +Ve | Yes & Painful |
Fever, cardio-pulmonary, renal, and gastrointestinal dysfunction | Keratinocyte necrosis “Hallmark” |
Supportive, IVIG, systemic steroids, others |
| Staphylococcal scalded skin syndrome (SSSS) | staphylococcus | More in infants & children |
TEN-like, peri-oral & peri-orbital crusts “Potato chips” desquamation | +Ve | No | Fever | Subcorneal cleavage without keratinocyte necrosis Frozen sections & Tzanck smear for a quicker diagnosis Isolation of the organism from colonisation sites |
Anti-Staph antibiotics |
| Toxic shock syndrome | TSST-1 | More in menstrual women | TEN-like with marked septic presentation | −Ve | No | Fever, hypotension, internal organs dysfunction | Blood culture Non-specific histopathology |
Anti-shock measures Antibiotics IVIG |
| Purpura fulminans | Endotoxin | Any age group | TEN-like, retiform purpura | +Ve | No | Multiorgan failure | DIC workup Blood culture Bacterial 16S rRNA gene sequencing Skin necrosis & embo-lism with microthrombi |
Immediate broad-spectrum antibiotics |
| Coxsackievirus-induced severe mucocutaneous disease | Coxsackie-B virus | Any age group | TEN-like | −Ve | Yes | Ocular | Viral culture PCR for throat swab |
Supportive |
| Chikungunya fever | Chikungunya virus | Any age group, endemic in some countries | TEN-like | −Ve | No or minimal | Fever | Anti-Chikungunya IgM nearly 5 days following symptoms Extensive keratinocyte necrosis or ballooning degeneration |
Supportive |
| Epstein-Barr virus positive T-cell lymphoproliferative disease | Epstein-Barr virus | Any age group | TEN-like | −Ve | No | Fever, cough, LNs, HSM, jaundice | Skin infiltration by atypical CD8+ lymphocytes, in situ hybridisation, EBV serology, PCR | Supportive |
| Lupus erythematosus | Autoimmune | Any age group, more in females, no drug causality | TEN-like, more on sun-exposed sites | −Ve | Yes & Painless |
Of SLE or SCLE, should be considered in a known case of lupus | Serology, complement levels, histopathology & DIF of lupus | Systemic steroids ± others |
| Dermatomyositis | Autoimmune MDA5-DM | Any age group | TEN-like | −Ve | No | Exacerbation of DM manifestations | Anti-MDA5, DM laboratory findings | Systemic steroids ± others |
| Pemphigus vulgaris | Autoimmune | Any age group | TEN-like | +Ve | Yes | No | Acantholysis, IF deposits of PV, anti-Dsg by ELISA | Systemic steroids ± others |
| Paraneoplastic pemphigus | Autoimmune | More in old adults | TEN-like | −Ve | Yes | Underlying tumour e.g., NHL | Acantholysis, anti-plakin & anti-Dsg antibodies | Systemic steroids ± others, treatment of related tumour |
| Lichen Planus Pemphigoides | Autoimmune | Any age group | TEN-like, both LP & BP lesions | −Ve | Yes | No | IF findings of BP | Systemic steroids ± others |
| Graft-versus-host-disease | Autoimmune | Any age group, after hematopoietic stem cell transplantation | TEN-like | −Ve | No | Jaundice, diarrhoea, ileus | Abnormal LFT with hyperbilirubinemia (>15mg/dL), ch.ch. GIT & liver pathology, Analysis of chimerism | Systemic steroids ± others |
| Inflammatory epidermolysis bullosa acquisita | Autoimmune | Any age group | TEN-like, with heterogeneous, overlapping or alternating clinical lesions of EBA, DH & BP | +Ve | Yes | No | Absent keratinocytes necrosis DIF, salt-split skin test is diagnostic |
Systemic steroids |
| Acute generalised exanthematous pustulosis (AGEP) | Drug-induced mainly | Any age group | Rapid onset of TEN-like lesions, pustules, facial oedema | +Ve | Yes in 20% of cases | No | Subcorneal /intra-epidermal pustules, marked dermal oedema, patch test | Stop the drug, Systemic steroids ± others |
| Generalised bullous fixed drug eruption | Drug-induced | Any age group | TEN-like, recurrent blistering, PIH | −Ve | Mild or absent | No | TEN-like histopathological changes | Stop the drug, Systemic steroids ± others |
| Drug-induced linear IgA bullous dermatosis | Drug-induced especially with vancomycin and phenytoin |
Any age group | TEN-like, annular, or polycyclic configuration of blisters | +Ve or −Ve |
No | No | DIF from perilesional skin with linear deposition of IgA along the basement membrane zone is diagnostic | Stop the drug, dapsone, Systemic steroids ± others |
| Drug reaction with eosinophilia and systemic symptoms (DRESS) | Drug-induced but with ch.ch. delayed presentation after drug intake | Any age group | urticarial, maculopapular, vesicles, bullae, pustules, purpura, target lesions, facial oedema, cheilitis, erythroderma and TEN-like | −Ve | Rare | Hepatitis, pneumonitis, myocarditis, pericarditis, nephritis, and colitis | Eosinophilia, lymphocytosis with atypical lymphocytes and other internal organ dysfunctions | Stop the drug, Systemic steroids ± others |
| Contact dermatitis to dendrimers | Drug-induced | Adults with history of contact with dendrimers | TEN-like | +Ve | No | None | Patch test, but not advised | Supportive |
| Pseudoporphyria | Metabolic | Any age group, with end-stage-renal disease on haemodialysis | TEN-like | −Ve | No | Chronic renal failure | Detection of porphyrins in serum or blister fluid, festooned dermal papillae with perivascular PAS reactive material in skin biopsy | Supportive |
| Boric acid intoxication | Acid Intoxication |
Mostly in children and infants | TEN-like, diffuse ‘boiled lobster’ skin | +Ve | No | Disturbed conscious level, tachycardia, abnormal breathing sounds, blue-green vomitus, body stiffness, fever | Absent keratinocyte necrosis Elevated serum boric acid level (normal <100 mcg/l) | Supportive ± haemodialysis |
| Bleaching agents | Topical bleaching compounds | Adults, more in females | TEN-like | −Ve | No | No | None | Supportive |
| Thermal | Fire or hot liquids Accidentally or intentionally in case of child abuse | Any age group | TEN-like, charred hairs in fire injuries, glove, and stocking with “donut sign” in immersion burns | −Ve | No | Fire injury: shock. carbon monoxide poisoning, stridor and respiratory failure | Carboxy-haemoglobin levels in suspected fire/inhalation burns | Supportive, immersion burns indicate physical abuse |
+Ve; Positive, -Ve; Negative, TEN; toxic epidermal necrolysis, IVIG; Intravenous immune globulin, TSST-1; toxic shock syndrome toxin-1, DIC; disseminated intravascular coagulation, PCR; polymerase chain reaction, EBV; Epstein-Barr virus, SLE; systemic lupus erythematosus, SCLE; subacute cutaneous lupus erythematosus, DIF; direct immunofluorescence, DM; dermatomyositis, MDA5; melanoma differentiation antigen-5, IF; immunofluorescence, PV; pemphigus vulgaris, ELISA; enzyme-linked immunoassay, NHL; non-Hodgkin lymphoma, Dsg; desmoglein, BP; bullous pemphigoid, LFT; liver function tests, GIT; gastrointestinal tract, EBA; epidermolysis bullosa acquisita, DH; dermatitis herpetiformis, PIH; post inflammatory hyperpigmentation, PAS; periodic acid Schiff.
The landscape of TEN-mimics
Proposed classification [Table 1]
Infections
Bacterial/Bacterial-Toxin:
Staphylococcal scalded skin syndrome (SSSS)
Staphylococcal scalded skin syndrome (SSSS) is caused by an infection with exfoliative toxin-producing Staphylococcus aureus. Typically, SSSS is seen in infants and children however, it can occur in susceptible adults.8
SSSS starts with fever and irritability. Then, the skin becomes abruptly tender, erythematous with sheets of a TEN-like epidermal detachment with a characteristic “potato chip desquamation”9 [Figure 1a] mainly at the mid-face and flexural sites associated with a positive Nikolsky’s sign. Within 24 hours, characteristic perioral and/or periorbital crusts can be seen without mucosal involvement. With appropriate treatment, the skin is expected to heal with no scarring within two weeks.8,10 The main histopathological finding in SSSS is a sub-corneal cleavage. Once a diagnosis of SSSS is established, anti-staphylococcal antibiotic therapy should be given immediately.11
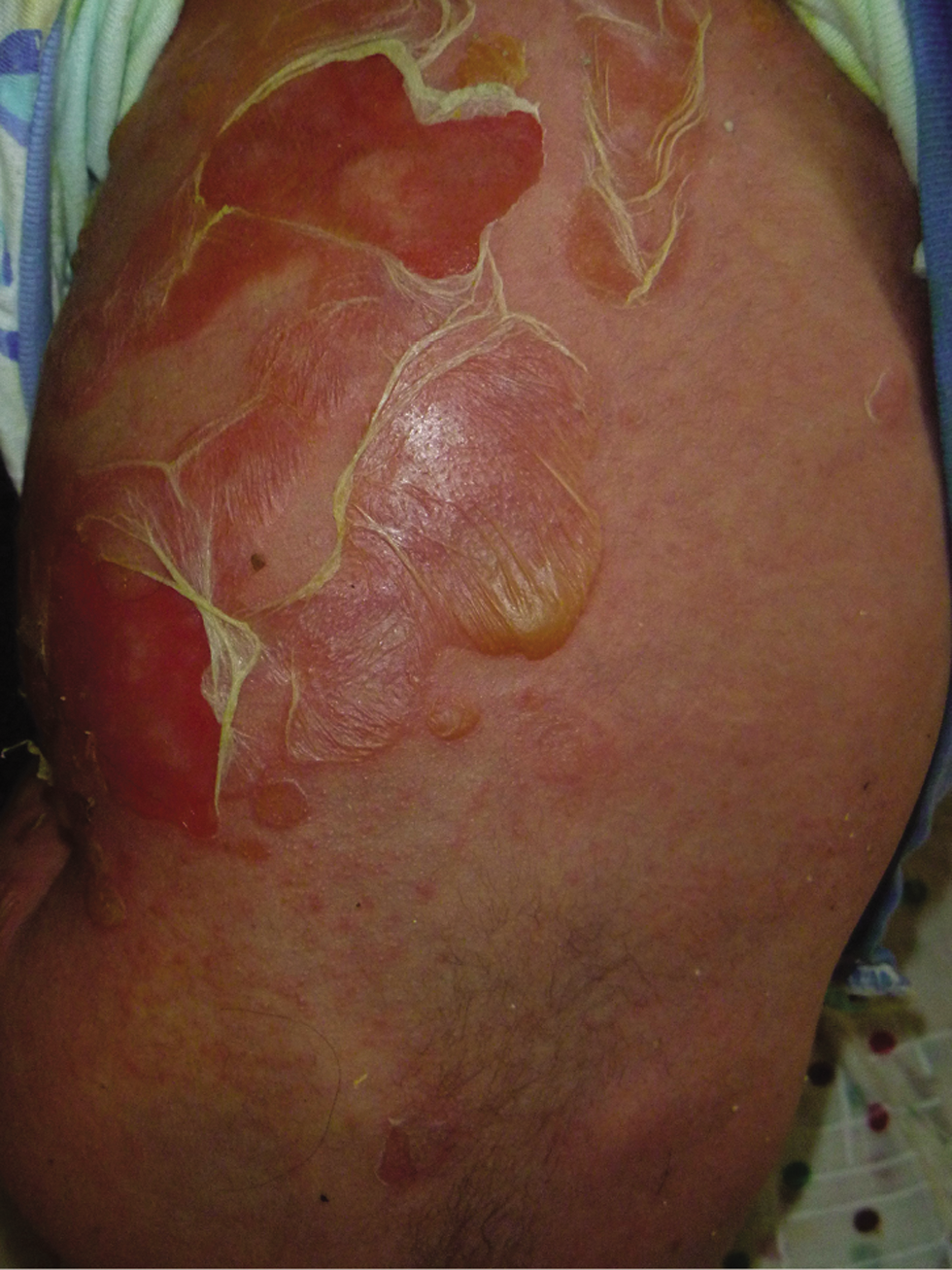
- Toxic epidermal necrolysis-mimics A-Staphylococcal scalded skin syndrome (SSSS) in a 6-months infant
Toxic shock syndrome (TSS)
Toxic shock syndrome (TSS) presents with a high fever of more than 38.9C, hypotension, the involvement of at least three organ systems as well as diffuse erythroderma and desquamation.12 Menstrual TSS accounts for most reported cases, but non-menstrual TSS cases have increased.13 TSS is mainly caused by Staphylococcus aureus that can produce toxic shock syndrome toxin 1 (TSST-1) and by toxigenic strains of Streptococcus pyogenes rarely.14 Blood culture is usually positive in streptococcal TSS, and an identifiable source of soft tissue infection can be found. In staphylococcal TSS, the blood culture is only positive in <5% of menstrual cases and in 50% of non-menstrual cases.15 The histopathological findings are non-specific without evidence of infectious organisms or necrotic keratinocytes.16 Treatment of TSS involves anti-shock measures, broad-spectrum antibiotics and IVIG.15
Purpura fulminans (PF)
Purpura fulminans is a haematological emergency. It is frequently associated with acute disseminated intravascular coagulation (DIC) secondary to sepsis by endotoxin-producing bacteria. The main features of (PF) include tissue necrosis and small vessel thrombosis.17 Initially (PF) presents with well-demarcated erythematous macules with irregular central areas of blue-black haemorrhagic necrosis.18
Epidermal necrolysis secondary to the thrombotic and haemorrhagic cutaneous infarction in (PF) might be confused with TEN, especially with a positive Nikolsky’s sign. Moreover, both syndromes have been reported simultaneously or successively.19 Multi-organ failure is very common.18 Prompt initiation of broad-spectrum antibiotics is the main treatment.17
Viral:
Coxsackievirus-induced severe mucocutaneous disease
Type B5 Coxsackievirus has been reported to induce TEN-like skin lesions. The eruption may present with generalised purpura and maculopapular/vesicular lesions. Mucosal lesions include conjunctival hyperaemia, and oral ulcers with haemorrhagic crusts on the lips and/or genitals. Skin lesions heal with hyperpigmentation and onychomadesis may supervene. The disease can result in significant corneal opacities.20 The histopathological changes mimic those of TEN.21,22
Chikungunya fever
An acute viral infection caused by the chikungunya virus. The virus can be transmitted via the Aedes aegypti mosquito to humans. There are several reported outbreaks of chikungunya fever in India with a TEN-like presentation. The histopathological findings are overlapping with TEN syndrome. The extensive keratinocyte necrosis or ballooning degeneration by the direct spreading of the virus into the skin, or via endothelial injury may explain the TEN-like features. The treatment of TEN-like chikungunya fever is only supportive in nature, and is associated with a favourable prognosis.23,25
Epstein-Barr virus) EBV (positive T-cell lymphoproliferative disease
Sharma et al. have described a TEN-like eruption in an infant with EBV-positive T-cell lymphoproliferative disease associated with haemophagocytic lymphohistiocytosis. Initially, the patient presented with fever, cough, maculopapular, jaundice, hepatosplenomegaly, and lymphadenopathy. Then, the rash became tender with extensive bullous eruptions and skin detachment.26 The histopathological findings included epidermal necrosis, basal layer vacuolar changes with apoptotic cells and dermo/epidermal infiltrating lymphocytes with irregular nuclei. Many of the lymphocytes were CD8 + T cells with positive staining for in situ hybridisations of EBV-encoded small RNA (EBER). Serum IgM and IgG antibodies against EBV were positive. The virus was isolated using PCR from the blister fluid. Bone marrow biopsy demonstrated histiocytosis and further infiltration by T-lymphocytes positive for EBER.26
Earlier to this report, Kawachi et al., have reported a patient demonstrating a similar clinical presentation but with classic TEN-like pathological findings associated with prominent infiltration by EBV-infected CD8 + T-lymphocytes.27
Immune-mediated
Lupus erythematosus:
Acute syndrome of apoptotic pan-epidermolysis (ASAP) is a new term that was 1st proposed by Ting et al. to designate clinical entities characterised by acute and massive cleavage of the epidermis resulting from hyperacute epidermal basal cell apoptotic injury.28 According to Sontheimer’s classification for vesiculobullous lesions occurring in the setting of lupus erythematosus, both acute cutaneous lupus or systemic lupus erythematosus (SLE), subacute cutaneous lupus erythematosus (SCLE), and even SLE patients who are not presenting with the lupus-specific skin lesions can mimic TEN,29 because of an extensive ASAP. Furthermore, TEN-like cutaneous lupus erythematosus is one of the clinical criteria for the diagnosis of SLE according to the Systemic Lupus International Collaborating Clinics classification system.30
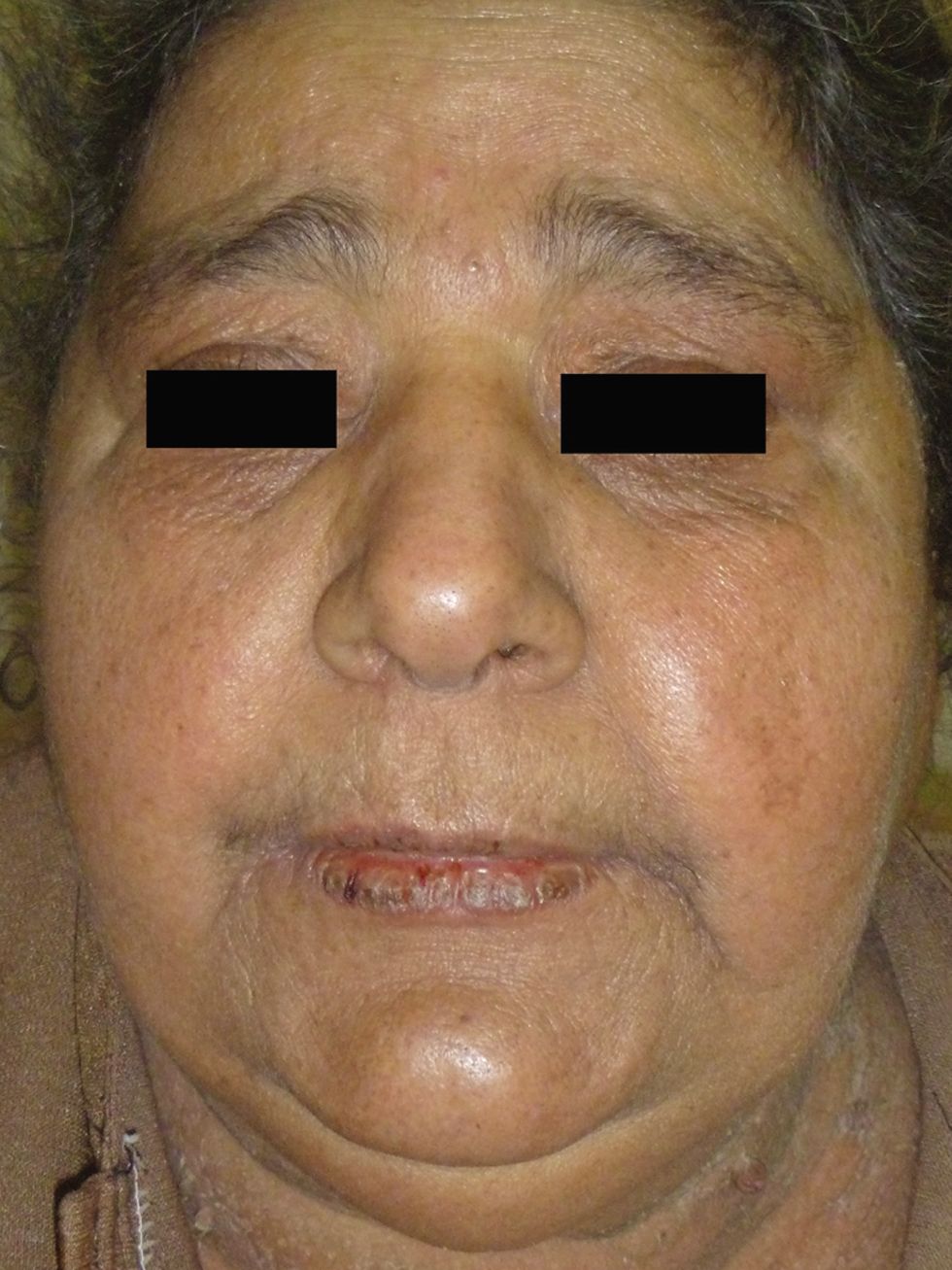
The TEN-like rash of lupus is clinically indistinguishable from the drug-induced TEN unless there is a high index of suspicion.31 However, the lack of a triggering medication, the subacute evolution of the TEN-like lesions over weeks, the photo-distribution [Figure 1b] of the TEN-like eruption (although it may also may occur in sun-protected sites), the presence of painless oral ulcers, associated malar or discoid rash, are all suggestive of TEN-like lupus. The serological profile including ANA is highly valuable to support the clinical suspicion of TEN-like lupus.31 The histopathological findings include both epidermal necrosis and features of cutaneous lupus.32 Direct immunofluorescence demonstrates positive IgG, IgM and C3 deposition in the basement membrane zone in a granular pattern.33 The most commonly reported treatments for TEN-like lupus are systemic corticosteroids with or without hydroxychloroquine, IVIG and mycophenolate mofetil.34
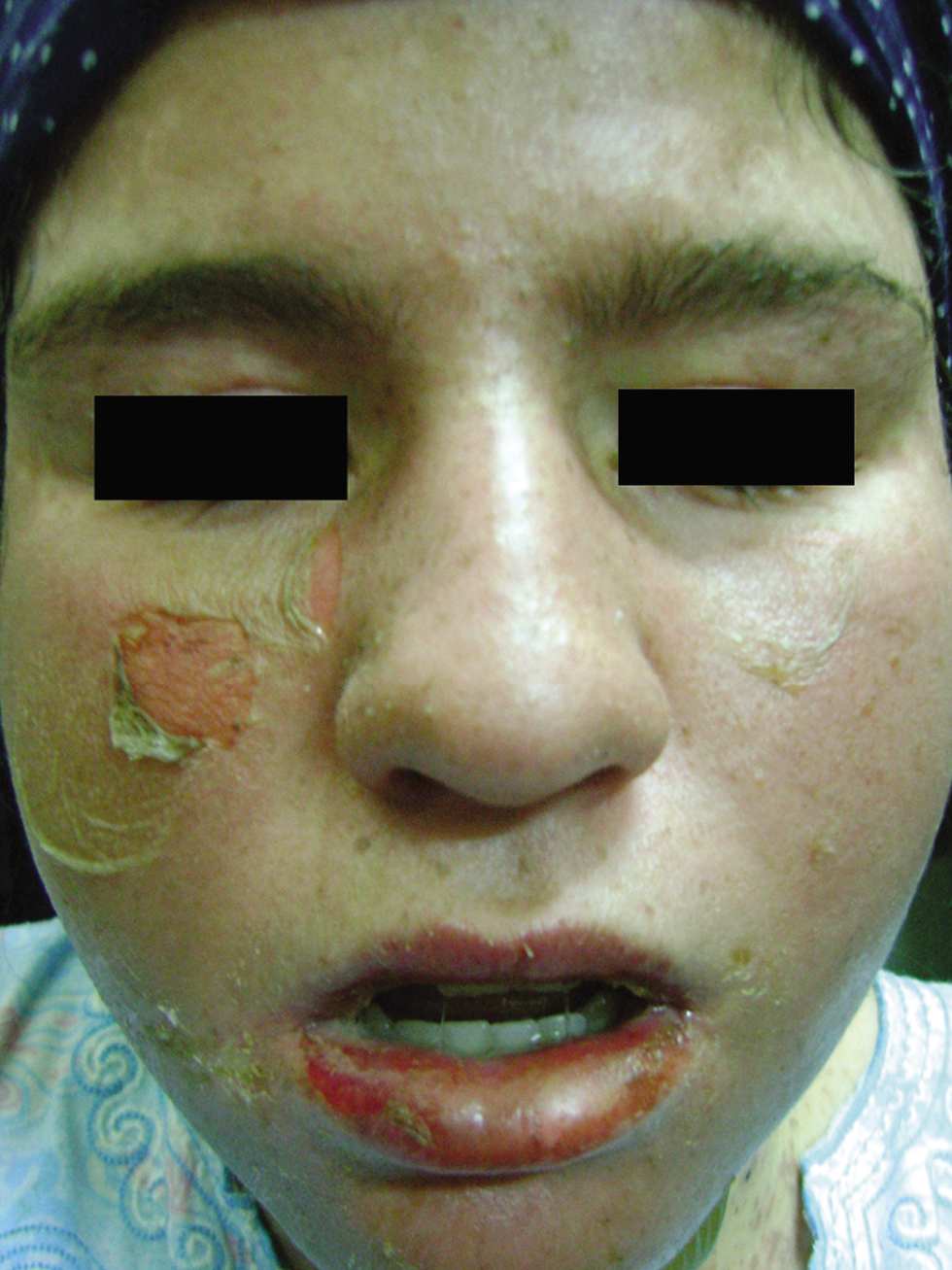
- Toxic epidermal necrolysis-like lupus Note the predilection to the sun-exposed sites
Dermatomyositis:
Kushnir-Grinbaum et al. have reported a patient with anti-melanoma differentiation antigen-5 (MDA5) dermatomyositis and a TEN-like skin eruption. The new TEN-like rash was mostly triggered by the patients’ abrupt quitting of prednisone therapy. However, Nikolsky’s sign was negative, and the mucous membranes were uninvolved. Skin biopsy revealed similar features to TEN.35,37
Pemphigus vulgaris (PV):
Wakumoto-Nakashima et al. have reported a rare presentation of PV associated with TEN-like keratinocytes necrosis. However, the patient also demonstrated lesional intercellular IgG and C3 by direct immunofluorescence (IF) associated with high serum anti-Dsg1 and anti-Dsg3 antibodies by ELISA.38
Paraneoplastic pemphigus (PNP):
There are five main clinicopathological presentations of PNP as described by Nguyen et al. 39 Additionally, a TEN-like presentation of PNP is a novel distinctive subtype.40 It is critical to differentiate true TEN from TEN-like PNP as the latter is associated with a greater mortality rate. Severe mucositis and keratinocyte necrosis with interface dermatitis are the main overlapping features between the two conditions. Also, the delayed appearance of frank acantholysis in PNP and the possibility of PNP preceding the occurrence of internal malignancy might add more confusion.40
In PNP, immunoblotting for envoplakin (EP) or periplakin (PP) is almost 100% sensitive, but with some false-positive results.41 IIF on rat bladder is commonly used to confirm PNP, given a recorded specificity near 100%; however, SJS/TEN sera also can react with rat bladder.42
Lichen planus pemphigoides (LPP):
Lichen planus pemphigoides (LPP) is an immunobullous disorder that is characterised by a concomitant eruption of bullous pemphigoid (BP) and lichen planus (LP) lesion.43 Immunofluorescence studies show BP findings with linear deposition of IgG and C3 along the BMZ. Circulating autoantibodies to BP180 is another distinguishing feature.44
Ondhia et al., have reported a patient with LPP with extensive TEN-like denuded skin and mucosal ulcers. A skin biopsy from the denuded skin revealed full-thickness epidermal necrosis while a biopsy from an intact blister showed a sub-epidermal split with a lichenoid inflammatory pattern. Direct and indirect IF studies revealed typical BP findings.44
Graft-versus-host-disease (GVHD):
Four clinical stages of cutaneous acute GVHD have been defined of which stage IV is characterised by a TEN-like dermo-epidermal detachment in addition to gastrointestinal and hepatic impairment.45,46 The differentiation of a TEN-like acute GVHD and true TEN is challenging as both may coexist. In GVHD, skin biopsies can show dyskeratotic keratinocytes, lymphocyte exocytosis, basal cell necrosis, depletion of Langerhans cells and satellite lymphocytes next to the dyskeratotic keratinocytes.47,49
Inflammatory epidermolysis bullosa acquisita (EBA):
Epidermolysis bullosa acquisita (EBA) of the immunopathological type (EBA-IP) has a heterogeneous inflammatory and/or non-inflammatory phase. The inflammatory phase may mimic bullous pemphigoid (BP), mucosal pemphigoid or dermatitis herpetiformis (DH) and the non-inflammatory mechanobullous phase of EBA displays skin fragility, blistering, scarring and milia at the sites of trauma.50
Madan et al. have described a rare presentation of inflam-matory EBA mimicking clinically both TEN and DH. DIF revealed linear basement membrane zone (BMZ) staining with IgG, IgA, IgM and C3. Salt-split skin test showed staining at the base of the blister, favouring the diagnosis of EBA.51
Drug-induced
Acute generalised exanthematous pustulosis (AGEP):
Acute generalised exanthematous pustulosis (AGEP) is a drug-induced cutaneous adverse reaction. The onset of AGEP is rapid, often occurring hours to days after drug exposure.52 Clinically, it is characterised by fever and the sudden eruption of numerous, tiny, non-follicular, pustules on oedematous erythematous skin. This eruption tends to be generalised with a predilection for the face and/or flexures. Mucosal involvement occurs in only 20% of cases.53 Atypical lesions in AGEP may include marked facial oedema, purpura, atypical target lesions and skin blisters.54 Occasionally, a coalescence of pustules occurs resulting in skin peeling resembling TEN. This effect is often referred to as a positive “pseudo” Nikolsky’s sign.55 The typical histopathology of AGEP shows spongiform sub-corneal and/or intra-epidermal pustules, often-marked oedema of the papillary dermis and perivascular infiltrate with neutrophils and exocytosis of some eosinophils.54 Patch testing with the causative drug is often helpful. Generally, AGEP is a self-limiting reaction that heals within two weeks following the withdrawal of the trigger.53
Generalised bullous fixed drug eruption (FDE):
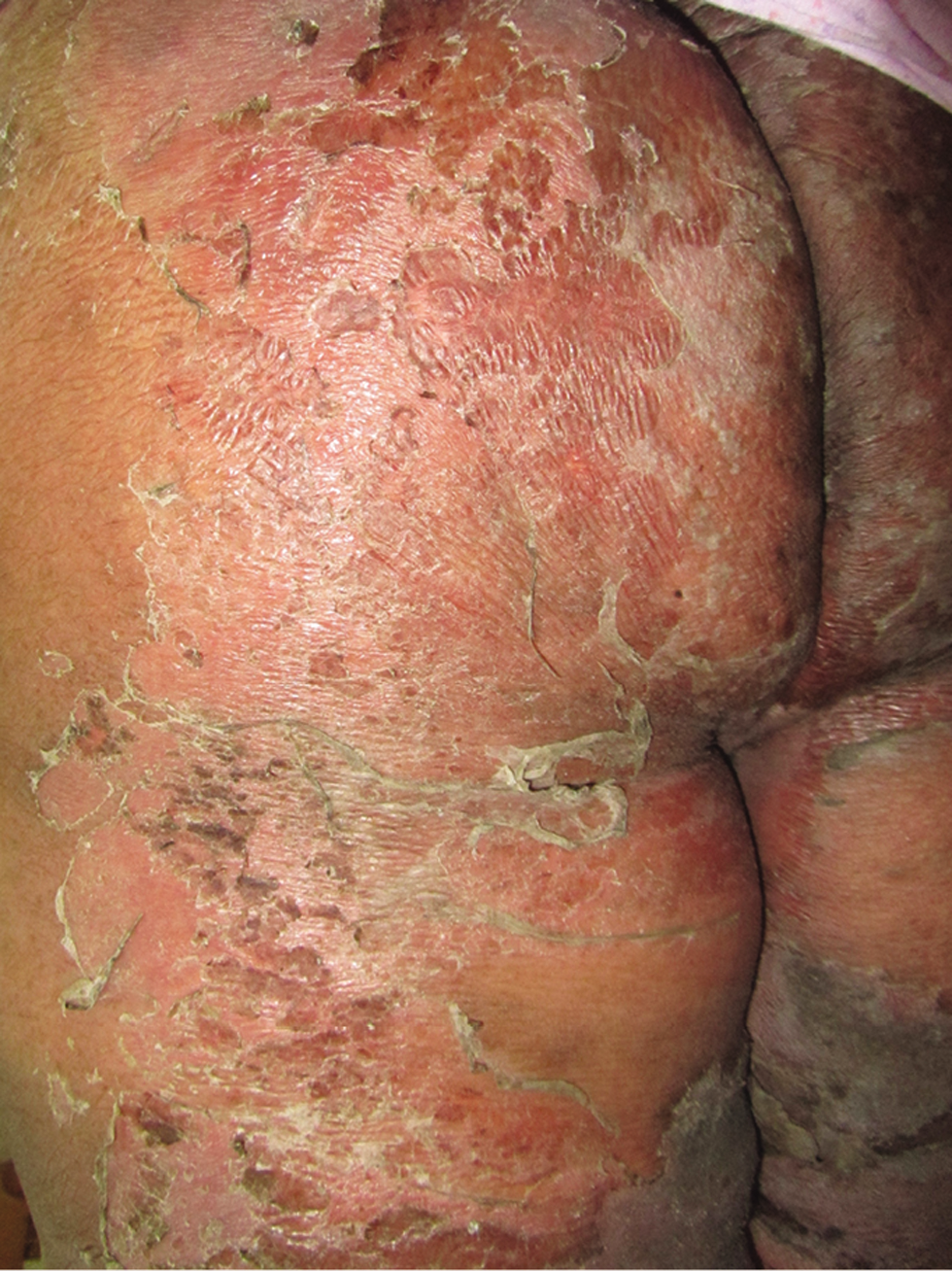
Fixed drug eruption (FDE) is characterised by the recurrence of the skin eruption at the same sites whenever the same drug is re-administered. The lesions usually develop within 30 minutes to 8 hours after drug intake. This eruption appears as well-defined round or oval violaceous plaques associated with itching; however, it can develop generalised blistering or extensive skin detachment simulating TEN.56 Lesions mostly heal with residual post-inflammatory hyperpigmentation [Figure 1c]. The pathological findings in FDE can be indistinguishable from TEN.57,58
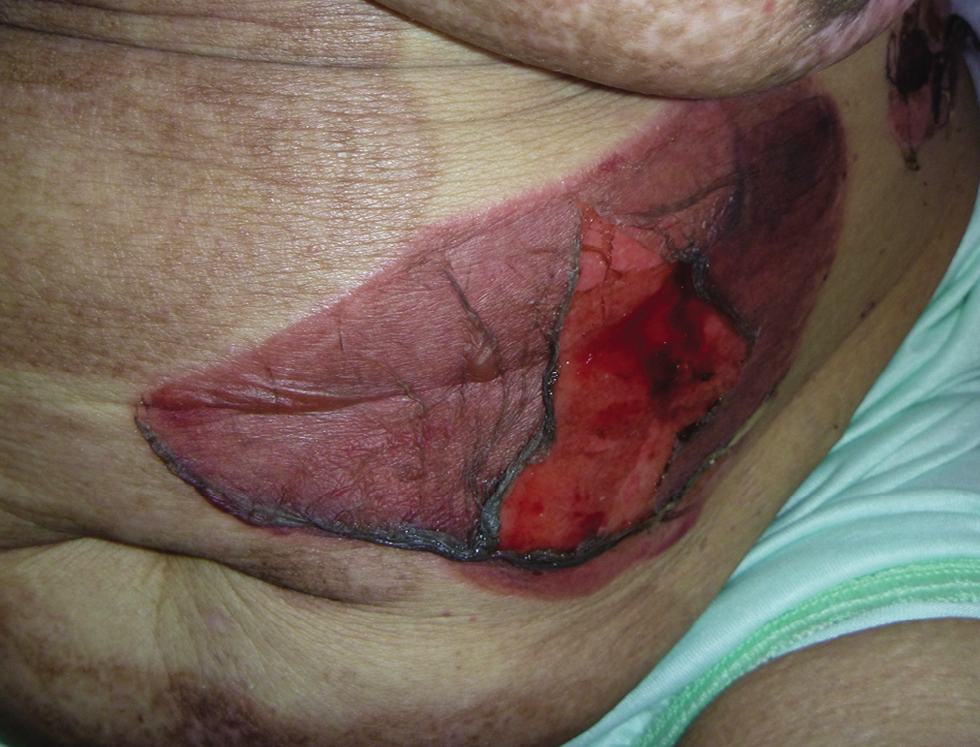
- Bullous fixed drug eruption Note the post-inflammatory hyperpigmentation affecting the abdominal skin from recurrent bullous eruptions
Drug-induced linear IgA bullous dermatosis (LABD):
Most cases of LABD arise spontaneously, but associations with drugs (most commonly vancomycin), infections, lymphoproliferative disorders and internal malignancies have been reported. The disease has a wide spectrum of clinical manifestations including a TEN-like presentation.59 DIF from perilesional skin is the main diagnostic tool for detecting the linear deposition of IgA along the basement membrane zone.60,61
Drug reaction with eosinophilia and systemic symptoms (DRESS):
Drug reaction with eosinophilia and systemic symptoms (DRESS) is a drug-induced delayed-type hypersensitivity reaction that includes skin eruption, haematologic abnormalities, lymphadenopathy and internal organ involvement.62 The clinical manifestations usually appear 2-8 weeks after drug intake63 adding to diagnostic difficulty. The most common drugs to cause this reaction can also induce TEN including anti-epileptics, allopurinol, and sulphonamides. The cutaneous manifestations [Figure 1d-f] typically consist of an urticarial, maculopapular eruption and, in some instances, vesicles, bullae, pustules, purpura, target lesions, facial oedema, cheilitis, erythroderma and TEN-like lesions.64,65 One important clinical finding in most patients is head and neck oedema which is often noticeable by looking at the ears.66 Mucous membrane involvement is very rare.62 Systemic involvement is the major cause of mortality in DRESS syndrome.67,68 Furthermore, the clinical features of DRESS may overlap with AGEP.69
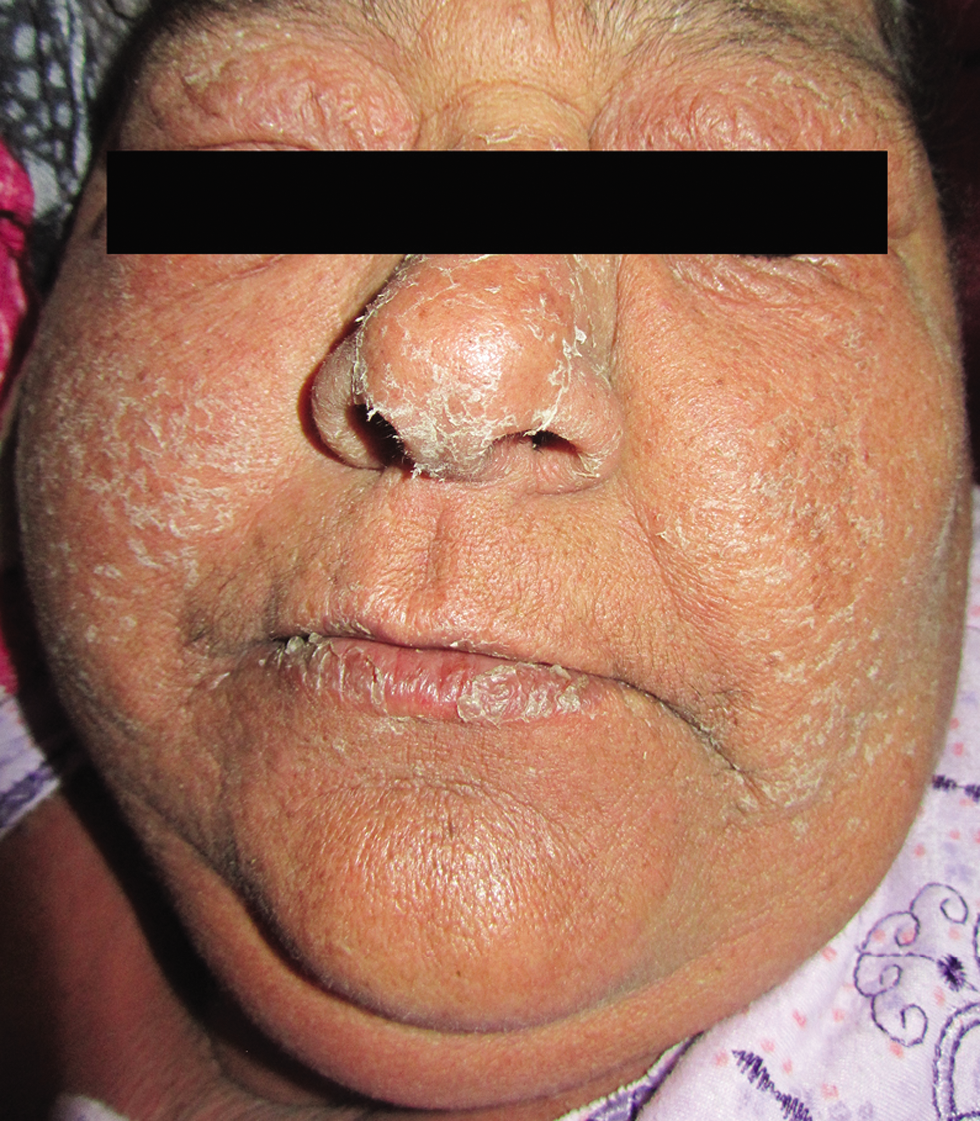
- Drug reaction with eosinophilia and systemic symptoms (DRESS). Note the marked facial oedema and erythroderma
- Drug reaction with eosinophilia and systemic symptoms (DRESS). TEN-like skin detachment
- Drug reaction with eosinophilia and systemic symptoms (DRESS). Resolution of facial oedema after treatment
The histopathological features of DRESS include spongiosis, a superficial perivascular lymphocytic infiltrate and an eosinophilic infiltrate in the dermis.69 Basal cell vacuolization and keratinocytes necrosis resembling EM may be found. Walsh et al. reported a correlation between those EM-like changes and severe liver dysfunction with higher mortality.70
Treatment consists of immediate withdrawal of all suspected medications, followed by careful monitoring and supportive care. Systemic corticosteroids are the first line of therapy, however; other immunosuppressive have been used.71
Contact dermatitis to dendrimers:
Dendrimers belong to a class of polymers that are recently used for drug delivery systems in nanotechnology. In 2008, Toyama et al. reported a severe case of contact dermatitis to dendrimers with a TEN-like presentation in a student who was handling dendrimers during some laboratory experiments. The skin biopsy showed confluent epidermal necrosis with partial eosinophilic degeneration and mononuclear cell infiltration at perivascular and subepidermal areas.72
Metabolic
Pseudoporphyria:
A TEN-like rash was reported in a patient on chronic haemodialysis who developed pseudoporphyria. Serum and vesicular fluid analysis using direct spectrofluorometry showed diagnostic findings of pseudoporphyria. Multiple skin biopsies revealed sub-epidermal blisters with an intact roof and a characteristic festooning of dermal papillae. Also, a Periodic Acid-Schiff (PAS) positive material was seen around the superficial blood vessels.73
Burn
Chemical:
Boric acid intoxication
Boric acid is an inorganic acid well absorbed across the gastric mucosa and abraded skin surfaces. It is colourless, odourless and water-soluble. The acid is used in wound irrigation solutions, eyewashes and mouthwashes. Toxicity mainly occurs in infants and children.74
Patients present with disturbed conscious levels, tachycardia, abnormal breathing sounds, blue-green vomitus, body stiffness, fever, and a skin rash.75 Within hours of ingestion, a diffuse erythematous “boiled lobster” desquamation occurs with a positive Nikolsky’s sign. Haemorrhagic lip crusts with intra-oral sparing and conjunctival oedema can be seen.74,76 Histopathologic examination reveals mainly an intact epidermis, but with alternating parakeratosis and orthokeratosis.74 There is no antidote to boric acid poisoning and only supportive care is recommended.77
Bleaching agents
In 2015, Totri et al. 78 reported a severe TEN-like presentation which was induced by a self-skin bleaching recipe of “betamethasone 0.05% ointment, hydrogen peroxide 12%, and potassium persulfate” combination. Furthermore, a skin biopsy revealed TEN-like pathology.
Thermal:
The early description of TEN syndrome was as a “burn-like” syndrome. Thermal burns can occur accidentally or intentionally as in the case of child abuse. Furthermore, thermal burn victims may develop later an attack of TEN. Clinically, patients with thermal burns present with painful, erythematous, blistering or deeply denuded skin with variable degrees of shock. Inhalation injury, carbon monoxide poisoning, upper airway swelling, stridor and respiratory failure are common in patients with fire-induced burns79. Furthermore, immersion burns of the buttocks and extremities are caused by dunking a child into scalding water. Immersion burns are usually sharply demarcated in a glove and stocking distribution. Sparing of the buttocks or “donut sign”80 suggests child abuse.81
Conclusion
Toxic epidermal necrolysis (TEN) is a serious dermatologic emergency. Many other dermatoses can present with TEN-like manifestations which is a critical diagnostic challenge. The proposed etiological classification and key differentiating points of all TEN-mimics would help our colleagues reach the correct diagnosis. Detailed history taking, careful skin and mucosal examination in addition to the proper selection of laboratory workup are mandatory to distinguish between TEN and TEN-mimics.
Abbreviations
TEN; toxic epidermal necrolysis
SJS; Stevens-Johnson syndrome
IVIG; intravenous immunoglobulin
SSSS; staphylococcal scalded skin syndrome
Dsg-1; desmoglein-1TSS; toxic shock syndrome
TSST-1; toxic shock syndrome toxin-1
DIC; disseminated intravascular coagulation
EBV; Epstein-Barr virus
EBER; Epstein-Barr virus EBV-encoded small RNA
PCR; polymerase chain reaction
ASAP; Acute Syndrome of Apoptotic Pan-Epidermolysis
SLE; systemic lupus erythematosus
SCLE; subacute cutaneous lupus erythematosus
IF; immunofluorescence
Ig; immunoglobulin
MDA5; anti-melanoma differentiation antigen-5
PV; pemphigus vulgaris
ELISA; enzyme-linked immunosorbent assay
PNP; paraneoplastic pemphigus
EP; envoplakin
PP; periplakin
IIF; indirect immunofluorescence
LPP; lichen planus pemphigoides
BP; bullous pemphigoid
LP; lichen planus
GVHD; graft versus host disease
EBA; epidermolysis bullosa acquisita
EBA-IP; epidermolysis bullosa acquisita-immunopathological
DH; dermatitis herpetiformis
DIF, direct immunofluorescence
BMZ; basement membrane zone
AGEP; acute generalised exanthematous pustulosis
FDE; fixed drug eruption
LABD; linear IgA bullous dermatosis
DRESS; drug reaction with eosinophilia and systemic symptoms
EM; erythema multiforme
PAS; Periodic acid-Schiff.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflict of interest
There are no conflicts of interest.
References
- Toxic epidermal necrolysis: Part I. Introduction, history, classification, clinical features, systemic manifestations, etiology, and immunopathogenesis. J Am Acad Dermatol. 2013;69:173.e1-13. quiz 185–6
- [CrossRef] [PubMed] [Google Scholar]
- Stevens-Johnson syndrome and toxic epidermal necrolysis: Assessment of medication risks with emphasis on recently marketed drugs. The Euro-SCAR-study. J Invest Dermatol. 2008;128:35-44.
- [CrossRef] [PubMed] [Google Scholar]
- Differential diagnosis of severe cutaneous drug eruptions. Am J Clin Dermatol. 2003;4:561-72.
- [CrossRef] [PubMed] [Google Scholar]
- The culprit drugs in 87 cases toxic epidermal necrolysis (Lyell’s syndrome) Arch Dermatol. 1987;123:1166-70.
- [PubMed] [Google Scholar]
- Clinical classification of cases of toxic epidermal necrolysis, Stevens-Johnson syndrome, and erythema multiforme. Arch Dermatol. 1993;129:92-6.
- [PubMed] [Google Scholar]
- UK guidelines for the management of Stevens Johnson syndrome/toxic epidermal necrolysis in adults 2016. J Plast Reconstr Aesthet Surg. 2016;69:e119-53.
- [CrossRef] [PubMed] [Google Scholar]
- Toxic epidermal necrolysis: Part II. Prognosis, sequelae, diagnosis, differential diagnosis, prevention, and treatment. J Am Acad Dermatol. 2013;69:187.e1-16. quiz 203–4
- [CrossRef] [PubMed] [Google Scholar]
- Staphylococcal scalded skin syndrome: Diagnosis and management in children and adults. J Eur Acad Dermatol Venereol. 2014;28:1418-23.
- [CrossRef] [PubMed] [Google Scholar]
- Tropical pyomyositis with staphylococcal scalded skin syndrome. Med J Armed Forces India. 2004;60:302-4.
- [CrossRef] [PubMed] [Google Scholar]
- Staphylococi, streptococci and the skin: Review of impetigo and staphylococcal scalded skin syndrome. Semin Dermatol. 1982;1:101-9.
- [Google Scholar]
- Staphylococcal scalded skin syndrome mimicking toxic epidermal necrolysis in a healthy adult. Our Dermatol Online. 2013;4:347-8.
- [CrossRef] [Google Scholar]
- Streptococcal toxic shock syndrome in the intensive care unit. Ann Intensive Care. 2018;8:88.
- [CrossRef] [PubMed] [Google Scholar]
- Toxic shock syndrome in the United States: Surveillance update, 1979-1996. Emerg Infect Dis. 1999;5:807-10.
- [CrossRef] [PubMed] [Google Scholar]
- Staphylococcal and streptococcal pyrogenic toxins involved in toxic shock syndrome and related illnesses. Crit Rev Microbiol. 1990;17:251-72.
- [CrossRef] [PubMed] [Google Scholar]
- Understanding toxic shock syndrome. Intensive Care Med. 2015;41:1707-10.
- [CrossRef] [PubMed] [Google Scholar]
- The use of etanercept for treatment of toxic epidermal necrolysis when toxic shock syndrome is in the differential. Dermatol Ther. 2018;31:e12684.
- [CrossRef] [PubMed] [Google Scholar]
- Purpura fulminans mimicking toxic epidermal necrolysis-Additional value of 16S rRNA sequencing and skin biopsy. Neth J Med. 2017;75:165-8.
- [PubMed] [Google Scholar]
- Purpura fulminans: Recognition, diagnosis and management. Arch Dis Child. 2011;96:1066-71.
- [CrossRef] [PubMed] [Google Scholar]
- Purpura fulminans in toxic epidermal necrolysis: Case report and review. J Burn Care Res. 2015;36:e274-82.
- [CrossRef] [PubMed] [Google Scholar]
- Coxsackievirus B5-induced severe mucocutaneous reaction mimicking drug-induced Stevens Johnson syndrome/toxic epidermal necrolysis. Indian J Dermatol Venereol Leprol. 2019;85:416-8.
- [CrossRef] [PubMed] [Google Scholar]
- Atypical hand, foot and mouth disease in adults associated with coxsackievirus A6: A clinico-pathologic study. J Cutan Pathol. 2016;43:940-5.
- [CrossRef] [PubMed] [Google Scholar]
- Epidermal necrolysis (Stevens-Johnson Syndrome and toxic epidermal necrolysis) In: Goldsmith LA, Katz SI, Gilchrest BA, Paller AS, Leffell DJ, Wolff K, eds. Fitzpatrick’s Dermatology in General Medicine. Vol 8. New York: The McGraw-Hill Companies; 2012. Ch. 40
- [Google Scholar]
- Purpuric macules with vesiculobullous lesions a novel manifestation of Chikungunya. Int J Dermatol. 2011;50:61-9.
- [CrossRef] [PubMed] [Google Scholar]
- Stevens Johnson syndrome and toxic epidermal necrolysis like cutaneous presentation of chikungunya fever: A case series. Pediatr Dermatol. 2018;35:392-6.
- [CrossRef] [PubMed] [Google Scholar]
- Chikungunya fever in travelers returning to Europe from the Indian Ocean region, 2006. Emerg Infect Dis. 2008;14:416-22.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- TEN-like eruption in setting of EBV positive T-cell lymphoproliferative disease with HLH, in a child. Australas J Dermatol. 2014;55:e44-7.
- [CrossRef] [PubMed] [Google Scholar]
- Epidermal cell necrosis with direct epidermal infiltration of Epstein-Barr virus (EBV)—encoded small nuclear RNA-positive T lymphocytes in a patient with EBV-associated haemophagocytic syndrome. Br J Dermatol. 2007;157:1053-6.
- [CrossRef] [PubMed] [Google Scholar]
- Toxic epidermal necrolysis-like acute cutaneous lupus erythematosus and the spectrum of the acute syndrome of apoptotic pan-epidermolysis (ASAP): A case report, concept review and proposal for new classification of lupus erythematosus vesiculobullous skin lesions. Lupus. 2004;13:941-50.
- [CrossRef] [PubMed] [Google Scholar]
- The lexicon of cutaneous lupus erythematosus-A review and personal perspective on the nomenclature and classification of the cutaneous manifestations of lupus erythematosus. Lupus. 1997;6:84-95.
- [CrossRef] [PubMed] [Google Scholar]
- Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum. 2012;64:2677-86.
- [CrossRef] [PubMed] [Google Scholar]
- Toxic epidermal necrolysis-like rash of lupus: A dermatologist’s dilemma. Indian J Dermatol. 2014;59:401-2.
- [CrossRef] [PubMed] [Google Scholar]
- Stevens-Johnson syndrome and toxic epidermal necrolysis-like lupus erythematosus. J Clin Rheumatol. 2019;25:224-31.
- [CrossRef] [PubMed] [Google Scholar]
- Toxic epidermal necrolysis-like acute cutaneous lupus erythematosus (TEN-like ACLE) in SLE patients: A report of two cases. Asian Pac J Allergy Immunol. 2012;30:83-7.
- [PubMed] [Google Scholar]
- Toxic epidermal necrolysis-like acute cutaneous lupus erythematosus: Report of a case and review of the literature. Dermatol Online J. 2018;24:13030/qt5r79d67k.
- [PubMed] [Google Scholar]
- Toxic epidermal necrolysis-like dermatomyositis associated with antimelanoma differentiation antigen 5. JAAD Case Rep. 2019;5:91-3.
- [CrossRef] [PubMed] [Google Scholar]
- An IFN-associated cytotoxic cellular immune response against viral, self-or tumor antigens is a common pathogenetic feature in ‘‘interface dermatitis.’. J Invest Dermatol. 2008;128:2392-402.
- [CrossRef] [PubMed] [Google Scholar]
- Activated STING in a vascular and pulmonary syndrome. N Engl J Med. 2014;371:507-18.
- [CrossRef] [PubMed] [Google Scholar]
- Rare appearance of toxic epidermal necrolysis-like histopathological features in a case of atypical pemphigus with serological pemphigus vulgaris character. J Dermatol. 2013;40:63-4.
- [CrossRef] [PubMed] [Google Scholar]
- Classification, clinical manifestations, and immunopathological mechanisms of the epithelial variant of paraneoplastic autoimmune multiorgan syndrome: A reappraisal of paraneoplastic pemphigus. Arch Dermatol. 2001;137:193-206.
- [PubMed] [Google Scholar]
- Paraneoplastic pemphigus mimicking toxic epidermal necrolysis: An underdiagnosed entity? JAAD Case Rep. 2018;4:67-71.
- [CrossRef] [PubMed] [Google Scholar]
- Sensitivity and specificity of clinical, histologic, and immunologic features in the diagnosis of paraneoplastic pemphigus. J Am Acad Dermatol. 2000;43:619-26.
- [CrossRef] [PubMed] [Google Scholar]
- Sera from patients with toxic epidermal necrolysis contain autoantibodies to periplakin. Br J Dermatol. 2006;155:337-43.
- [CrossRef] [PubMed] [Google Scholar]
- Lichen planus pemphigoides: Two case reports. Clin Exp Dermatol. 1998;23:132-5.
- [CrossRef] [PubMed] [Google Scholar]
- Lichen planus pemphigoides mimicking toxic epidermal necrolysis. Am J Dermatopathol. 2019;41:e144-7.
- [CrossRef] [PubMed] [Google Scholar]
- Acute graft-versus-host disease: From the bench to the bedside. Blood. 2009;114:4327-36.
- [CrossRef] [PubMed] [Google Scholar]
- Many faces of graft-versus-host disease. Australas J Dermatol. 2010;51:1-10; quiz 11.
- [CrossRef] [PubMed] [Google Scholar]
- Skin biopsy in allogeneic and autologous bone marrow transplant patients: A histologic and immunohistochemical study and review of the literature. Mod Pathol. 1995;8:59-64.
- [PubMed] [Google Scholar]
- Gastrointestinal graft-versus-host disease in man. A clinicopathologic study of the rectal biopsy. Am J Surg Pathol. 1979;3:291-9.
- [CrossRef] [PubMed] [Google Scholar]
- Acute graft versus host disease. Orphanet J Rare Dis. 2007;2:35.
- [CrossRef] [PubMed] [Google Scholar]
- Epidermolysis bullosa acquisita of the immunopathological type (dermolytic pemphigoid) J Invest Dermatol. 1985;85:79s-84s.
- [CrossRef] [PubMed] [Google Scholar]
- Inflammatory epidermolysis bullosa acquisita mimicking toxic epidermal necrolysis and dermatitis herpetiformis. Clin Exp Dermatol. 2009;34:e705-8.
- [CrossRef] [PubMed] [Google Scholar]
- Acute generalized exanthematous pustulosis simulating toxic epidermal necrolysis: A case report and review of the literature. Arch Dermatol. 2011;147:697-701.
- [CrossRef] [PubMed] [Google Scholar]
- Severe flucloxacillin-induced acute generalized exanthematous pustulosis (AGEP), with toxic epidermal necrolysis (TEN)—like features: Does overlap between AGEP and TEN exist? Clinical report and review of the literature. Br J Dermatol. 2014;171:1539-45.
- [CrossRef] [PubMed] [Google Scholar]
- Acute generalized exanthematous pustulosis (AGEP)-A clinical reaction pattern. J Cutan Pathol. 2001;28:113-9.
- [CrossRef] [PubMed] [Google Scholar]
- History and clinical significance of mechanical symptoms in blistering dermatoses: A reappraisal. J Am Acad Dermatol. 2003;48:86-92.
- [CrossRef] [PubMed] [Google Scholar]
- Textbook of Dermatology (8th ed). United Kingdom: Wiley-Blackwell Publisher (P) Ltd; 2010. p. :75.28-75.29.
- Generalized bullous fixed drug eruption mimicking toxic epidermal necrolysis caused by paracetamol. Clinical Dermatology Review. 2018;2:34-7.
- [CrossRef] [Google Scholar]
- Doxycycline induced generalized bullous fixed drug eruption. Indian J Dermatol. 2016;61:128.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- A toxic epidermal necrolysis-like presentation of linear IgA bullous dermatosis treated with dapsone. Dermatol Online J. 2017;23:13030/qt4443157h.
- [PubMed] [Google Scholar]
- IgA linear dermatosis of childhood (chronic bullous disease of childhood) Br J Dermatol. 1979;101:535-42.
- [CrossRef] [PubMed] [Google Scholar]
- Linear IgA bullous disease presenting as toxic epidermal necrolysis: A case report and review of the literature. Dermatology. 2013;227:209-13.
- [CrossRef] [PubMed] [Google Scholar]
- Drug reaction with eosinophilia and systemic symptoms (DRESS): An original multisystem adverse drug reaction. Results from the prospective RegiSCAR study. Br J Dermatol. 2013;169:1071-80.
- [CrossRef] [PubMed] [Google Scholar]
- Risk of Stevens-Johnson syndrome and toxic epidermal necrolysis during first weeks of antiepileptic therapy: A case-control study. Study Group of the International Case Control Study on Severe Cutaneous Adverse Reactions. Lancet. 1999;353:2190-4.
- [CrossRef] [PubMed] [Google Scholar]
- Twelve-year analysis of severe cases of drug reaction with eosinophilia and systemic symptoms: A cause of unpredictable multiorgan failure. Arch Dermatol. 2009;145:67-72.
- [CrossRef] [PubMed] [Google Scholar]
- Variability in the clinical pattern of cutaneous side-effects of drugs with systemic symptoms: Does a DRESS syndrome really exist? Br J Dermatol. 2006;155:422-8.
- [CrossRef] [PubMed] [Google Scholar]
- Drug reaction with eosinophilia and systemic symptoms (DRESS): A clinical update and review of current thinking. Clin Exp Dermatol. 2011;36:6-11.
- [CrossRef] [PubMed] [Google Scholar]
- Colitis may be part of the antiepileptic drug hypersensitivity syndrome. Epilepsia. 1999;40:1780-3.
- [CrossRef] [PubMed] [Google Scholar]
- Longterm sequelae of drug reaction with eosinophilia and systemic symptoms: A retrospective cohort study from Taiwan. J Am Acad Dermatol. 2013;68:459-65.
- [CrossRef] [PubMed] [Google Scholar]
- Severe cutaneous drug reactions: Do overlapping forms exist? Actas Dermosifiliogr. 2016;107:23-33.
- [CrossRef] [PubMed] [Google Scholar]
- Clinicopathological features and prognosis of drug rash with eosinophilia and systemic symptoms: A study of 30 cases in Taiwan. J Eur Acad Dermatol Venereol. 2008;22:1044-9.
- [CrossRef] [PubMed] [Google Scholar]
- Drug reaction with eosinophilia and systemic symptoms: Is cutaneous phenotype a prognostic marker for outcome? A review of clinicopathological features of 27 cases. Br J Dermatol. 2013;168:391-401.
- [CrossRef] [PubMed] [Google Scholar]
- Drug reaction with eosinophilia and systemic symptoms (DRESS): An interplay among drugs, viruses, and immune system. Int J Mol Sci. 2017;18:1243.
- [CrossRef] [PubMed] [Google Scholar]
- A case of toxic epidermal necrolysis-like dermatitis evolving from contact dermatitis of the hands associated with exposure to dendrimers. Contact Dermatitis. 2008;59:122-3.
- [CrossRef] [PubMed] [Google Scholar]
- Pseudoporphyria: An atypical variant resembling toxic epidermal necrolysis. J Cutan Med Surg. 2001;5:479-85.
- [CrossRef] [PubMed] [Google Scholar]
- Boric acid ingestion clinically mimicking toxic epidermal necrolysis. J Cutan Pathol. 2013;40:962-5.
- [CrossRef] [PubMed] [Google Scholar]
- Ellenhorn’s medical toxicology: Diagnosis and treatment of human poisoning. (2nd). Baltimore MA: Williams & Wilkins; 1997.
- [Google Scholar]
- ‘‘Boiled Lobster’’ rash of acute boric acid toxicity. Clin Toxicol (Philia). 2009;47:432.
- [CrossRef] [PubMed] [Google Scholar]
- Successful treatment of a rare case of boric acid overdose with hemodialysis. Am J Kidney Dis. 2006;48:e95-7.
- [CrossRef] [PubMed] [Google Scholar]
- Severe chemical burns mimicking toxic epidermal necrolysis in an adolescent female following a self-skin bleaching protocol. JAAD. 2015;72:AB203.
- [CrossRef] [Google Scholar]
- Toxic epidermal necrolysis after acute burn injury. Ann Burns Fire Disasters. 2018;31:266-70.
- [PubMed] [Google Scholar]
- Book: Rook's Textbook of Dermatology (Seventh edition). Oxford: Blackwell Science Ltd Oxford; 2004.
- [Google Scholar]






